By Kevin E. Noonan —
On July 15th, Junior Party the University of California/Berkeley, the University of Vienna, and Emmanuelle Charpentier (collectively, "CVC") filed its opposition to Senior Party ToolGen's Substantive Motion No. 1 for benefit of priority to U.S. Provisional Application No. 61/837,481, filed June 20, 2013 ("P3" or "ToolGen P3"), or alternatively, International Application No. PCT/KR2013/009488, filed Oct. 23, 2013 ("PCT"), in Interference No. 106,127. On August 27th ToolGen filed its Reply.
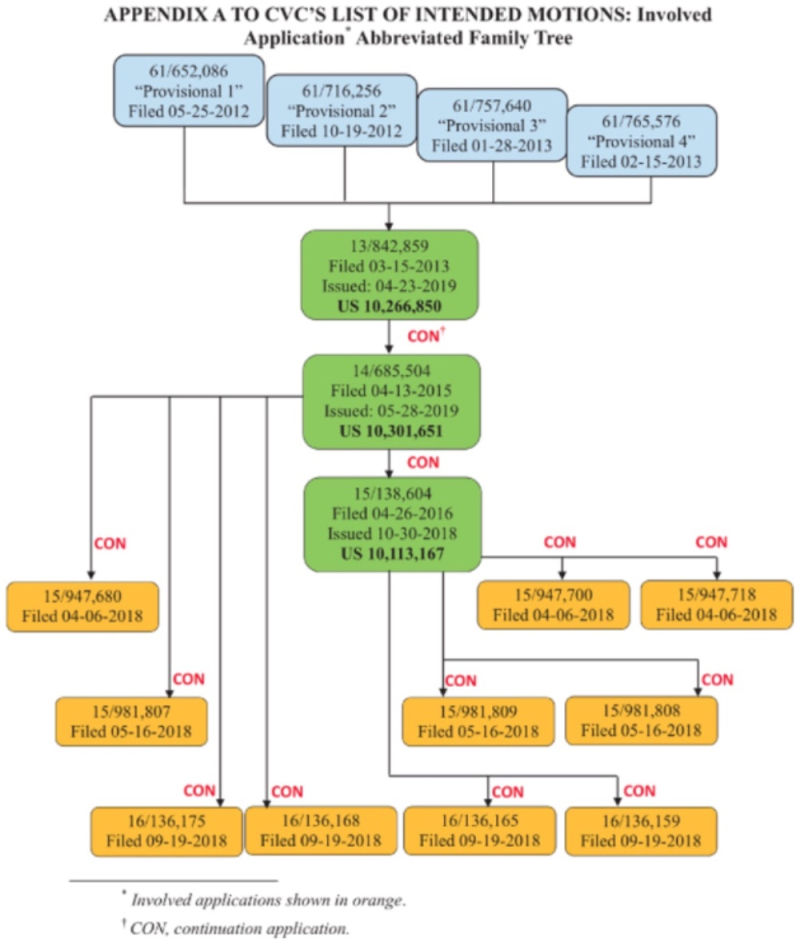
In its Substantive Motion No. 1, ToolGen had set out graphically the relationship of these priority documents, including U.S. provisional application No. 61/717,324 ("P1") for which the Board had granted ToolGen priority upon institution:

The basis ToolGen asserted for its priority claim was satisfaction of the written description and enablement requirements under 35 U.S.C. § 112(a) with regard to two embodiments falling within the scope of the Interference Count. ToolGen argued satisfaction of the alternative language of the Count that recites claim 18 (dependent on claim 15) of Broad's U.S. Patent No. 8,697,359, the brief annotating the limitation recited therein to facilitate identification of ToolGen's disclosure corresponding thereto:
[1] An engineered, programmable, non-naturally occurring Type II CRISPR-Cas system comprising
[2] a Cas9 protein and
[3] at least one guide RNA that targets and hybridizes to a target sequence of a DNA molecule in a eukaryotic cell,
[4] wherein the DNA molecule encodes and the eukaryotic cell expresses at least one gene product and
[5] the Cas9 protein cleaves the DNA molecules,
[6] whereby expression of the at least one gene product is altered; and,
[7] wherein the Cas9 protein and the guide RNA do not naturally occur together;
[8] wherein the guide RNAs comprise a guide sequence fused to a tracr sequence.
The brief asserted Examples 3 and 4 of the P3 (PCT) priority document in support of its satisfaction arguments, noting that one such embodiment (designated 3-1) comprises a Foxn1-specific sgRNA and a Cas9 mRNA, while embodiment 3-2 comprises the same sgRNA and recombinant Cas9 protein. And in each case, ToolGen argued the Examples illustrate CRISPR-mediated cleavage and editing of the target Foxn1 DNA in mouse embryos expressed in the resultant genetically engineered mice. The CRISPR-Cas9 complex is illustrated in the brief by this drawing:

wherein "target DNA [is] in the green box, DNA-targeting sequence of crRNA [is] in the blue box, crRNA:tracrRNA duplex linked together by a -GAAA- loop [is] in the red box, remaining tracrRNA portion shown with brown underline, Cas9 protein with label shown with purple underline depicted as a shaded oval, which is in complex with sgRNA and cleaves the target sequence in the target DNA."
The brief also noted the portions of the P3 priority document showing that such CRISPR-Cas9 complexes successfully cleaved and edited the target Foxn1 DNA, which ToolGen argued provided extensive disclosure in the P3 priority document of "abundant working examples and considerable guidance to a [person of ordinary skill in the art] to make and use CRISPR/Cas9 in eukaryotic cells." ToolGen also noted that the P3 priority document shares the same specification with U.S. Application No. 14/685,510, to which the Board has recognized ToolGen's entitlement to priority.
CVC disagreed in its opposition brief. CVC's argument tracked much of the argument made in CVC's Substantive Preliminary Motion No.2 that the Board deny ToolGen benefit of priority to it P1 provisional application (see "CVC Substantive Preliminary Motion No. 2 to Deny Priority Benefit"). In that argument, CVC maintained that the Board should deny ToolGen priority benefit to the P1application based on party admissions that the provisional application did not disclose an operative embodiment falling within the scope of the Interference Count. Specifically, CVC argued that ToolGen in the prosecution of the P1 application leading to allowance (and declaration of this interference) had argued to the Patent Examiner (and PTAB) that "a codon-optimized Cas9 nucleic acid is required for CRISPR-Cas9 to function in eukaryotic cells" and that "a skilled artisan would have no idea what the outcome may be if one were to codon optimize a Cas9 nucleic acid" (and with the additional argument here that ToolGen's P3 and PCT applications do not disclose Cas9 embodiments comprising a nuclear localization signal, either, wherein both of these alterations CVC alleges ToolGen represented as the "secret sauce" before the Board during ex parte prosecution). This position was consistent with the prokaryotic source of Cas9, and the Board and Examiner relied upon these arguments to find allowable claims that are now claims designated as corresponding to the Count in this interference, CVC asserts. All such claims require use of a Cas9-encoding nucleic acid that is codon-optimized for expression in eukaryotic cells, and ToolGen added this limitation to claims-in-interference to overcome anticipation and obviousness rejections based on the prior art. But neither of ToolGen's '481 provisional application nor its PCT application discloses a codon-optimized Cas9 nucleic acid, according to CVC, nor by ToolGen's own argument (unpredictability) would the skilled worker be able to discern such a nucleic acid with any reasonable basis for expecting such an embodiment could be produced using the disclosure in either the '481 provisional application or its PCT application. Accordingly, CVC argues in its motion, ToolGen cannot in this interference renounce these arguments and rely on the priority date of the either the '481 provisional application or its PCT application to constitute a constructive reduction to practice for eukaryotic CRISPR-Cas9 embodiments falling within the scope of the interference Count. And mere "generalized language" relating to these modifications are inadequate, CVC argues here, pursuant to University of Rochester v. G.D. Searle & Co., 358 F.3d 916, 923 (Fed. Cir. 2004). Thus, according to CVC, the Board should deny ToolGen priority benefit to the '481 provisional application or its PCT application.
CVC also took issue with ToolGen's alleged argument that its disclosure satisfies CVC's half of the count (which doesn't recite these modifications) saying "the issue is what embodiments ToolGen may rely on given that it unambiguously told the Patent Office that NLS addition and codon optimization are required and unpredictable." And the disclosure of the P3 provisional and PCT application to which ToolGen seeks to claim priority benefit do not pass muster under this precedent according to CVC.
Finally, CVC argued that "[p]rinciples of party admission and judicial estoppel preclude ToolGen from taking positions inconsistent with its prior assertions," citing Zedner v. United States, 547 U.S. 489 (2006), and Louis v. Okada, 59 U.S.P.Q.2d 1073, 1075 (B.P.A.I. 2001) (precedential).
CVC then argued that these infirmities extended to ToolGen's P3 and PCT applications that were the subject to ToolGen's Substantive Motion No. 1.
In its Reply, ToolGen addresses with particularity CVC's arguments raised in its Opposition. The Reply begins by asserting that CVC argued against a strawman Motion ToolGen asserts it did not bring. Specifically, ToolGen argues that its Motion No. 1, contingent on the Board granting CVC's Motion No. 1 to deny benefit to ToolGen's P1 application, was predicated on the P3 and PCT applications describing multiple embodiments that correspond to CVC's half of the Count (the Board declaring the interference based on a Count derived from claims in each Party's patents or applications-in-interference in the alternative, a demonstration of correspondence and support to either satisfying a demand for priority benefit. According to ToolGen, CVC's arguments and expert testimony in support thereof do neither address nor rebut ToolGen's ground for asking the Board to recognize its priority claim with regard to the P3 or PCT applications.
ToolGen reiterated its argument that P3 and PCT provide a constructive reduction to practice of embodiments falling within the scope of CVC's portion of the Count, which does not require NLS-tagged nor eukaryotic cell codon-optimized Cas9. ToolGen's general objection to CVC's argument was that "CVC relies on contextual fragments of prosecution arguments, omits critical words from statements, stitches together disparate questions and answers, and misrepresents ToolGen's positions, and its expert Dr. Cullen's opinions, during prosecution, in order to craft a false narrative that ToolGen achieved patentability by focusing on codon optimization and NLSs, not the unpredictability in prokaryote-to-eukaryote translation," something ToolGen contends is "revisionist history." CVC denies it ever asserted that there was a "secret sauce" (specifically, NLS-tagged or eukaryotic cell codon-optimized Cas9) ("never happened," according to the brief). ToolGen takes the portions of the transcript of its arguments to the PTAB in ex parte prosecution to illustrate that "CVC's excerpted quote takes a question asked by Judge Flax and matches it with ToolGen's answer to a different question from a different Judge" (emphasis in brief). ToolGen accuses CVC of "cherry picking" one (specifically, NLS-tagged or eukaryotic cell codon-optimized Cas9) out of six reasons it asserted as to why the skilled artisan would not have had a reasonable expectation of success in practicing CRISPR in eukaryotic cells, not that NLS-tagged or eukaryotic cell codon-optimized Cas9 was required to do so, in the context of non-obviousness without the disclosure of the application that matured into the P1 application. In fact, ToolGen reminds the Board, it was the Examiner's focus on eukaryotic CRISPR embodiments requiring NLS-tagged or eukaryotic cell codon-optimized Cas9 that led to ToolGen's successful appeal in its application-in-interference.
ToolGen further argues that CVC's prosecution history estoppel arguments are unsupported by the evidence in its prosecution history and also legally insufficient. After making the distinction between the issue here (support in the P3 and PCT application for at least one embodiment falling within the scope of the count and the role of PHE in claim construction and infringement contexts and the inapplicability of the estoppel under the procedural issues before the Board in the interference), ToolGen then asserts is arguments that judicial estoppel as urged by CVC also does not apply. ToolGen's reasoning is based on its earlier arguments regarding NLS-tagged or eukaryotic cell codon-optimized Cas9 that it never took the position CVC asserts it did during ex parte prosecution and thus judicial estoppel does not apply.
Finally, turning to the substantive question of whether NLS-tagged or eukaryotic cell codon-optimized Cas9 was needed to show constructive reduction to practice of an embodiment of eukaryotic CRISPR falling within the scope of the interference Count, ToolGen argues that both the P3 and PCT applications satisfy this requirement because both NLS-tagging and eukaryotic cell codon-optimization were well known in the art at the time both these applications were filed, relying on Spectra–Physics, Inc. v. Coherent, Inc., for the rubric that "[a] patent need not teach, and preferably omits, what is well known in the art," 827 F.2d 1524, 1534 (Fed. Cir. 1987). Nevertheless, ToolGen also asserts that in fact both the P3 and PCT applications expressly disclose examples of how each of these modifications are made to adapt CRISPR for the eukaryotic environment. These include knowledge in the art regarding nucleic acids encoding the amino acid sequence of S. pyogenes Cas9, the existence and use of codon-optimization tables for achieving codon optimization in eukaryotic cells, and express reference in both the P3 and PCT applications of the protein/amino acid sequence of a specific NLS used (which ToolGen notes was first disclosed in the art in 1984). ToolGen completes this portion of its argument by distinguishing the circumstances here before the Board and those in Univ. of Rochester v. G.D. Searle &Co., 358 F.3d 916, 929 (Fed Cir. 2004), where the specification of the asserted patents did not disclose support for the claims at issue.
 Schwegman Lundberg & Woessner will be offering a webinar entitled "Inventorship and Assignment: Two Vital Concepts in Patent Law" on September 28, 2021 starting at 12:00 pm (CT). Douglas Portnow and Ryan Connell Schwegman Lundberg & Woessner will provide an overview of how inventorship is determined and why assignment is important, including hypotheticals for audience participation, and will also discuss some of the common errors and how to correct them.
Schwegman Lundberg & Woessner will be offering a webinar entitled "Inventorship and Assignment: Two Vital Concepts in Patent Law" on September 28, 2021 starting at 12:00 pm (CT). Douglas Portnow and Ryan Connell Schwegman Lundberg & Woessner will provide an overview of how inventorship is determined and why assignment is important, including hypotheticals for audience participation, and will also discuss some of the common errors and how to correct them.