By Kevin E. Noonan —
Bans on gene patenting and the possibility of
having a court, Congress, or czar declare them banned are all the rage recently,
spurred in part by the lawsuit against Myriad Genetics and the University of
Utah (among other defendants) in the Federal District Court for the Southern
District of New York (see "Association for Molecular Pathology v. U.S. Patent and Trademark Office"). Whether that lawsuit will go forward is
expected to be decided by the end of the month, when a decision on standing for
many of the plaintiffs is expected from the District Court. No matter how the judge rules, however, interests groups
like the American Civil Liberties Union are unlikely to refrain from efforts to
have gene patenting banned.
Under the circumstances, it is reasonable to wonder
whether gene patents are in fact inhibiting innovation or otherwise harming
patients, physicians, or researchers (since the existential harm asserted by
those against gene patents — that it is fundamentally "wrong" — is
harder to assess quantitatively). The latest report from academia on the question is consistent with
several other reports refuting negative effects predicted on theoretical
grounds (such as the "tragedy of the anticommons" idea). Indeed, the report's conclusions are that the
greatest difficulty surrounding gene patents arises from the legal
uncertainties created in part by efforts to ban gene patenting.
 The report, from Isabelle Huys, Nele Berthels, Gert Matthijs & Geertrui Van
The report, from Isabelle Huys, Nele Berthels, Gert Matthijs & Geertrui Van
Overwalle, researchers at the Catholic University of Leuven,
Belgium, is found in the October 2009 edition of Nature Biotechnology (see "Legal uncertainty in the area of genetic diagnostic testing"). The authors' express intent was to assess whether gene patents, either
as "blocking patents" or as a "patent thicket," were
inhibiting genetic technology. The
concern stemmed from the belief that such patents would be "difficult or
impossible to circumvent." Their research is current as of February 6, 2009 (and thus, it does not
reference the ACLU-backed lawsuit filed earlier this year).
Defined as a "large-scale empirical study,"
the authors describe the following "heart of the problem: which types of claims occur in
disease-specific patents and to what extent are these claims essential for
carrying out genetic diagnostic tests?"
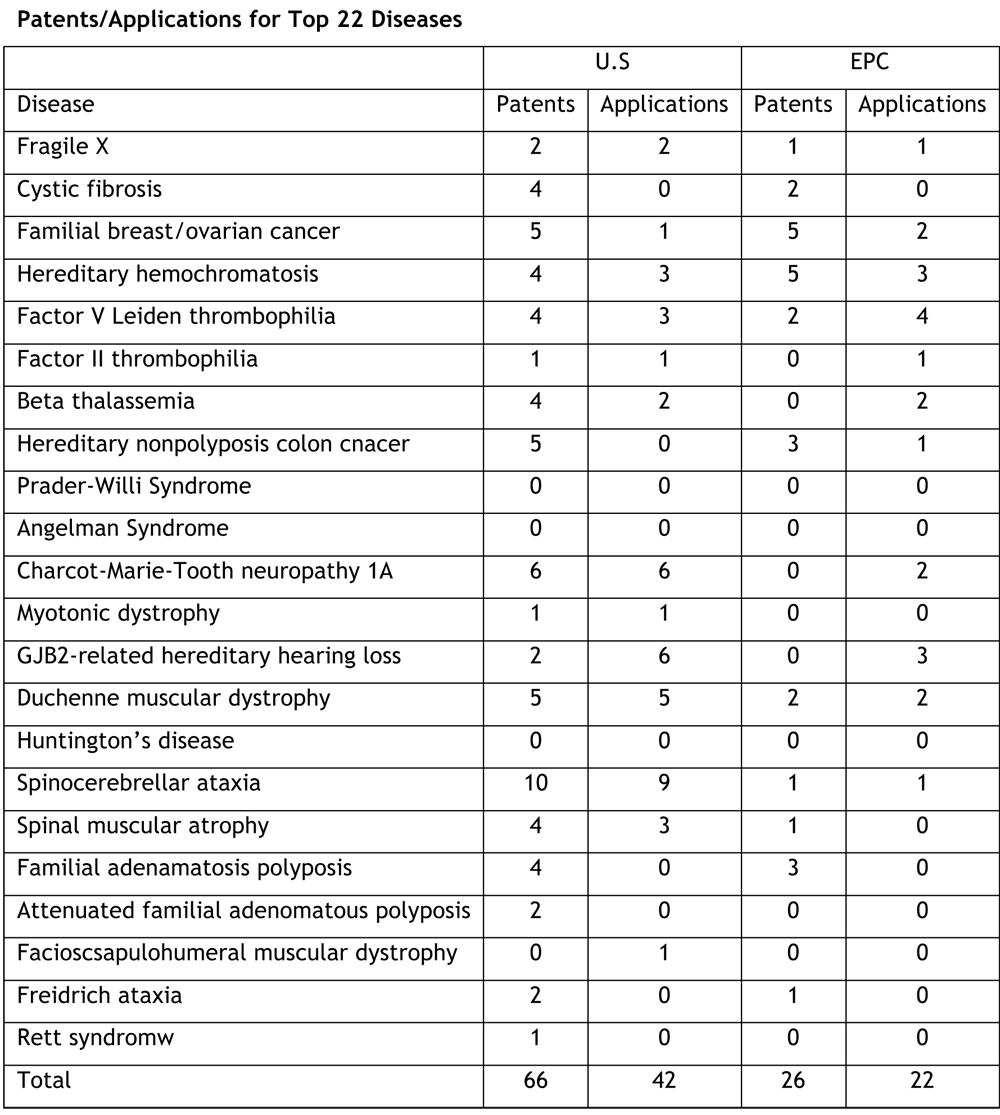
The sample set was U.S. and European (EP) patents
and patent applications directed at twenty-two inherited, monogenic diseases (see Table 1 below; click on table to enlarge). The legal
status of the patent documents reviewed included patent applications, granted
patents, patent applications "dropped" (i.e., abandoned during
prosecution) and abandoned, withdrawn, revoked, or expired patents. 267 claims from 145 patents were
assessed, with regard to whether they were "easy," "difficult,"
or almost "impossible" to circumvent, based on comparing the claimed
methods to the "best practices" guidelines for genetic susceptibility
screening; claims deemed "impossible" to circumvent were
characterized as "blocking" claims. The claims studied were independent claims for genes,
oligonucleotides, methods, and kits. However, they did not review the file history nor does the report compare
the commercial embodiments with the claims or their licensing status. Finally, U.S. claims were assessed with
regard to the "written description," "enablement," and "best
mode" requirements, and the European patents with regard to EPC Art. 83
and 69.
The patents and applications (250 patent documents)
were grouped into 72 different patent families (where a "family" is "a
group of patents or applications ('patent documents') taken in
various countries to protect a single invention"). U.S. patents were found in 66
families whereas EP patents were found in only 26 families. Twenty-one families had both U.S. and EP
patents, while 46 families had only U.S. patents and only 5 families had EP but
not U.S. patents. There were 42
patent families containing U.S. applications versus 22 families with EP
applications pending. "Remarkably," according to the authors, "about
twice as many patent applications are dropped during the patent prosecution
procedure in Europe (34) in EP than in US (16)." Also, in Europe there were about the same number of granted
patents (26) as pending applications (22), whereas in the U.S. there are 66
granted patents and 42 pending applications.
There were 56 different applicants for these
patents/applications, with 62.5% of the applicants coming from non-profit
sector, predominantly universities and research organizations, 58.9% of applicants from the U.S., 25%
from EP, and 16.1% from JP or CA, with U.S. applicants owning 55.8% of the EP
patents and 67% of the U.S. patents. There were also 11 different co-ownerships with 17 different partners. The top applicants from the U.S. were
Johns Hopkins University (6 patent families) and Baylor College of Medicine (6
families); the top European applicant was Leiden University (3 patent
families).
The vast majority of genetic diseases (19/22) have
patented tests (Huntington's disease a surprising exception); the status of the
diseases and the patents or applications relating to each are shown in Table 2 below (click on table to enlarge). Four diseases were covered by
granted patents in Europe, while six diseases have U.S. patent coverage.
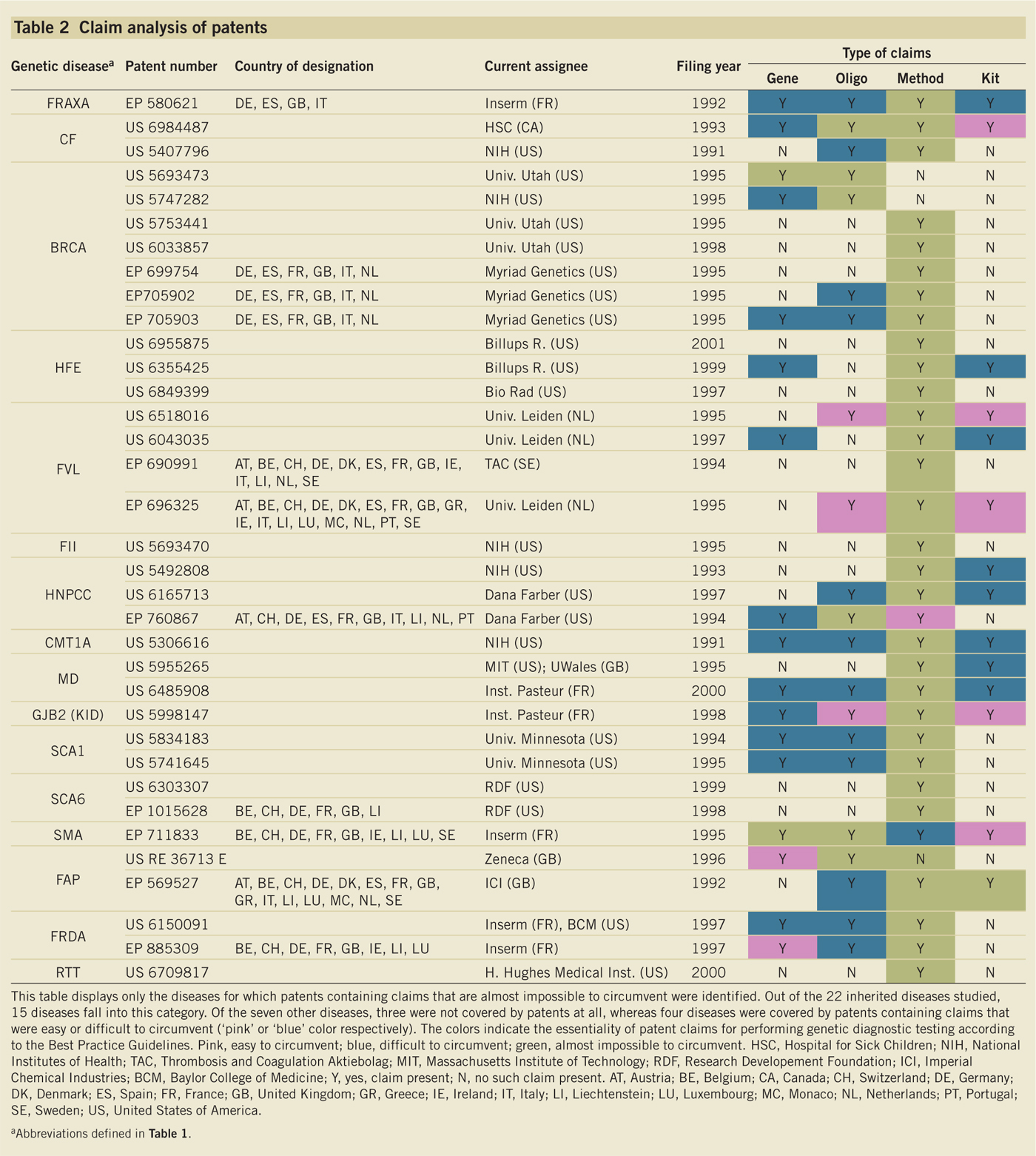
(Huys et al., 2009, Nature Biotechnology 27: 903-909, Table 2, p. 907).
The "[m]ost
heavily patented [diseases] in Europe are hereditary hemochromatosis and
familial breast cancer testing," while in the U.S., "most patents are
for spinocerebellar ataxia, Charcot-Marie-Tooth neuropathy, hereditary
nonpolyposis colon cancer and familial breast and ovary cancer."
145 granted patents were reviewed (118 U.S. patents
and 27 EP patents) and 267 claims from these patents characterized: of these 38% were method claims, 25%
gene claims, 23% oligonucleotide/probe claims, and 14% kit claims. For the patents having claims to genes
(66/145), the majority were claims for cDNA copies of genes (although there
were 22 patents having claims to genomic DNA embodiments of the claimed
genes). When assessed under the "easy,"
"difficult," or "impossible" (i.e., "blocking")
standard, only 3% of these gene claims were "blocking." Accordingly, the authors concluded that
these claims did not represent a "patent thicket" or otherwise
provide an impediment to innovation.
For method claims, on the other hand, 30%
impossible, 47% difficult, and 23% easy to circumvent, and 35/145 patents
contained at least one blocking method claim.
Claim 1 of U.S. Patent No. 5,693,470 was recited as
exemplary of a "blocking" method claim (for hereditary nonpolyposis colon cancer):
1. A
method of determining a predisposition to cancer comprising:
testing a body sample of
a human to ascertain the presence of a mutation in a gene identified as hMSH2
(human analog of bacterial MutS and Saccharomyces cerevisine MSH2) which
affects hMSH2 expression or hMSH2 protein function, the presence of such a
mutation indicating a predisposition to cancer.
60% of patent families (21/35) had no kit claims,
and 15/22 diseases have claims impossible to circumvent.
The authors reported a number of observations that
they characterized as "surprising." These included the supremacy of U.S. patents on genetic
inventions persists, almost 30 years after Diamond
v. Chakrabarty. They also
noted that, despite the requirement in the U.S. since 2001 that gene patents
can be obtained in the U.S. only if "biochemical, biological, or genetic
data describing the function are included" (they reported 63 such
patents), 11 "key" patents were directed to genes without this
information. The situation is
different in Europe, with many more applications abandoned during prosecution
or otherwise not pursued. The
authors speculate that this many be due "perhaps" to a more
restrictive and expensive patent system. The authors also noted that 60% of applicants were U.S. universities
and research institutes, due to the "progressive attitude" of the U.S.
for universities to be able to patent their inventions.
With regard to the claim analysis, the data clearly
showed no evidence of a patent thicket for gene claims. The situation is different for
diagnostic method claims, with most of these claims being difficult or
impossible to circumvent. Because
the authors found there to exist claims to the same disease owned by different
applicants, they noted the possibility that a patent thicket could exist but
provide no evidence for any. They
also noted that for method claims, there was a failure to link the biological
observation to specific method steps, which may make it difficult to find
infringement for these claims. (For example, a claim reciting an isolated cDNA encoding a human gene is
unlikely to be infringed by a test for identifying a mutation in DNA from a
human sample.)
The authors present these conclusions:
In conclusion,
the present analysis and accompanying observations do not point to the
existence of a wide patent thicket in genetic diagnostic testing. Rather, they
highlight a problem of lack of transparency and clarity, leading to legal
uncertainty. Neither case law nor patent legislation resolves the legal
uncertainty related to patents on genetic inventions. In 2006, the US Supreme
Court dismissed a judicial review on the case Laboratory Corporation of
America Holdings v. Metabolite Laboratories Inc. . . . As a substantial
number of patent claims would have been affected by a decision in this case,
this refusal further increased legal
uncertainty, including in the genetic field. The consequence of this high
level of legal uncertainty is that either enormous
risks are taken if genetic tests are performed without knowingly infringing
a specific patent, or much time and energy goes into establishing patent
landscapes and freedom-to-operate analyses or to efforts to use different
techniques and methods that may eventually be below the state of the art that
is clinically requested.As this study shows that not that many blocking
gene patents exist, proposals aiming at banning patents on human
genes do not provide a plausible solution, unless the ban would be on
patents for broad genetic diagnostic
methods as such. For instance, the European Society of Human Genetics (Vienna)
has recently recommended avoiding patenting of the pure link between a mutation
and disease. More attention should
be paid to the licensing practices in a 'responsible' way. Otherwise, the risk exists that the control by owners of patents containing
those broad claims with respect to genetic diagnostic testing may in the future dissociate actual
genetic diagnostic testing from genetic counseling and clinical investigation, which
is to the detriment of progress of the genetic diagnostic service and public healthcare
system.(Emphasis added)
These conclusions reflect two realities. The first is that researchers have
identified practices that may, in the future, lead to negative
outcomes, but have not detected any evidence of such outcomes using current
data. The second is that the legal
uncertainty occasioned by "decisions" such as Justice Breyer's
dissent in the LabCorp case have
consequences for research, investment, and healthcare. Judge Rader may note, in the Prometheus decision, that this dissent
is "not controlling law," but these sentiments, combined with the
risk of a gene patenting ban, are (according to these authors) on balance
having an effect more negative on innovation than the gene patents that are at
risk.