By Donald Zuhn –-
 Last month, the U.S. Patent and Trademark Office released its Performance and Accountability Report for Fiscal Year 2021 (FY 2021). The Report notes that in 2018 the Office issued its 2018-2022 Strategic Plan, which "outlines the framework that enables the USPTO to respond to domestic and international customers' demands for vital and timely IP products and services and builds on the agency's current organizational effectiveness and efficiency." The Report also notes that "[t]o fulfill the mission and goals included in the 2018-2022 Strategic Plan, the USPTO developed a comprehensive Strategic Performance Framework."
Last month, the U.S. Patent and Trademark Office released its Performance and Accountability Report for Fiscal Year 2021 (FY 2021). The Report notes that in 2018 the Office issued its 2018-2022 Strategic Plan, which "outlines the framework that enables the USPTO to respond to domestic and international customers' demands for vital and timely IP products and services and builds on the agency's current organizational effectiveness and efficiency." The Report also notes that "[t]o fulfill the mission and goals included in the 2018-2022 Strategic Plan, the USPTO developed a comprehensive Strategic Performance Framework."
The FY 2021 Report specifies nine strategic plan key performance outcome measures that are designed to monitor progress as the Office implements initiatives to achieve its strategic goals (two of the measures are related to the Office's goal to optimize patent quality and timeliness). According to the Report, the Office met or exceeded its targets for seven of the nine key performance metrics. Table 3 of the Report provides data for the two patent-related performance targets (click on any table to expand):
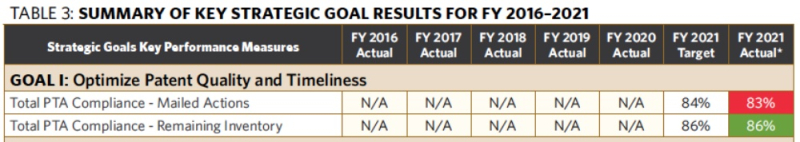
The two patent-related performance targets relate to PTA compliance, with the Office continuing its efforts to achieve a pendency goal that 90% of both total performance (i.e., mailed actions) and total (i.e., remaining) inventory be within statutory time frames set by 35 U.S.C. § 154(b). The Report notes that the statutory time frames (i.e., PTA categories) are as follows:
• Fourteen months from the filing date of a patent application to the mailing date of a first office action (a statement of the Examiner's position and the primary action to establish the issues that exist)
• Four months to respond to an amendment, which includes an RCE
• Four months to act on a decision from the PTAB
• Four months to issue a patent after payment of the issue fee
• Thirty-six months from the filing date of an application to the issue date of a patent
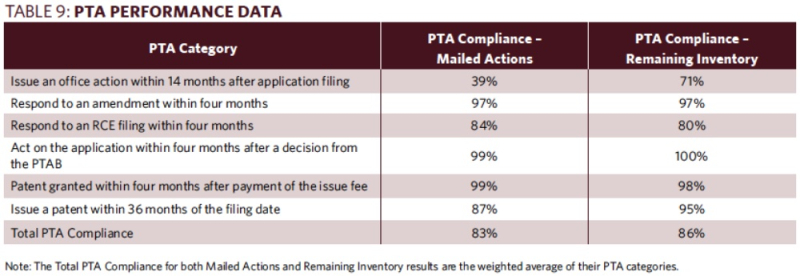
Table 9 of the Report provides a breakdown of the Office's performance for each of these PTA categories with respect to mailed actions and remaining inventory:
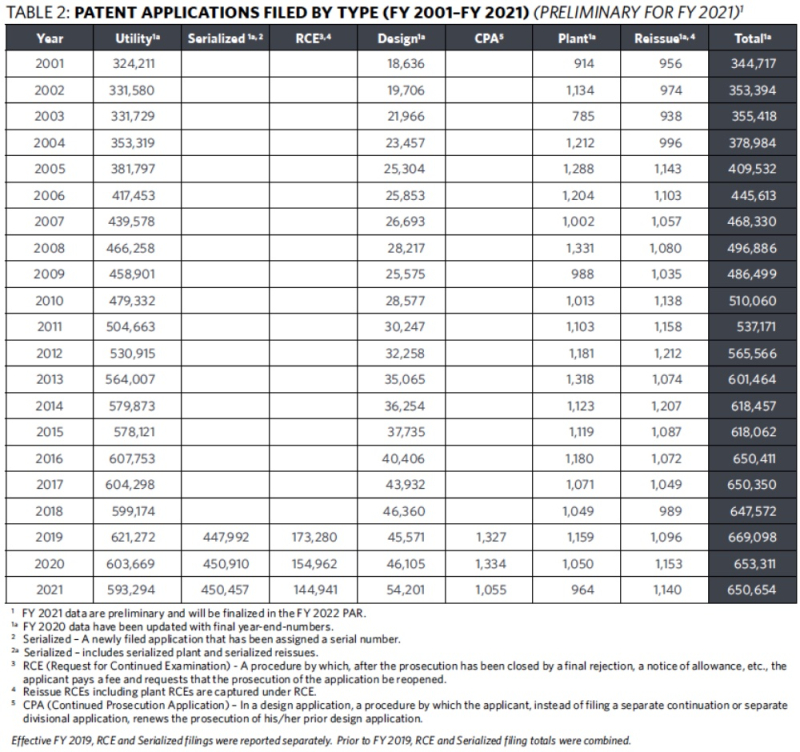
The Report also notes that the number of utility applications filed decreased from 603,669 in FY 2020 to 593,294 in FY 2021, which constituted a 1.7% decrease in filings (see Table 1 below). This followed a 2.5% decrease in application filings in FY 2020 and a 3.3% increase in application filings in FY 2019.
The Report also indicates that while the Office accepted more than 600,000 patent applications for the ninth straight year and topped 500,000 applications for the twelfth consecutive year (see Table 2 below), and despite the second consecutive drop in applications filed, the number of applications awaiting action rose from 630,873 in FY 2020 to 645,573 in FY 2021 (see Table 3 below). It was the Office's fourth consecutive increase in the number of applications awaiting action. The total number of pending applications also increased from 1,011,827 in FY 2020 to 1,045,177 in FY 2021.
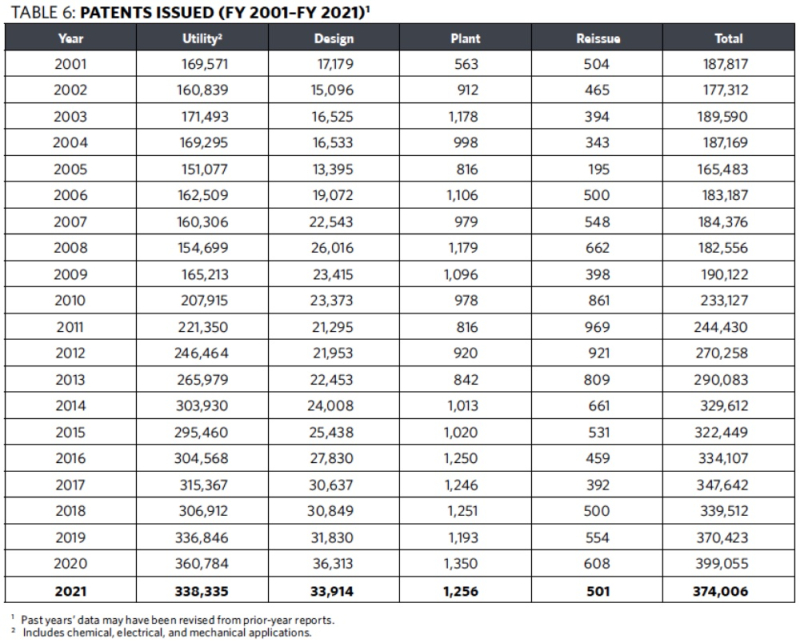
After increasing from 336,846 utility patent issuances in FY 2019 to 360,784 in FY 2020, patent issuances dropped in FY 2021, falling to 338,335 (see Table 6 below). It was the first drop in utility patent issuances since FY 2018.
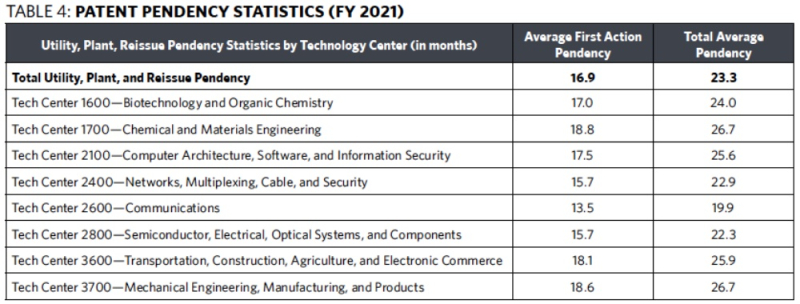
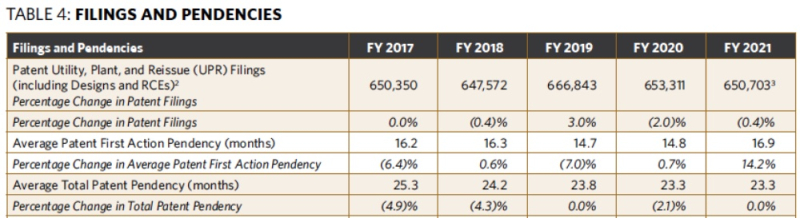
As noted above, the Office now uses PTA compliance rather than first action and total pendency as patent-related performance targets. Given the mixed results obtained for first action and total pendency, the change in patent-related performance targets was convenient. In particular, first action pendency increased from 14.8 months in FY 2020 to 16.9 months in FY 2021 despite a drop in patent filings, and total pendency remained unchanged in FY 2021 at 23.3 months (see Table 4 below).
When comparing pendency statistics by Technology Center, Tech Center 2600 (communications) produced the best average first action pendency (13.5 months), and Tech Center 1700 (chemical and materials engineering) produced the worst average first action pendency (18.8 months) (see Table 4 below). As for total average pendency, Tech Center 2600 also produced the best total average pendency (19.9 months), with Tech Center 1700 and Tech Center 3700 (mechanical engineering, manufacturing, and products) producing the worst total average pendency (26.7 months).
For additional information regarding this and other related topics, please see:
• "USPTO Releases Performance and Accountability Report for FY 2018," February 6, 2019
• "USPTO Releases Performance and Accountability Report for FY 2016," February 6, 2017
• "USPTO Issues Performance and Accountability Report for FY 2015," March 3, 2016
• "USPTO Issues Performance and Accountability Report for FY 2014," April 7, 2015
• "USPTO Releases Performance and Accountability Report for FY 2013," January 9, 2014
• "USPTO Releases Performance and Accountability Report for FY 2012," November 28, 2012
• "USPTO Releases Performance and Accountability Report for FY 2011," November 30, 2011
• "USPTO Releases 2010 Performance and Accountability Report," November 17, 2010
• "USPTO Announces 'Highest Performance Levels in Agency's History' in 2008," November 18, 2008
• "USPTO Announces 'Record Breaking' 2007 Performance," November 15, 2007
• "Patent Office Announces Record-Breaking Year," December 27, 2006