By
Donald Zuhn —
 On
On
New Year's Eve, we kicked off our third annual list of top biotech/pharma
stories by counting down the first three of the top ten stories covered on Patent Docs in 2009 (see "Top Stories of 2009: #10, #9,
and #8"). Yesterday, we listed stories #7, #6,
and #5 (see "Top Stories of 2009: #7, #6, and #5"). Today, we present our top four stories
for 2009. As with the 2008 list
and 2007 list, links to
our coverage of the 2009 top stories (as well as links to a few articles on
related topics) have been provided in case you missed the articles the first
time around or wish to go back and have another look. As always, we love to hear from Patent Docs readers, so if you think we left something off the list
or disagree with our rankings, please let us know.
#4 — Gene Patenting
Debate Rages On
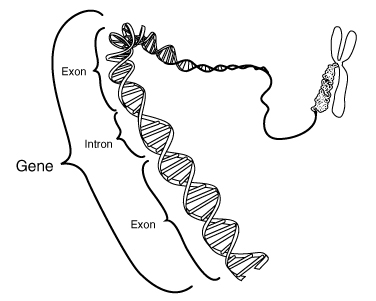 Gene
Gene
patenting has been a hotly debated topic for at least the past three
years. Only two days after Patent Docs began posting new content,
Chicago-Kent College of Professor of Law Lori Andrews and the late Michael
Critchton published an article in the November 11, 2006 issue of Parade magazine, in which the two
advocated for a Genetic Bill of Rights that would have included a prohibition
on gene patents (see "Gene
Patenting in the News Again"). In response, Patent Docs author Dr. Kevin Noonan countered with an article refuting
the factual misstatements in the Andrew-Critchton article (see "In Support of Gene Patents"). In February 2007, Rep. Xavier Becerra
(D-CA) grabbed the anti-gene patent baton when he introduced a bill (H.R. 977)
in the House that would have prohibited patents from being granted for "a
nucleotide sequence, or its functions or correlations, or the naturally
occurring products it specifies" (see "The Continuing Threat to Human
Gene Patenting").
Last
year, however, the issue of gene patenting moved into the courts — and the
debate reignited — when a group of patients, physicians,
academic researchers, and medical societies filed suit against the U.S. Patent
and Trademark Office and Myriad Genetics, among others, over several patents
claiming human BRCA1 and BRC2 genes and methods for detecting mutations in
these genes to diagnose a predisposition to breast and ovarian cancer. As a result of this high-profile litigation,
gene patenting was debated not once, not twice, but three separate times on
Public Radio. In June, Dr. Hans
Sauer, the Associate General Counsel for Intellectual Property for the
Biotechnology Industry Organization (BIO); Joshua Sarnoff, Professor of the
Practice of Law at American University's Washington College of Law; and Shobita
Parthasarathy, Co-Director of the Science, Technology and Public Policy Program
at the Ford School of Public Policy at the University of Michigan discussed the
topic on WAMU, Washington, DC's leading public radio station. And last August, Kevin Keenan,
Executive Director of the ACLU for San Diego and Imperial Counties; Stacey
Taylor, a partner at DLA Piper; and Dr. Leonard Deftos, Professor of Medicine
in Residence at the University of California, San Diego, and Adjunct Professor
of Law at California Western School of Law tackled the issue on San Diego's
KPBS "These Days" program.
Finally, Patent Docs author
Kevin Noonan and Daniel Ravicher, the Executive Director of the Public Patent
Foundation (PUBPAT), which is serving as co-counsel in the Association for Molecular Pathology case, squared off in a third
debate on the topic on NPR's Science
Friday last month. And gene
patenting is not just being debated in the U.S.; the debate has also been
heating up in Australia. With the Association for Molecular Pathology case
moving forward, gene patenting could very well end up on next year's top
stories list as well.
For
information regarding this and other related topics, please see:
•
"Gene Patenting: Australian Potpourri," December 28, 2009
•
"Science Progress Article Examines Impact of Gene Patents on Research,"
December 21, 2009
•
"Gene Patenting Debate Continues – Round Three," December 17, 2009
•
"BRCA Patent Suit to Continue in Southern District of New York,"
November 2, 2009
•
"Empirical Research Fails to Support Gene Patenting Ban," October 22,
2009
•
"The Tragedy of a Bad Idea," August 25, 2009
•
"Gene Patenting Debate Continues – Round Two," August 4, 2009
•
"The Unwanted Consequences of Banning Gene Patenting," June 16, 2009
•
"Falsehoods, Distortions and Outright Lies in the Gene Patenting Debate,"
June 15, 2009
•
"Gene Patenting Debate Continues," June 9, 2009
•
"Association for Molecular Pathology v. U.S. Patent and Trademark Office,"
May 17, 2009
•
"Court Report: Special Edition," May 13, 2009
•
"BIO IP Policy Briefing," May 11, 2009
•
"Gene Patenting and the Wisdom of Judge Lourie," April 12, 2009
•
"The Effects of Intellectual Property Protection on Agricultural Research:
Patents Are Not The Problem," February 3, 2009
•
"Newsweek Joins the Anti-patent Bandwagon," January 26, 2009
#3 — Federal Circuit
Hears Ariad v. Lilly En Banc
 On
On
April 3rd, the Federal Circuit issued its decision in Ariad Pharmaceuticals, Inc. v. Eli Lilly & Co., reversing the
District Court's denial of Lilly's motion for JMOL in view of a jury verdict of
infringement and validity of the asserted claims, and affirming the District
Court's ruling that Lilly failed to establish the affirmative defense of
inequitable conduct. Ariad
Pharmaceuticals filed a petition for a panel rehearing of that decision in
June, and the Federal Circuit determined in August that the appeal warranted en banc consideration. The CAFC's en banc order presented two questions: whether 35 U.S.C. § 112, first paragraph, contains a written
description requirement separate from an enablement requirement, and if so,
what the scope and purpose of that requirement is.
 Armed
Armed
with the parties' principal briefs, Ariad's reply brief, and twenty-five amicus briefs, the Federal Circuit heard
oral argument on December 7th. By
our count, nineteen of the twenty-five amicus
briefs supported Defendant-Appellant-Respondent Eli Lilly & Co. and/or judgment
of affirmance and six briefs supported neither party. Patent Docs provided summaries of both principal briefs, the
reply brief, and fifteen of the amicus
briefs (see links below). Regardless of the en banc panel's decision, the Ariad
appeal is almost certain to make next year's list of top stories.
For
information regarding this and other related topics, please see:
•
"Ariad v. Lilly: Oral Argument – Addendum," December 14, 2009
•
"Ariad v. Lilly: Ariad's Reply Brief," December 10, 2009
•
"Ariad v. Lilly: Oral Argument," December 9, 2009
•
"Amicus Briefs in Ariad v. Lilly: Regents of University of California et
al.," December 6, 2009
•
"Amicus Briefs in Ariad v. Lilly: Briefs by Companies," December 6,
2009
•
"Amicus Briefs in Ariad v. Lilly: American Intellectual Property Law
Association," December 1, 2009
•
"Amicus Briefs in Ariad v. Lilly: Federal Circuit Bar Association,"
November 29, 2009
•
"Amicus Briefs in Ariad v. Lilly: Intellectual Property Owners
Association," November 25, 2009
•
"Amicus Briefs in Ariad v. Lilly: GlaxoSmithKline," November 24, 2009
•
"Amicus Briefs in Ariad v. Lilly: United States," November 23, 2009
•
"Amicus Briefs in Ariad v. Lilly: Google, Verizon Communications Inc. and
Cisco Systems, Inc.," November 22, 2009
•
"Amicus Briefs in Ariad v. Lilly: Professor Christopher Holman,"
November 19, 2009
•
"Lilly Files Principal Brief for Ariad v. Lilly Rehearing En Banc,"
November 16, 2009
•
"Next Up: Ariad v. Lilly Rehearing En Banc," November 10, 2009
•
"Federal Circuit Grants En Banc Review in Ariad v. Lilly," August 21,
2009
•
"Ariad Files Petition for Rehearing in Ariad v. Lilly," June 3, 2009
•
"Amgen, Inc. v. Ariad Pharmaceuticals, Inc. (Fed. Cir. 2009)," June
1, 2009
•
"Ariad Decision Voids Attempt to Use Broad Claiming to Avoid the Written
Description Requirement," April 14, 2009
•
"Ariad Pharmaceuticals, Inc. v. Eli Lilly and Co. (Fed. Cir. 2009),"
April 6, 2009
#2 – USPTO Content with
Dismissal in Tafas v. Doll Despite
Federal Circuit Win
 The
The
Tafas/GSK case (and the claims and
continuations that are at the heart of the case) have, not surprisingly, dominated
our top stories lists for each of the past three years. Last year, the decision by Judge James Cacheris
of the District Court for the Eastern District of Virginia to permanently
enjoin the claims and continuations rules, and the USPTO's appeal of this
injunction, took the top spot on our list (see
"Top Stories of 2008: #5 to #1"). And stories related to the new claims
and continuations rules took the #3 (promulgation of new rules), #2 (USPTO
clarification of new rules), and #1 (new rules preliminarily enjoined) spots on
our 2007 list of top stories (see
"Top Stories of 2007: #5 to #1"). The Federal Circuit's decision in the Tafas v. Doll appeal and the USPTO's decision to rescind the rules and withdraw the appeal claim the #2 spot on this year's list.
On
March 20th, the Federal Circuit stunned many practitioners and applicants by
determining, in a split decision authored by Judge Prost and joined by Judge
Bryson, that Rules 75, 78, 114, and 265 are procedural, but that Rule 78 is
inconsistent with 35 U.S.C. § 120.
Judge Rader dissented in part, contending that all four of the rules at
issue are substantive. In June,
both Dr. Tafas and GSK filed petitions seeking a rehearing, and in July, the Federal
Circuit ordered en banc review of Tafas v. Doll, vacating the earlier
panel decision. On month later,
however, the Department of Justice (DOJ) filed a joint motion with Dr. Tafas
and GSK seeking to stay briefing and oral argument in Tafas v. Kappos (previously Tafas
v. Doll) until 60 days after the U.S. Senate confirmed new Director David
Kappos. And on October 8th, the
USPTO shocked the patent community by announcing that Director Kappos had signed
a new Final Rule rescinding the claims and continuations rules. A little more than a month later, Chief
Judge Paul Michel, writing for most of the en
banc Court of Appeals for the Federal Circuit (Judge Lourie took no part in
the decision), granted the joint motion of all parties to dismiss the appeal of
Tafas v. Kappos, ending nearly
twenty-seven months of angst for most (but not all) patent practitioners and
applicants. With any luck, this
story has made its last appearance on our year-end list.
For
information regarding this and other related topics, please see:
•
"If It Ain't Over 'Til It's Over, Today It's Over," November 13, 2009
•
"Dr. Tafas Files Reply to USPTO/GSK Motion to Dismiss Appeal and Vacate
District Court Judgment," October 20, 2009
•
"Response to Rescission of Claims and Continuations Rules," October
8, 2009
•
"'New Rules' Officially Rescinded," October 8, 2009
•
"Federal Circuit Grants Stay in Tafas v. Kappos," August 21, 2009
•
"Could the "New Rules" Nightmare Finally Be Over?" July 27,
2009
•
"Federal Circuit Grants En Banc Review in Tafas v. Doll," July 6,
2009
•
"GSK Files Petition for Rehearing in Tafas v. Doll," June 4, 2009
•
"Tafas Files Petition for Rehearing in Tafas v. Doll," June 3, 2009
•
"USPTO Will Not Enforce New Claims and Continuation Rules . . . For Now,"
March 25, 2009
•
"Tafas v. Doll (Fed. Cir. 2009)," March 22, 2009
• "Anyone Remember What These 'New Rules' Are All About?"
March 22, 2009
•
"Federal Circuit Issues Decision in Tafas v. Doll," March 20, 2009
#1 — Biotech/Pharma
Industry Tries to Recover from Great Recession
In
2009, the country suffered through its worst recession since the Great
Depression. The biotech/pharma
industry was not immune to the recession's impact. In February, BIO President and CEO Jim Greenwood noted that
almost a third of BIO's membership had less than six months of cash on hand,
45% had less than one year of cash remaining, and only 10% of the 370
publicly-traded biotechnology companies reported a positive income. In April, the National Venture Capital
Association (NVCA) and PricewaterhouseCoopers announced that venture
capitalists had invested $3.0 billion in 549 deals in the first quarter of
2009, a 47% decrease in terms of dollars invested and a 37% decrease in terms
of the number of deals as compared with the fourth quarter of 2008. The NVCA noted that venture funding had
dropped to levels not seen since 1997.
The first quarter drop followed a 2008 drop in venture funding, the
first yearly decline in total investments since 2003.
However,
all of these negative indicators appear to be giving way to some more positive
signs as we move into 2010. As we noted in August, Nashville-based Cumberland Pharmaceuticals
snapped a two-year industry dry spell when it conducted an initial public
offering (IPO) that raised $85 million, and in October, Talecris Biotherapeutics
and Omeros Corp. conducted their own IPOs, raising $950 million and $68.2
million, respectively. At least
eight biotech/pharma companies have filed IPOs since October. In addition, the NVCA released the
results of its latest venture funding study in October, finding that venture capitalists
invested $4.8 billion in the third quarter of 2009, a 17% increase in dollars
as compared to the second quarter of 2009. More importantly (at least for the biotech/pharma industry), the Life Sciences sector, which comprises the
biotechnology and medical device industries, received the highest level of
funding when compared with the other sixteen sectors the NVCA tracks (as a
point of comparison, the Software sector dropped to its lowest level of funding
since 1996, and ten of the seventeen sectors examined by the NVCA experienced
dollar declines in the third quarter, including Semiconductors (14% decline; a
10-year low), Healthcare Services (57% decline), Computers and Peripherals (40%
decline) and Telecommunications (17% decline)). While many expect a slow recovery in 2010, it appears that the worst may be behind us.
For
information regarding this and other related topics, please see:
•
"Biotech/Pharma Companies Look to Market for Capital," December 29,
2009
•
"NVCA Study Shows Increase in Third Quarter Venture Funding," October
23, 2009
•
"Biotech/Pharma IPO Binge," October 9, 2009
•
"BIO Top Brass Meet the Press," September 9, 2009
•
"VC CEO Believes Biotech Is Entering Golden Era," September 6, 2009
•
"Biotech/Pharma Financing Improving, R&D Spending Up," August 31,
2009
•
"Investors Saw Biotech Rebound Coming," August 17, 2009
•
"Is Biotech/Pharma Beginning to Bounce Back?" August 12, 2009
• "Bristol-Myers Squibb to Acquire Medarex," July 23, 2009
•
"Docs at BIO: "Perfect Storm" Super Session," May 28, 2009
•
"Docs at BIO: Steve Burrill's State of the Biotechnology Industry Report
2009," May 19, 2009
•
"Docs at BIO: Ernst & Young Hosts Super Session Addressing Financial
Performance of Biotech Industry," May 19, 2009
•
"First Quarter Venture Capital Funding at 12-Year Low," April 23,
2009
•
"Roche and Genentech Agree on Terms of Merger," March 13, 2009
• "Merck and Schering-Plough to Merge," March 9, 2009
•
"BIO Meets the Press," February 26, 2009
• "Bio-irony: Biotech Turns a Profit in 2008," February 26, 2009
•
"The Future of Biotechnology," February 17, 2009
•
"NVCA Study Shows Decline in 2008 Investment; BIO Study Predicts Biotech
Rebound in 2009," February 16, 2009
•
"Pfizer Expected to Announce Wyeth Acquisition on Monday," January
25, 2009
•
"PatentBuddy.com Examines Impact of Economic Recession on Patent Practice,"
January 13, 2009
 A recent study published in The Columbia Science and Technology Law Review challenges the traditional
A recent study published in The Columbia Science and Technology Law Review challenges the traditional To
To First
First Productivity
Productivity Social
Social Data
Data