By
Donald Zuhn —
 In a decision that will significantly impact the amount
In a decision that will significantly impact the amount
of patent term adjustment ("PTA") that many patentees can expect to obtain under
U.S. Patent Law, the Court of Appeals for the Federal Circuit determined today that the U.S. Patent and Trademark Office had erred in making PTA calculations for two patents owned by Plaintiffs-Appellees Wyeth and Elan Pharma
International Ltd. ("Wyeth"). In Wyeth v. Kappos, the Federal Circuit affirmed
an order by the District Court for the District of Columbia granting summary
judgment in favor of Wyeth, in which the lower court found that the USPTO had
misconstrued 35 U.S.C. § 154(b)(2)(A), and as a result, had denied Wyeth a
portion of the patent term to which it was entitled. In an opinion
authored by Judge Rader and joined by Judges Plager and Moore, the panel
determined that § 154(b) "expressly permits" the legal relief requested by
Wyeth, and that Wyeth was therefore entitled to "extended patent term
adjustments" under § 154(b).
 The
The
case concerns the interplay between two of the three patent term guarantee
provisions of § 154(b)(1), which were introduced into Title 35 as a result of
the enactment of the American Inventors Protection Act (AIPA) in 1999. The two guarantee provisions at issue provide
a one-day extension of patent term for every day that issuance of a patent is
delayed by a failure of the USPTO to comply with various enumerated statutory
deadlines (see § 154(b)(1)(A)) and a
one-day extension of patent term for every day greater than three years after
the filing date that it takes a patent to issue (see § 154(b)(1)(B)). The
above guarantee provisions (often referred to as A or B delays, periods, or
guarantees) are subject to the limitations of § 154(b)(2)(A), which states that
"[t]o the extent that periods of delay attributable to grounds specified
in paragraph (1) overlap, the period of any adjustment granted under this
subsection shall not exceed the actual number of days the issuance of the
patent was delayed."
 In
In
2000, the Patent Office promulgated 37 C.F.R. § 1.703(f), which stated that
"[t]o the extent that periods of adjustment attributable to the
[guarantees] overlap, the period of adjustment granted under this section shall
not exceed the actual number of days the issuance of the patent was
delayed." The Office amended
§ 1.703(f) in 2004 to replace "period of adjustment" with
"period of delay" in order to clarify the regulation, explaining that:
The language of former § 1.703(f)
misled applicants into believing that [periods of A-delay] and [periods of
B-delay] were overlapping only if the [period of A-delay] occurred more than
three years after the actual filing date of the application. If an application is entitled to a
[B-]adjustment . . . the entire period during which the application was
pending before the [PTO] . . ., and not just the period beginning three
years after the actual filing date of the application; is the period of delay
under 35 U.S.C. 154(b)(1)(B) in determining whether periods of delay overlap
under 35 U.S.C. 154(b)(2)(A).
69
Fed. Reg. 21706
(emphasis and changes by panel).
As Judge Rader writes in his opinion, "the 'period of delay,' according to the PTO's
new definition, caused the B guarantee to start with the filing of the
application, not three years later," and therefore, "[u]sing this
framework, the PTO uses either the greater of the A delay or B delay to
determine the appropriate adjustment but never combines the two."
As
we noted in our summary of the District Court decision (see "Wyeth v. Dudas
(D.D.C. 2008)"), the
differences between Wyeth's and the USPTO's perspectives on an exemplary PTA
calculation can be represented schematically as follows (using the example
presented in District Judge Robertson's opinion):

The
USPTO's view (shown above): the
patentee is entitled to three years of PTA.
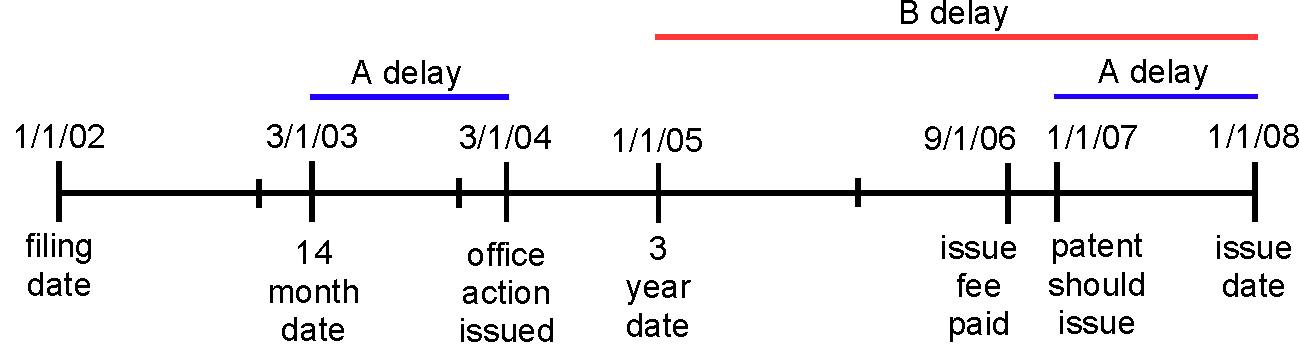
Wyeth's
view (shown above): the patentee
is entitled to four years of PTA.
In
affirming the District Court's grant of summary judgment for Wyeth, the Federal
Circuit states:
This court detects no ambiguity in the
terms "periods of delay" and "overlap" [of § 154(b)(2)(A)]. Each term has an evident meaning within
the context of section 154(b). The
limitation in section 154(b) only arises when "periods of delay"
resulting from violations of the three guarantees "overlap." 35 U.S.C. § 154(b)(2)(A). Significantly, the A and B guarantees
expressly designate when and for what period they each respectively apply.
In
particular, the Court found that "[t]he 'period of delay' for purposes of
the A clause therefore runs from the date the PTO misses the specified deadline
to the date (past the deadline) of response to the underlying action," and
"[t]he 'period of delay' under the express language of the B clause
therefore runs from the three-year mark after filing until the application issues."
As
for the "overlap," the panel states that it is clear that "no
'overlap' happens unless the violations occur at the same time," adding
that "[b]efore the three-year mark, no 'overlap' can transpire between the
A delay and the B delay because the B delay has yet to begin or take any effect." However, "[u]nder the PTO’s
strained interpretation, B delay can occur anytime after the application
is filed," which the Court determined "cannot be reconciled with the
language of the statute (emphasis by panel).
While
the USPTO offered several arguments in defense of its interpretation of the
statute, the panel found none to be persuasive. With respect to the Office's double-counting argument (i.e., A delays during the first three
years of prosecution ultimately lead to B delays after the three-year mark from
filing), the Court responded that "the statute requires as
much." As to the Office's
suggestion that its statutory interpretation prevents situations in which one patentee
might receive a windfall adjustment while a similarly positioned patentee might
receive no adjustment, the Federal Circuit replied that it "perceives
potential perverse results as well under the PTO's suggested interpretations." The Court added:
Regardless of the potential of the
statute to produce slightly different consequences for applicants in similar
situations, this court does not take upon itself the role of correcting all
statutory inequities, even if it could.
In the end, the law has put a policy in effect that this court must enforce,
not criticize or correct.
The
panel also found nothing in the legislative history of the statute that would support
the Office's interpretation, stating that "legislative history — always a
very dull instrument for extracting the essence of statutory meaning — provides
no reason to depart from the language of section 154(b)." Finally, in response to the Office's
assertion that it was entitled to deference under either Chevron U.S.A., Inc. v. Natural Resources Defense Council, Inc.,
467 U.S. 837 (1984), or Skidmore v. Swift
& Co., 323 U.S. 134 (1944), the Court stated that "[b]ecause
the language of the statute itself controls this case and sets an unambiguous
rule for overlapping extensions, this court detects no reason to afford special
deference to the PTO's interpretation." As a result, the Federal Circuit concluded that:
Section
154(b)'s language is clear, unambiguous, and intolerant of the PTO's suggested
interpretation. For that reason,
this court accords no deference to the PTO’s greater-of-A-or-B rubric.
Wyeth
v. Kappos (Fed. Cir. 2010)
Panel:
Circuit Judges Rader, Plager, and Moore
Opinion
by Circuit Judge Rader
For
additional information regarding this and other related topics, please see:
•
"Patent Term Adjustment: 37 C.F.R. § 1.704(b)'s Three-Month Provision,"
November 17, 2009
•
"USPTO Says Some Requests for PTA Reconsideration Are Premature,"
September 7, 2009
•
"Fish & Richardson Catches Error in Patent Office's PTA Calculation,"
July 30, 2009
•
"More § 154(b)(4)(A) Actions Filed against Director," February 8,
2009
•
"Three More Patentees File § 154(b)(4)(A) Actions against Director Dudas,"
January 12, 2009
•
"More Patentees Follow Wyeth's Lead in Seeking Additional PTA,"
December 3, 2008
•
"USPTO to Appeal Wyeth v. Dudas," December 2, 2008
•
"Two Patentees Follow Wyeth's Lead in Seeking Additional PTA,"
November 12, 2008
•
"Wyeth v. Dudas (D.D.C. 2008)," October 16, 2008
 Industry
Industry
















