By Michael Borella —
 Patent examiners have a hard job. They are given a relatively short amount of time in which they are supposed to thoroughly review a patent application, search for relevant prior art, and write a well-reasoned Office action. Given the challenges of doing so, each of these tasks — reviewing, searching, and writing — may not get the attention that it needs. Further, examiners are sometimes asked to examine applications from technology areas in which they lack the appropriate expertise. Moreover, relatively few examiners are attorneys and thus may struggle to apply the relevant law in a cogent fashion.
Patent examiners have a hard job. They are given a relatively short amount of time in which they are supposed to thoroughly review a patent application, search for relevant prior art, and write a well-reasoned Office action. Given the challenges of doing so, each of these tasks — reviewing, searching, and writing — may not get the attention that it needs. Further, examiners are sometimes asked to examine applications from technology areas in which they lack the appropriate expertise. Moreover, relatively few examiners are attorneys and thus may struggle to apply the relevant law in a cogent fashion.
With all of this, it is not surprising that the quality of patent examination can vary dramatically. Just based on examination statistics, the average allowance rate across all examiners is 76% but some examiners have an allowance rate of less than 10% and others have an allowance rate of greater than 90%.[1] It is highly unlikely that all of this variability can be attributed to technology area or chance — every patent attorney has stories of interactions with both notably cooperative and notably difficult examiners from different groups and art units within the USPTO.[2]
I'll have to admit that I generally enjoy interactions with competent, thoughtful examiners even when the ultimate outcome is not what I was hoping for. When an examiner can clearly articulate a solid factual and legal basis for rejecting claims — and better yet, is willing to make suggestions that would advance prosecution — that makes my job easier, simplifies prosecution, and reduces applicant cost. A win all around.
But what about examiners at the other end of the spectrum and the tactics they employ — perhaps intentionally, perhaps not — to reject claims without sufficient basis? Here are a few common techniques that I've seen used over the years, and how to counter them. I am not just talking about poor spelling and grammar, or fuzzy reasoning. These examination annoyances are more fundamental.
Before we get started, let me reiterate the importance of examiner interviews. Even when faced with the most confusing or seemingly incorrect Office action, a conversation with the examiner can often help, sometimes in a major way, and will rarely if ever hurt the application's chances for allowance. Interviews can be crucial when faced with any of the annoyances below, with the caveat that some annoyances do take place during interviews.
1. The Low-Effort First Office Action
Some experienced examiners will provide these Office actions because they know how the game is played. They put as little effort as possible into the application review and search, typically just reading the claims without consideration of much of the specification, and finding 1-2 (maybe 3) references that "seem close." The resulting Office action is typically very short and very wrong, with a § 102 or § 103 rejection of all pending claims based on the same set of references.
The good news is that the low-effort first Office action is usually followed by the examiner putting more time and thought into subsequent Office actions, once they have a sense of the applicant's commitment. The key is to convince the examiner that you are going to make them work. This may involve rebutting the rejection of the independent claims on multiple grounds, as well as arguing separately for some of the dependent claims. Schedule as many interviews as you can and in them casually state that the applicant believes that the invention is important and plans on seeing it through. Most of the time, the examiner will eventually come around and either provide a more reasonable grounds for rejection or negotiate an allowance.
2. The <Ctrl>-F Rejection
As the cliché goes, it can be hard to see the forest for the trees. Here, the examiner focuses entirely on the trees rather than the forest. This type of rejection is based on the examiner selecting certain keywords from the claims, finding prior art references that contain the same keywords, and stitching these references together, usually into an obviousness rejection. Of course, the context in which the keywords are used in the claim is lost. The prior art may contain the same language as the claims but is often not otherwise related. It is not unusual to have a strong ground for rebutting the examiner's application of the prior art to most if not all claim elements.
In addition to rebutting the examiner on an element-by-element basis, the lack of a more contextual approach usually results in the examiner's motivation to combine references being weak at best. So this motivation may be rebutted as well. When applicable, it may also be helpful to quote the stated goals of the prior art references back to the examiner to further illustrate how far off they are from the applicant's claimed invention.
3. The Over-Reliance on Broadest Reasonable Interpretation
It is axiomatic that claims are given their broadest reasonable interpretation (BRI) during examination. But some examiners take this to mean that they can come up with an alleged BRI that is a purely literal reading of each word or phrase in the claim. That is not what BRI is all about.
BRI is tempered by how claimed elements fit into the overall structure of the claims and how they relate to other claim elements, as well as how the constituent words and phrases are defined in the specification and drawings. There is a helpful section of the MPEP to quote back to examiners when rebutting an examiner's misuse of BRI:
The broadest reasonable interpretation does not mean the broadest possible interpretation. Rather, the meaning given to a claim term must be consistent with the ordinary and customary meaning of the term (unless the term has been given a special definition in the specification), and must be consistent with the use of the claim term in the specification and drawings. Further, the broadest reasonable interpretation of the claims must be consistent with the interpretation that those skilled in the art would reach.[3]
Pointing out where the examiner's interpretation of claim language conflicts with how it is used in the claims or the specification can be helpful. During interviews, it can also help to ask the examiner for the basis of their claim construction — if it is faulty, that will often become readily apparent.
4. The Hand-Wave over Dependent Claims
Let's say the examiner provides reasonable grounds for rejecting the independent claims. In some cases, substantive examination more or less stops there, with the examiner using the same references to improperly reject all of the dependent claims as well. Often (but not always) used in conjunction with the Low-Effort First Office Action, this type of rejection typically ignores the actual features of the dependent claims.
In these situations, it is likely that you will end up amending the claims but it may not be clear what amendment should be made. If multiple dependent claims are rejected on sketchy grounds, which one should be incorporated into the independent claims? Or should you select a previously-unclaimed feature from the specification?
One approach is to find one or more dependent claims that appear to distinguish over the references, and present arguments on each to the examiner during an interview. Tell the examiner that you are not sure which one to amend into the independent claims and therefore would appreciate the examiner's opinion on each. Another approach would be to file a response with no amendments that argues for allowance of the favorable dependent claims. This latter tactic might result in reception of a final rejection that again ignores the dependent claims and your arguments as well. Thus, the interview might bear more fruit.
5. The Kitchen Sink Rejection
"These claim features can be found in Reference A at paragraphs [0003]-[0117] and figures 1-18." I wish I was kidding, but this quote is not far off from what I've seen in actual Office actions (I've paraphrased to protect the guilty). The kitchen sink rejection fails to pinpoint exactly what in the reference allegedly teaches the claim elements, taking an "it's in there somewhere" approach.
The catch here is that sometimes the cited reference does disclose at least some claim elements, so it cannot be dismissed out of hand. But rather than play "Where's Waldo?", one strategy is to argue that the examiner has not established a prima facie case for the rejection due to a lack of compliance with 37 C.F.R. § 1.104(c)(2). That regulation states "When a reference is complex or shows or describes inventions other than that claimed by the applicant, the particular part relied on must be designated as nearly as practicable. The pertinence of each reference, if not apparent, must be clearly explained and each rejected claim specified."
Nonetheless, interviewing the examiner is the most efficient way to vet these rejections. In the interview, try to get the examiner on your side by explaining that you are confused, that you need to provide the applicant with a specific rationale for narrowing the claims, and that you have been unable to find one in the Office action as mailed. Ask the examiner to at least narrow the cited sections down to a few specific paragraphs and/or figures.
6. The No-Preparation Interview
Every so often an examiner does not properly prepare for an interview. I certainly can empathize, as we all lead busy lives. But some examiners are quite insistent that it is okay for them to do this. It is not.
There are two flavors of no-preparation interview. The more common is that the examiner clearly has no idea what the invention is, what the claims recite, or what the cited references disclose. The interview quickly becomes a waste of time. As a next step, ask the examiner for a follow up interview "to give you more time to fully consider the case." Sometimes this works. But when it does not, it is time to get the examiner's supervisor involved. Doing so in a polite fashion is often the most effective, such as "I have attempted to conduct an interview with Examiner Jones on multiple occasions but they have not been ready to discuss the application. My client really wants to move this case forward — what should I do?" Sometimes requesting that the supervisor sit in on the interview can ultimately result in a more productive discussion.
The second flavor is rarer — the examiner states that interviews are only for a high-level discussion of the claims. This is not the case, and is usually a sign that the examiner is not interested in conducting the interview and just wants to get out of it quickly. Nonetheless, sometimes useful information can be gleaned from these examiners by just jumping into discussion of the claim elements and the prior art. This at least gives the examiner a chance to respond to your questions and arguments. Once the examiner learns that you are not going to just give up, they may decide that it is in their interest to put effort into the interview.
7. The "I Know of Better Art" Dodge
Here's the pattern: the examiner's written rejections are not great, and you make your point successfully during an interview that this is the case. The examiner admits that the rejection could have been better but then contends that it does not matter because "I know of better art that will maintain the rejection."
This is the examiner saving face, so let them do so. But follow up by asking the examiner to put this new art on the record in the next Office action. Tell the examiner that you can only respond to the rejections that appear in writing and not to uncited references — though you will be happy to do so when they are used in a formal rejection. Sometimes the examiner will provide patent or application numbers of these references, which you can then enter into the record in an information disclosure statement. Still, remind the examiner that the burden of proof remains with the USPTO to provide a rejection based on these references.
8. The "Expert" Examiner
This does not happen often, but every few years I find myself discussing a case with an examiner who takes the position that they are a bona-fide expert in the field of the invention. For example, they might state in an interview, "I worked in the industry for 15 years on the same type of technology, and there is nothing innovative about what you are claiming — the claims are entirely obvious." This stance regarding the invention may or may not be the case, but such a statement is almost always attached to an Office action that is poorly reasoned and cites prior art that is off-base.
The examiner's self-aggrandizing is of limited relevance since any alleged obviousness of the claimed invention must be from the point of view of a person of ordinary skill at the time of the invention.[4] If the examiner has stated that they are a person of exceptional skill, then their position is by definition not that of one of ordinary skill in the art. This is different from when an examiner relies on common knowledge to take official notice of facts not in the record.[5]
Dealing with this sort of examiner can be tricky, but first try to get them to put as much as possible in writing for the next Office action. This will give you something more substantial to rebut. Or, you can appeal to their ego and point out that their expertise goes beyond the "person of ordinary skill at the time of the invention" and that they should reconsider their rejection by putting themselves in the shoes of a less experienced and knowledgeable individual.
Conclusion
These eight examination annoyances are just a few of the most common. There certainly are more. But the main point is that each requires a slightly different style of legal jiu-jitsu to get the best outcome for your client. Good luck.
[1] Juristat’s Prosecution Metrics dashboard, accessed April 23, 2022.
[2] To be fair, just about any profession has a similar distribution of individual worker quality — plumbers, electricians, rideshare drivers, and yes, even patent attorneys.
[3] MPEP § 2111.
[4] See, e.g., MPEP § 2141(II).
[5] MPEP § 2144.03.
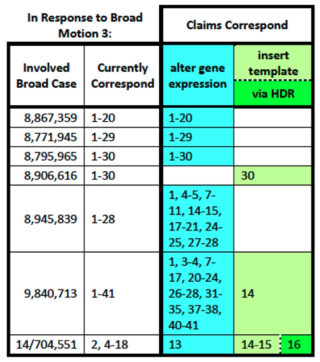
 On November 19th, Junior Party the University of California, Berkeley; the University of Vienna; and Emmanuelle Charpentier (collectively, "CVC") filed its Substantive Preliminary Motion No. 3 in Interference No. 106,132 (which names Sigma-Aldrich as Senior Party), asking the Patent Trial and Appeal Board to substitute the Count of the interference pursuant to 37 C.F.R. §§ 41.121(a)(1)(iii) and 41.208(a)(1). On February 18th, Senior Party Sigma-Aldrich filed its Opposition, and on April 7th CVC filed its Reply.
On November 19th, Junior Party the University of California, Berkeley; the University of Vienna; and Emmanuelle Charpentier (collectively, "CVC") filed its Substantive Preliminary Motion No. 3 in Interference No. 106,132 (which names Sigma-Aldrich as Senior Party), asking the Patent Trial and Appeal Board to substitute the Count of the interference pursuant to 37 C.F.R. §§ 41.121(a)(1)(iii) and 41.208(a)(1). On February 18th, Senior Party Sigma-Aldrich filed its Opposition, and on April 7th CVC filed its Reply.