 By Kevin E. Noonan —
By Kevin E. Noonan —
On July 15th, Junior Party the University of California/Berkeley, the University of Vienna, and Emmanuelle Charpentier (collectively, "CVC") filed its Opposition to Senior Party ToolGen's Substantive Motion No. 1 for benefit of priority to U.S. Provisional Application No. 61/837,481, filed June 20, 2013 ("P3" or "ToolGen P3"), or alternatively, International Application No. PCT/KR2013/009488, filed Oct. 23, 2013 ("PCT"), in Interference No. 106,127.
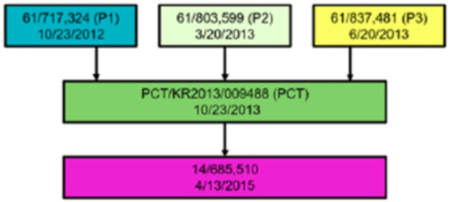
In its motion, ToolGen set out graphically the relationship of these priority documents, including U.S. provisional application No. 61/717,324 ("P1") for which the Board had granted ToolGen priority upon institution:
The basis ToolGen asserted for its priority claim was satisfaction of the written description and enablement requirements under 35 U.S.C. § 112(a) with regard to two embodiments falling within the scope of the Interference Count. ToolGen argued satisfaction of the alternative language of the Count that recites claim 18 (dependent on claim 15) of Broad's U.S. Patent No. 8,697,359, the brief annotating the limitation recited therein to facilitate identification of ToolGen's disclosure corresponding thereto:
[1] An engineered, programmable, non-naturally occurring Type II CRISPR-Cas system comprising
[2] a Cas9 protein and
[3] at least one guide RNA that targets and hybridizes to a target sequence of a DNA molecule in a eukaryotic cell,
[4] wherein the DNA molecule encodes and the eukaryotic cell expresses at least one gene product and
[5] the Cas9 protein cleaves the DNA molecules,
[6] whereby expression of the at least one gene product is altered; and,
[7] wherein the Cas9 protein and the guide RNA do not naturally occur together;
[8] wherein the guide RNAs comprise a guide sequence fused to a tracr sequence.
The brief asserted Examples 3 and 4 of the P3 (PCT) priority document in support of its satisfaction arguments, noting that one such embodiment (designated 3-1) comprises a Foxn1-specific sgRNA and a Cas9 mRNA, while embodiment 3-2 comprises the same sgRNA and recombinant Cas9 protein. And in each case, ToolGen argued the Examples illustrate CRISPR-mediated cleavage and editing of the target Foxn1 DNA in mouse embryos expressed in the resultant genetically engineered mice. The CRISPR-Cas9 complex is illustrated in the brief by this drawing:

wherein "target DNA [is] in the green box, DNA-targeting sequence of crRNA [is] in the blue box, crRNA:tracrRNA duplex linked together by a -GAAA- loop [is] in the red box, remaining tracrRNA portion shown with brown underline, Cas9 protein with label shown with purple underline depicted as a shaded oval, which is in complex with sgRNA and cleaves the target sequence in the target DNA."
The brief also noted the portions of the P3 priority document showing that such CRISPR-Cas9 complexes successfully cleaved and edited the target Foxn1 DNA, which ToolGen argued provided extensive disclosure in the P3 priority document of "abundant working examples and considerable guidance to a [person of ordinary skill in the art] to make and use CRISPR/Cas9 in eukaryotic cells." ToolGen also noted that the P3 priority document shares the same specification with U.S. Application No. 14/685,510, to which the Board has recognized ToolGen's entitlement to priority.
CVC disagrees in its opposition brief. CVC's argument tracks much of the argument made in CVC's Substantive Preliminary Motion No. 2 that the Board deny ToolGen benefit of priority to it P1 provisional application (see "CVC Substantive Preliminary Motion No. 2 to Deny Priority Benefit"). In that argument, CVC maintained that the Board should deny ToolGen priority benefit to the '324 application based on party admissions because this provisional application does not disclose an operative embodiment falling within the scope of the interference Count. Specifically, CVC argued that ToolGen in the prosecution of the '324 patent application leading to allowance (and declaration of this interference) had argued to the Patent Examiner (and PTAB) that "a codon-optimized Cas9 nucleic acid is required for CRISPR-Cas9 to function in eukaryotic cells" and that "a skilled artisan would have no idea what the outcome may be if one were to codon optimize a Cas9 nucleic acid" (and with the additional argument here that ToolGen's P3 and PCT applications do not disclose Cas9 embodiments comprising a nuclear localization signal, either, wherein both of these alterations CVC alleges ToolGen represented as the "secret sauce" before the Board during ex parte prosecution). This position was consistent with the prokaryotic source of Cas9, and the Board and Examiner relied upon these arguments to find allowable claims that are now claims designated as corresponding to the Count in this interference, CVC asserts. All such claims require use of a Cas9-encoding nucleic acid that is codon-optimized for expression in eukaryotic cells, and ToolGen added this limitation to claims-in-interference to overcome anticipation and obviousness rejections based on the prior art. But neither of ToolGen's '481 provisional application nor its PCT application discloses a codon-optimized Cas9 nucleic acid, according to CVC, nor by ToolGen's own argument (unpredictability) would the skilled worker be able to discern such a nucleic acid with any reasonable basis for expecting such an embodiment could be produced using the disclosure in either the '481 provisional application or its PCT application. Accordingly, CVC argues in its motion, ToolGen cannot in this interference renounce these arguments and rely on the priority date of the either the '481 provisional application or its PCT application to constitute a constructive reduction to practice for eukaryotic CRISPR-Cas9 embodiments falling within the scope of the interference Count. And mere "generalized language" relating to these modifications are inadequate, CVC argues here, pursuant to University of Rochester v. G.D. Searle & Co., 358 F.3d 916, 923 (Fed. Cir. 2004). Thus, according to CVC, the Board should deny ToolGen priority benefit to the '481 provisional application or its PCT application.
CVC also takes issue with ToolGen's alleged argument that its disclosure satisfies CVC's half of the count (which doesn't recite these modifications) saying "the issue is what embodiments ToolGen may rely on given that it unambiguously told the Patent Office that NLS addition and codon optimization are required and unpredictable." And the disclosure of the P3 provisional and PCT application to which ToolGen seeks to claim priority benefit do not pass muster under this precedent according to CVC.
Finally, CVC argues that "Principles of party admission and judicial estoppel preclude ToolGen from taking positions inconsistent with its prior assertions," citing Zedner v. United States, 547 U.S. 489 (2006), and Louis v. Okada, 59 U.S.P.Q.2d 1073, 1075 (B.P.A.I. 2001) (precedential).
The brief then points out with particularity ToolGen's arguments in its motion, pursuant to PTAB procedural rules in interferences, in a "call and response" format and hies back to its argument and the structure thereof in CVC's own motion to deny ToolGen benefit of priority to its '428 provisional application. These arguments are all tied to CVC's assertion that "ToolGen's motion fails to show that its P3 and PCT provide a [constructive reduction to practice] of CVC's half of the count because neither application describes an embodiment with an NLS-tagged and codon-optimized Cas9 consistent with its prosecution representations," based on the priority benefit requirements as set forth inter alia in Falkner v. Inglis, 448 F.3d 1357, 1362 (Fed. Cir. 2006), and 37 C.F.R. § 41.201. CVC's arguments further sound in familiar written description precedent (University of Rochester v. G.D. Searle & Co; Ariad Pharm., Inc. v. Eli Lilly & Co., 598 F.3d 1336, 1351 (Fed. Cir. 2010) (en banc); In re Alonso, 545 F.3d 1015, 1021 (Fed. Cir. 2008)) as applied to the description contained in the specification of ToolGen's P3 and PCT applications. Specifically, CVC's brief focuses on Embodiment 3-1 and Embodiment 3-2 cited by ToolGen in support of its motion for priority benefit, each of which comprise a codon-optimized Cas9 protein having an NLS tagged therewith. In particular, CVC argues that the P3 provisional application has no disclosure of a codon-optimized Cas9 sequence and lacks a formal sequence listing pursuant to 37 C.F.R. § 1.821 (despite the specification making reference to "SEQ ID NO:2" as an amino acid sequence encoding Cas9). Moreover, the absence of this disclosure was conceded by ToolGen's expert according to CVC. In addition, CVC argues that:
P3 lacks the following disclosures that a skilled artisan would otherwise need to understand that ToolGen possessed a Cas9 mRNA of the count: (1) the bacterial Cas9 species from which the Cas9 mRNA was derived, (2) whether the Cas9 mRNA was codon-optimized or encoded an NLS-tagged Cas9 protein, (3) which NLS, if any, was attached; or (4) which Cas9 mRNA sequence was injected into the mouse cells,
and ToolGen's expert merely cobbled together "disparate statements from other portions of the specification" in an attempt to establish sufficiency of disclosure. (Perhaps infelicitously, ToolGen relied on the same expert to support its arguments before the Board during ex parte examination.) CVC further argues that at the time the invention was made there were many codon optimization tables known in the art and that they differed with regard to how a bacterial protein like Cas9 would be effectively codon-optimized for expression un eukaryotic cells (and CVC notes that P3 does not inform the skilled worker which table or program should be used). CVC asserts similar arguments with regard to choice of NLS tags known in the art. According to CVC both embodiments have these flaws fatal to satisfaction of the requirements for a constructive reduction to practice.
CVC asserts similar deficiencies in the disclosure of ToolGen's PCT application; in both arguments CVC focuses on element [2] in ToolGen's explication of Claim 18 of Broad's '359 patent corresponding to one half of the Count. CVC also asserts deficiencies in Embodiment 4 in ToolGen's PCT application while "purports to transfect an NLS tagged Cas9 protein complexed with a sgRNA into the protoplast cells of the Arabidopsis plant (CVC noting in a footnote that codon optimization was not relevant because Cas9 protein was microinjected into the cells). Again relying on ToolGen's purported arguments in related patents, CVC argues that the disclosure of the PCT is deficient for not disclosing the identity of the Cas9 protein nor the NLS signal (out of the "dozens and dozens" that CVC asserts were known in the art), despite ToolGen disclosing "five Cas9 nucleic acid sequences and two Cas9 amino acid sequences in a sequence listing." CVC bases this portion of its argument on deficiencies in the sequence listing with regard to identifying which sequences used in which disclosed embodiment, and in other instances identifying certain sequences for human cell experiments but not for use in mouse or plant cells, or for use in transfection but not microinjection experiments.
The brief concludes with CVC's arguments regarding party admissions and judicial estoppel in view of ToolGen's purported arguments before the Board during ex parte prosecution. Citing Springs Window Fashions LP v. Novo Indus., L.P., 323 F.3d 989, 995 (Fed. Cir. 2003), CVC argues that "[t]he public notice function of a patent and its prosecution history requires that a patentee be held to what he declares during the prosecution of his patent" (emphasis in brief) and were not "simply an inadvertent mistake by the prosecuting attorney" because these statements were "detailed, consistent, and repeated" and were "effected with reasonable clarity and deliberateness" by ToolGen. And in an interference context CVC relies on the BPAI's decision in Louis v. Okada that ToolGen must be bound by its earlier averments.
CVC further argues that judicial estoppel also prevents ToolGen from eschewing its earlier arguments, citing Zedner v. United States, New Hampshire v. Maine, 532 U.S. 742, 742 (2001); and Wilson v. Martin, 789 Fed. Appx. 861, 872 (Fed. Cir. 2019). CVC relies on the rubric that what is prohibited is a party taking a position that is "clearly inconsistent" with an earlier position (although to be fair the bulk of CVC's arguments is that ToolGen's P3 and PCT applications are insufficient to support the arguments it made previously before the Board in ex parte examination). But CVC contends that the clears inconsistency is that "ToolGen's motion, by relying on generic disclosures of Cas9 mRNA and protein in P3 and PCT, directly contradicts ToolGen's prosecution position that NLS addition and codon optimization are required, yet unpredictable, to get CRISPR-Cas9 to function in eukaryotic cells." Permitting ToolGen to garner priority benefit under these circumstances would work an unfair advantage and, on a broader policy plane, "would encourage other parties to advocate one position during prosecution to initiate an interference and then advocate the opposite position to survive the interference."
For all these reasons CVC asks the Board to deny ToolGen's Substantive Motion No. 1.








