By
Donald Zuhn —
 Last
Last
week, National Public Radio's Science
Friday, which is broadcast nationwide as part of NPR's Talk of the Nation, examined the issue of gene patenting. The debate over gene patenting reignited
in May as a result of the Association for
Molecular Pathology v. United States Patent and Trademark Office case, in
which a group of patients, physicians, academic researchers, and medical
societies filed suit against the U.S. Patent and Trademark Office and Myriad
Genetics, among others, over several patents claiming human BRCA1 and BRC2
genes and methods for detecting mutations in these genes to diagnose a
predisposition to breast and ovarian cancer (see Patent Docs reports here and
here). Since then, two public
radio debates have focused on the issue.
 In
In
June, Dr. Hans Sauer, the Associate General Counsel for Intellectual Property
for the Biotechnology Industry Organization (BIO); Joshua Sarnoff, Professor of
the Practice of Law at American University's Washington College of Law; and
Shobita Parthasarathy, Co-Director of the Science, Technology and Public Policy
Program at the Ford School of Public Policy at the University of Michigan
discussed the topic on WAMU, Washington, DC's leading public radio station (see "Gene Patenting Debate
Continues"). And last August, Kevin Keenan,
Executive Director of the ACLU for San Diego and Imperial Counties; Stacey
Taylor, a partner at DLA Piper; and Dr. Leonard Deftos, Professor of Medicine
in Residence at the University of California, San Diego, and Adjunct Professor
of Law at California Western School of Law tackled the issue on San Diego's
KPBS "These Days" program (see
"Gene Patenting Debate Continues – Round Two"). This past Friday, Patent Docs author Dr. Kevin Noonan, a partner at McDonnell Boehnen
Hulbert & Berghoff, and Daniel Ravicher, the Executive Director of the
Public Patent Foundation (PUBPAT), which is serving as
co-counsel in the Association for
Molecular Pathology case, squared off in a third debate
on the topic on NPR's Science Friday.
 Science Friday
Science Friday
host Ira Flatow (at right) began the program by asking Mr. Ravicher why the lawsuit was
brought. Mr. Ravicher replied that
the suit was "about whether or not the government has the right to grant
exclusive rights to one company over our genes" as well as the knowledge
of gene mutations that might relate to a predisposition for particular types of
diseases. He added that the suit
was filed because Myriad Genetics, which co-owns the seven patents-in-suit, had
been asserting the patents in a way that "crack[s] down on anyone else
doing any research" and "anyone else doing . . . second opinion
testing." After Mr. Flatow
noted that the PUBPAT website describes gene patent holders as having "the
right to prevent anyone from studying, testing, or even looking at a [patented]
gene," Mr. Ravicher declared that "there's absolutely conclusive
ownership of the gene by the patent holder."
 Mr.
Mr.
Flatow then turned to Dr. Noonan (at left) for a response, asking whether he agreed with
Mr. Ravicher's assessment. Dr.
Noonan conceded that Mr. Ravicher was "technically correct," but before
he could opine further, Mr. Flatow asked whether it was true that "people
aren't even able to look at the gene?" Noting that "[t]he National Library of Medicine has a
list of most of the major scientific journals in the U.S. and abroad," Dr.
Noonan revealed that a search of the collection for BRCA references yielded
"almost 3,500 papers that have been published about these genes, some as
recent as last month." (In
addition, a search of the NCBI PubMed database
yields 1,145 references containing "BRCA" in the title or abstract.) In view of the NLM search results, Dr.
Noonan contended that "the idea that people are not doing scientific
research on these genes is just wrong," explaining that "[w]hat
people are not being [allowed] to do, and what some of the named plaintiffs in
this lawsuit are trying to do, is provide clinical testing." According to Dr. Noonan, it was the
commercial use of the BRCA sequences, and not the research use of the
sequences, that was precluded. Mr.
Ravicher, however, replied that he represented researchers at the University of
Pennsylvania, Emory University, and the University of California-Los Angeles, all
of whom had received "very nasty letters" from Myriad saying that
"[w]e know that you are doing things with these genes that relate to
breast and ovarian cancer, and you shall stop or pay us money." He added that "BRCA analysis at
the University of Pennsylvania [had been] shut down because of Myriad's patent
assertion."
Returning
from a short break, Dr. Noonan noted that he was on the program to defend gene
patenting (and not necessarily Myriad), stating that "it would be a
mistake for the United States government — whether through the courts, through
Congress, or through the Patent Office — to not allow genes to be patented." When asked why this would be a mistake,
he replied that "[b]esides being contrary to 200 years of natural products
patenting," a prohibition on gene patenting would adversely impact the disclosure
of gene sequences and investment in biotechnology. Explaining that "in the biotechnology space, investment
is critical," Dr. Noonan noted that "[m]ost of the companies that do
this are small companies like Myriad, and unless there's some way for investors
— in something that's very high risk and [where] most of the products fail —
to get their investment back, you're not going to have investment." As a result, "you don't have the
drugs and the diagnostics that the biotechnology industry has developed over
the past 25 years, and I think that we're in worse shape if we don't have them."
 In
In
response, Mr. Ravicher (at right) argued that "[t]here are valid reasons to have
patents on many things, but you can have patents on too many things, and
there's a line that's been drawn by the Supreme Court, and we fully believe
that these patents land on the wrong side of that line." When pressed by Mr. Flatow to discuss
the impact of the lawsuit on investment, Mr. Ravicher noted that "patents
have both an incentivizing and a chilling effect," and analogized gene
patents with a patent on the law of gravity, stating that "the Supreme
Court has said that you can't patent some things [that] are basic laws of
nature because that would have such a dramatic, countervailing chilling effect
on everyone else — all 300 million other Americans and any research that they
might do on that subject — that granting a [patent] to that one party isn't
worthwhile."
Mr.
Flatow next asked Dr. Noonan whether he could patent a cure for cancer
discovered in the course of doing research on a patented gene. After Mr. Flatow outlined a lengthy
hypothetical involving Bill Gates, Dr. Noonan observed that "we could spin
hypotheticals all day, but it gets down very much to what exactly is it that
you're doing." Dr. Noonan
added that "if you isolate [a patented] gene and use it, then you would be
infringing," but "whether or not that would be something that you
would be sued for . . . would depend on the circumstances." When Mr. Flatow asked whether a patent
holder could bring suit for "any arbitrary reason," Dr. Noonan
responded that Rule 11 of the Federal Rules of Civil Procedure precluded such a
suit, and "[y]ou have to have a good faith belief that somebody has
infringed your patent." Mr.
Ravicher, however, noted that Rule 11 did not apply "to sending nasty
letters." Explaining that a
lot of researchers were either universities that have "very skittish
university counsel offices" or small businesses that "could never
afford to defend themselves," Mr. Ravicher argued that "[m]erely the
threat of a lawsuit just through a letter, which doesn't have to meet Rule 11,
often chills conduct that most reasonable people would agree would never be
covered by a valid claim of any patent."
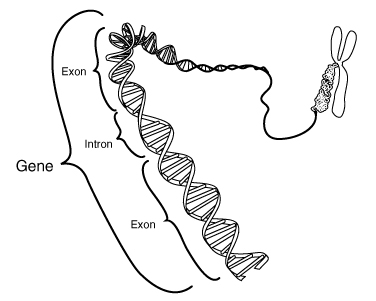
Turning
to the impact of the claim term "isolated," Mr. Ravicher argued that:
[M]ost reasonable people would agree
that if the patents were just on the BRCA genes, that would not be valid
because that exists in our body — the genes exist in our body. It's that these patents say an
"isolated gene," and the Patent Office . . . said that . . . if you
isolated the gene, then that's patentable. And we just think that's incorrect. That's like saying I can go out into
nature, I can dig up some gold, I can isolate it from the mountain, and well
now I deserve a patent on gold, and that's just not the law.
(Mr.
Ravicher also contended that the decision to grant gene patents containing
claims reciting "isolated" sequences was related to the "very
pro-business, pro-patent Administration" under which authority the Office operated.) With regard to the patentability of
natural products, Dr. Noonan countered that antibiotics made by bacteria and
the anti-cancer drug taxol, which is produced in the bark of the yew tree, were
both examples of patentable natural products. As for the BRCA genes, he contended that:
(a) they're not identical to what's in
the chromosome, (b) they're not in the chromosome anymore, and (c) they are
much more converted [or] changed as the Supreme Court would say by the
"hand of man," than . . . taxol from the yew tree. So if you're going to use the traditional
standards of what you have to do to change something that is found in nature to
make it patentable, these genes satisfy that criteria hands down.
Acknowledging
that "Kevin does an excellent job of presenting our opponent's point of
view," Mr. Ravicher noted that the plaintiffs in the BRCA case
nevertheless disagreed. According
to Mr. Ravicher, "[a]ll those isolated natural product claims have never
been upheld by the Supreme Court, and we think they would be invalid under
binding precedent."
For
the last portion of the program, Mr. Flatow took calls from listeners. The first caller stated that while she
couldn't "fault the company Myriad, because they are the holders of the patent
that was granted legally," and therefore she didn't "see any point in
any lawsuits against them," she appeared to favor Mr. Ravicher's argument
regarding the claim term "isolated," and thus saw "quite a bit
of validity in complaints or suits against the U.S. Patent Office." Mr. Ravicher noted that he:
[W]ould disagree [with the caller] when she concedes that these patents are
legal. There are many things done
under our previous Administration that were not legal. Just because the Patent Office has
granted a patent doesn't mean it's actually valid. In fact, about half of the patents granted by the Patent
Office and that are later litigated in court are found to be invalid. They were wrongly granted by the Patent
Office. That's because the Patent
Office has been created to be a pure patent-issuing Office under previous
Administrations. Under the new
Administration, we expect that to change.
So I would not concede that these patents are valid just because they
were granted.
Unfortunately,
Mr. Ravicher's attack on patent validity has some holes. As an initial matter, Mr. Ravicher
provided no support for his assertion that "about half of the patents . .
. litigated in court are found to be invalid." According to the Patstats.org,
however, in cases where patent validity was at issue, the patent holder
prevailed 59.8% of the time in 2008.
Mr. Ravicher's comment about patent validity also glosses over the
motivation of accused infringers to invalidate asserted patents, as well as the
impact of recent Federal Circuit decisions (e.g.,
KSR International Co. v. Telflex Inc.)
on patents that were examined and granted years ago. With respect to Mr. Ravicher's comment that the USPTO had
been "created to be a pure patent-issuing Office under previous
Administrations," one hopes that he was not referring to the most recent
Administration, as the Office's efforts to push through the claims and
continuations rules (among other rules packages) and "reject, reject,
reject" mantra contradict such a characterization.
Turning
to the cost of Myriad's test, Mr. Flatow asked Dr. Noonan whether there wasn't "something
ethically, morally wrong with not allowing someone to come up with a cheaper
test that potentially cancer patients can afford." Dr. Noonan responded that he didn't
have "any evidence that Myriad is the most profitable company in the
country, or that what they're charging is excessive," adding that if
Myriad was "making money hand over fist and w[as] on Forbes' front cover as the most profitable biotechnology company,
maybe you'd have an argument."
He argued that Myriad appeared to be charging an amount that merely "justified
the investment to [develop] the test in the first place."
The
second caller to the program admitted that she was "still a little unclear
on this isolating of the gene," and asked:
I have the gene, right? It's in me? So they've isolated it, but I have the gene, right? So, why don't I have the patent on the
gene, or why can't I sue Myriad?
Agreeing
with the caller, Mr. Flatow exclaimed that Myriad was "using your
gene!" and asked Dr. Noonan for comment (while also accusing him of
"trying to make light" of the caller's question). Dr. Noonan argued that he was not
making light of the question, and added that the caller's (and Mr. Flatow's) comment
was "the emotional crux of the ACLU
lawsuit." He contended that:
[N]obody is going to knock on your
door, like Michael Crichton said in a New
York Times editorial a few years ago, and give you a bill for using your
gene. That's just — it's great —
it gets people all excited and all upset, but it's just — frankly, at the end
of the day, it's a lie.
The
third (and final) caller observed that "the recommendation typically for
women who test positive for the BRCA gene is to have a double mastectomy and
their ovaries removed, and it's troubling that you can do this and you can't
really get a second opinion," and noted that "[t]here's no other
company you can go to get this test done before you have this very radical life
changing surgery." Dr. Noonan
replied that he had "spoken with some people at Myriad [who] say nobody
ever asked them for the second diagnosis," and therefore speculated that
"the second opinion option is one that may still be out there." Mr. Ravicher, however, asserted that he
had "actually written a letter to Myriad on behalf of my client who has
already paid [for one test]," and that Myriad had "refused to grant
our client in this lawsuit the right to get a second opinion." Dr. Noonan asked Mr. Ravicher whether
this letter had been sent to Myriad after the lawsuit was filed, and Mr.
Ravicher acknowledged that it had.
After
Mr. Ravicher suggested that an individual could get a second opinion outside
the U.S. "because these gene patents are not allowed in most other
countries in the world," Mr. Flatow compared such an option with the
dilemma facing women prior to Roe v. Wade. Mr. Ravicher noted that he had "seen
another analogy, which was with respect to patents on human embryonic stem
cells in this country, which were granted [in the U.S.] but not granted in
other countries." He
suggested that these patents had forced "many researchers . . . to move
their labs to foreign countries like Singapore because the [stem cell] patents
didn't exist there." Exposing
the flaw in Mr. Ravicher's analogy, Dr. Noonan pointed out that "the
reason those labs moved offshore was the Bush Administration's restriction on
the cell lines," rather than the assertion of stem cell patents.
While
Mr. Flatow's guests found little common ground, they did agree on one thing at
the conclusion of the debate: that
the case would likely be appealed to the Federal Circuit regardless of who wins
at the District Court level.
For additional information regarding this and other related topics, please see:
• "BRCA Patent Suit to Continue in Southern District of New York," November 2, 2009
• "Empirical
Research Fails to Support Gene Patenting Ban," October 22, 2009
• "Gene
Patenting Debate Continues – Round Two," August 4, 2009
• "The
Unwanted Consequences of Banning Gene Patenting," June 16, 2009
• "Falsehoods,
Distortions and Outright Lies in the Gene Patenting Debate," June 15, 2009
• "Gene
Patenting Debate Continues," June 9, 2009
• "Association for Molecular Pathology v. U.S.
Patent and Trademark Office," May 17, 2009
• "Court Report: Special Edition," May 13, 2009
• "Gene Patenting and
the Wisdom of Judge Lourie," April 12, 2009
• "Science Article
Should Help Allay Gene Patenting Fears," December 17, 2008
 About
About