By Kevin E. Noonan —
 In the Federal Circuit's decision in Association for Molecular Pathology v. U.S. Patent and Trademark Office (the Myriad case), Judge Kimberly Moore wrote a separate opinion in which she concurred with the opinion of the panel affirming the District Court's judgment on the standing issues and the method claims, and concurred with the judgment reversing the District Court's judgment that the composition of matter claims were not patent-eligible under 35 U.S.C. § 101 as being "products of nature." Judge Moore joined Judge Lourie's majority opinion fully with regard to some of the composition of matter claims at issue (specifically, claims to cDNA embodiments of the BRCA genes and claims to subsequences such as primers) and concurred in the result regarding claims that encompassed isolated genomic DNA. This post will discuss her reasoning.
In the Federal Circuit's decision in Association for Molecular Pathology v. U.S. Patent and Trademark Office (the Myriad case), Judge Kimberly Moore wrote a separate opinion in which she concurred with the opinion of the panel affirming the District Court's judgment on the standing issues and the method claims, and concurred with the judgment reversing the District Court's judgment that the composition of matter claims were not patent-eligible under 35 U.S.C. § 101 as being "products of nature." Judge Moore joined Judge Lourie's majority opinion fully with regard to some of the composition of matter claims at issue (specifically, claims to cDNA embodiments of the BRCA genes and claims to subsequences such as primers) and concurred in the result regarding claims that encompassed isolated genomic DNA. This post will discuss her reasoning.
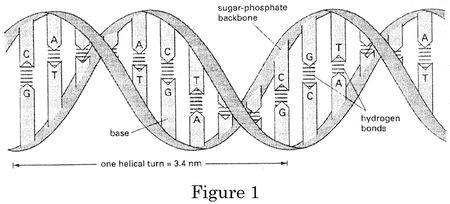
Like Judge Lourie, Judge Moore begins her opinion with her view on the subject matter at issue, stating that "DNA is a chemical polymer. In principle, a polymeric DNA sequence is no different than any other well known polymer, for example, nylon." Illustrating this concept, her opinion contains a drawing, showing that "polymerization changes the monomers and results in a molecule with a different ionic charge, different chemical bonds, and a different chemical composition, as compared to the monomers in aggregate."

The opposite of polymerization, "deconstructing" in her parlance, "leads to similar results: a fragment of a DNA sequence has different properties than the parent molecule from which it is derived." Judge Moore uses the example that:
[A] two nucleotide sequence (T-C), has a different chemical structure, and different chemical connections than the same subunit found within the larger A-T-C-G-T structure. Despite many similarities, it is impossible to find the isolated T-C structure in the A-T-C-G-T molecule. This is because, instead of being connected to a phosphate, the C subunit terminates in a different functional group, a hydroxyl. Likewise, instead of being connected to another sugar via a phosphodiester bond, the T subunit instead terminates in a phosphate. The isolated T-C sequence is a different molecule than the "T-C" sequence appearing as part of the larger A-T-C-G-T polymer.
The relevance of these chemical facts is that, for Judge Moore, "[t]he isolated DNA sequences at issue in this case have the same type of chemical changes, but on a much bigger scale":
[L]ike the simple sequences discussed above, just because the same series of letters appears in both the chromosome and an isolated DNA sequence does not mean they are the same molecule. While the isolated DNA molecules claimed in this case are undoubtedly inspired by the corresponding naturally occurring sequence present on the chromosome, man must create these isolated DNA molecules. This can be accomplished by building them de novo using chemical or biological means, or by chemically altering the larger polymer to cleave off adjacent portions.
Judge Moore makes a distinction similar to Judge Lourie's between "isolating" and "purifying" DNA molecules: "Isolation of a DNA sequence is more than separating out impurities: the isolated DNA is a distinct molecule with different physical characteristics than the naturally occurring polymer containing the corresponding sequence in nature. These differences, of course, are directly related to the change in chemical bonds in the isolated DNA."
She also cites the distinction that the isolated DNA molecule claimed by Myriad is no longer contained in the chromosome, and that for some of the claims at issue these DNA molecules are "as short as fifteen nucleotides, and the isolated BRCA1 cDNA sequence has approximately six thousand nucleotides (see, e.g., '282 col.67-80 (SEQ ID NO:1))," noting the further distinction between he structure of cDNA (i.e., lacking introns) and genomic DNA. "Even the isolated BRCA1 gene, however, is substantially smaller than chromosome 17, which includes the unisolated BRCA1 gene as well as many other genes. Isolation of a DNA sequence thus results in a substantially smaller molecule compared to the naturally occurring sequence as part of the chromosome" (citations omitted).
Next Judge Moore discusses the distinctions between cDNA, which "unlike isolated or unisolated DNA, has a unique sequence of DNA bases (A, C, G, T) which is not actually present in nature." These include "form[ing] DNA sequence that is completely different than the corresponding RNA" as well as the fact that "[t]here is no contiguous sequence on the chromosome that duplicates the cDNA sequence" due to the lack of introns (as shown in the figure below from Judge Moore's opinion).

For both genomic DNA and cDNA, Judge Moore recognizes that "[c]reating isolated DNA allows a scientist, among other things, to remove potentially confounding sequences that are naturally present in the larger chromosomal polymer, and instead focus on just the sequence of interest. This aspect of isolated DNA has important practical consequences and leads to additional utility, particularly for the smaller isolated fragments" (citing, inter alia, the use of primer sequences to detect mutations in the BRCA1 genes in a patient sample). Having set forth her understanding of the relevant scientific principles ("[a]rmed with this scientific background"), Judge Moore's concurrence sets the legal basis for her conclusions regarding "the isolated DNA claims at issue."
Like Judge Lourie, Judge Moore considers Funk Bros. Seed Co. v. Kalo Inoculant Co. and Diamond v. Chakrabarty as controlling precedent. "[T]erms as 'the work of nature' and the 'laws of nature' . . . are vague and malleable terms . . . . Arguments drawn from such terms for ascertaining patentability could fairly be employed to challenge almost every patent," she writes, citing Funk Bros. Seed Co., 333 U.S. at 134-45 (Frankfurter, J., concurring). Like the majority opinion, Judge Moore contrasts the patent-ineligible mixture of bacteria claimed in Funk Brothers with the patent-eligible recombinant bacterium claimed by Chakrabarty. However, she states that "Funk Brothers and Chakrabarty do not stake out the exact bounds of patentable subject matter. Instead, each applies a flexible test to the specific question presented in order to determine whether the claimed invention falls within one of the judicial exceptions to patentability." Funk Brothers sets forth one end of this spectrum, where an invention that "serve[s] the ends nature originally provided" is likely unpatentable subject matter, but an invention that is an "enlargement of the range of . . . utility" as compared to nature may be patentable. 333 U.S. at 131. Chakrabarty, on the other hand, is an example of an invention having "a distinctive name, character, and use, e.g., markedly different characteristics with the potential for significant utility, is patentable subject matter. 447 U.S. at 309-310." While she admits that these cases come to different conclusions, "the inquiry itself is similar."
In discussing Learned Hand's decision in Parke-Davis & Co. v. H.K. Mulford Co., 189 F. 95, 103 (S.D.N.Y. 1911), that adrenaline was patent-eligible, and the same result in Merck & Co. v. Olin Mathieson Chem. Corp., 253 F.2d 156 (4th Cir. 1958), for vitamin B12, Judge Moore's analysis reveals an important consideration for her reasoning: "mere purification of a naturally occurring element is typically insufficient to make it patentable subject matter," which explains the decisions in the In re Marden and DeForest Radio cases (which the majority recognize to be novelty rather than patent-eligibility cases). Unfortunately, this doctrinal admixture confuses the analytics, and while the relevance of its casting aside of the neat analytical boundaries set forth by Judge Rich in In re Bergy raises jurisprudential questions, perhaps its greater significance is that it undercuts the reasoned basis for determining patent-eligibility by conflating it with the different question of patentability.
Regardless, Judge Moore concludes that there is "no reason to deviate from this longstanding flexible approach in this case. Keeping these principles in mind, I analyze the isolated DNA claims below, to determine whether they have markedly different characteristics with the potential for significant utility, e.g., an "enlargement of the range of . . . utility" as compared to nature. Chakrabarty, 447 U.S. at 309-310; Funk Bros., 333 U.S. at 131."
The cDNA embodiments of the claimed invention "present the easiest analysis." She rejects the District Court's determination that these molecules are "products of nature" for the simple reason that they "do not exist in nature." In addition, these molecules have different "uses" (an "enlargement of the range of . . . utility") because "it can be used to express a protein in a cell which does not normally produce it." She also cites the various chemical distinctions (different constituent sugars and bases) and consequent differences in chemical stability as reasons to conclude that cDNAs are not "products of nature." In the language of the Supreme Court precedent relevant to her decision:
cDNA sequences thus have a distinctive name, character, and use, with markedly different chemical characteristics from either the naturally occurring RNA or any continuous DNA sequence found on the chromosome. The claimed isolated cDNA sequences are the creation of man, made using biological tools and the naturally occurring mRNA as a template. cDNA is therefore not one of the "'manifestations of . . . nature, free to all men and re-served exclusively to none'" that falls outside of the patent system. Chakrabarty, 447 U.S. at 309 (quoting Funk Bros., 333 U.S. at 130).
The "more difficult issue" are "DNA sequences that have the same pattern of DNA bases as a natural gene, in whole or in part." These sequences are present in the human body, "albeit as part of a much larger molecule, the chromosome." For Judge Moore, the "markedly different chemical structure" of such DNAs when isolated, relied upon in Judge Lourie's opinion, are not a sufficient basis for determining that such isolated DNA molecules are patent-eligible because they are not "markedly different" enough, citing by analogy Funk Bros., 333 U.S. at 130-31 (Creation of "a new and different composition" of bacterial strains was nevertheless not patentable subject matter).
However, "the mere fact that the larger chromosomal polymer includes the same sequence of nucleotides as the smaller isolated DNA is not enough to make it per se a law of nature and remove it from the scope of patentable subject matter." The chemical distinctions noted earlier in the opinion distinguish these isolated DNA molecules from minerals, for example, because minerals are "created by nature without the assistance of man. Instead, the claimed isolated DNA molecules . . . are not naturally produced without the intervention of man. Cf. Chakrabarty, 447 U.S. at 312-13. For Judge Moore, the question then resolves into a determination of "whether these differences impart a new utility which makes the molecules markedly different from nature." The "short" DNA molecules (as claimed for example in claim 5 of U.S. Patent No. 5,747,282):
5. An isolated DNA having at least 15 nucleotides of the DNA of claim 1.
pass muster under this analysis:
The shorter isolated DNA sequences have a variety of applications and uses in isolation that are new and distinct as compared to the sequence as it occurs in nature. These smaller isolated DNA sequences — including isolated radiolabeled sequences mirroring those on the chromosome — can also be used as the basis for probes. Naturally occurring DNA cannot be used to accomplish these same goals. Unlike the isolated DNA, naturally occurring DNA simply does not have the requisite chemical and physical properties needed to perform these functions.
This new utility ("use [of the] isolated DNA molecules as the basis for diagnostic genetic testing"]) satisfies the Funk Brothers requirement for "an 'enlargement of the range of . . . utility,'" at least because "[d]iagnostic testing . . . is not a natural utility" and "the claimed DNA does not 'serve the ends nature originally provided.'" These isolated DNA molecules have "markedly different properties which are directly responsible for their new and significant utility," she writes, noting that "[t]he same sequence, as it appears in nature as part of the chromosome, simply cannot be used in the same way." Accordingly, "small, isolated DNA fragments are patentable subject matter" in her opinion.
The "[l]onger strands of isolated DNA" are what apparently provided most of the impetus for Judge Moore's concurring opinion, since for her (as opposed to Judge Lourie), the patent-eligibility of these molecules present "a much closer case." And while Judge Moore "ultimately conclude[s] that these longer isolated sequences, including the isolated gene sequence as a whole, are also patentable subject matter," her reasoning is different than for the shorter sequences. She concedes that "[a]ll of the same structural arguments apply to any length of isolated DNA so, like the shorter strands, an isolated DNA coding for a gene does have a literal chemical difference from the gene as it appears on the chromosome." However, "[u]nlike the shorter strands of isolated DNA, the chemical and structural differences in the isolated gene do not clearly lead to an "enlargement of the range of . . . utility" as compared to nature she says, citing Funk Bros., 333 U.S. at 131, and this difference raises her concerns for whether isolated genomic DNA molecule should be included within the scope of patent-eligible subject matter.
In view of these concerns, it may seem curious that Judge Moore decides that such molecules are patent-eligible. She explains that "[i]f I were deciding this case on a blank canvas, I might conclude that an isolated DNA sequence that includes most or all of a gene is not patentable subject matter." This is due to the fact that "[d]espite the literal chemical difference, the isolated full length gene does not clearly have a new utility and appears to simply serve the same ends devised by nature, namely to act as a gene encoding a protein sequence." This statement ignores the real-world uses to which isolated DNA molecules can be put (uses Judge Moore appreciates, citing Amgen's EPO product later in her opinion) specifically the basis for the biotechnology industry: providing useful quantities of proteins (erythropoietin, human growth hormone, interferon, blood clotting factors, and a host of other products) that would otherwise be in insufficient quantities from biological sources or have deleterious side-effects (such as adverse immunological reactions to animal insulins despite their ready availability). However, Judge Moore recognizes that the Court does not write with a blank slate:
This case, however, comes to us with a substantial historical background. Congress has, for centuries, authorized an expansive scope of patentable subject matter. Likewise, the United States Patent Office has allowed patents on isolated DNA sequences for decades, and, more generally, has allowed patents on purified natural products for centuries. There are now thousands of patents with claims to isolated DNA, and some unknown (but certainly large) number of patents to purified natural products or fragments thereof. As I explain below, I believe we must be particularly wary of expanding the judicial exception to patentable subject matter where both settled expectations and extensive property rights are involved. Combined with my belief that we should defer to Congress, these settled expectations tip the scale in favor of patentability.
Judge Moore cites as one example U.S. Patent No. 3,067,099 (claiming vancomycin, an antibiotic produced by bacteria found in soil) and U.S. Patent No. 4,552,701 (claiming a vancomycin fragment produced by removing a sugar unit), and then (curiously) focuses on the second and ignores the first in discussing patent-eligibility of biologically derived molecules. She further cites U.S. Patent No. 141,072, claiming "[y]east, free from organic germs of disease," issued to Louis Pasteur in 1873 (ironically, in view of how the opinion has been treated, one of the patents cited by Judge Rich in In re Bergy a generation ago), as well as the Patent Office justification that "isolated DNA is no different from the isolated natural products of Parke-Davis. See 66 Fed. Reg. at 1093 (quoting Parke-Davis)." She also notes that such claims have not been strangers to the Federal Circuit, citing (again, ironically in view of its author) the Court's opinion in Amgen, Inc. v. Chugai Pharmaceutical Co., 927 F.2d 1200 (Fed. Cir. 1991), which "involved a claim to "'[a] purified and isolated DNA sequence consisting essentially of a DNA sequence encoding human erythropoietin.'" Id. at 1203-04 (quoting U.S. Patent No. 4,703,008, claim 2). "We affirmed that this claim was valid and infringed. Id. at 1219."
In addition, she notes that:
The settled expectations of the biotechnology industry — not to mention the thousands of issued patents — cannot be taken lightly and deserve deference. This outpouring of scientific creativity, spurred by the patent system, reflects a substantial investment of time and money by the biotechnology industry to obtain property rights related to DNA sequences. The type of fundamental alteration in the scope of patentable subject matter argued in this case "risk[s] destroying the legitimate expectations of inventors in their property." Festo Corp. v. Shoketsu Kinzoku Kogyo Kabushiki Co., 535 U.S. 722, 739 (2002).
In this regard, she notes that Isolated DNA claims, at least in the case of Amgen, represent crucial and exceedingly valuable property rights, citing Amgen's EPO product and the basis this product (protected inter alia by patents to isolated DNA molecules) has had for the company's success. Accordingly, Judge Moore asserts that "leaving intact the settled expectaions of property owners is particularly important in light of the large number of property rights involved, both to isolated DNA and to purified natural products generally." Specifically, "[t]o change so substantially the rules of the game now," after more than a century of practice, "'could very well subvert the various balances the PTO sought to strike when issuing the numerous patents which have not yet expired and which would be affected by our decision.'" Festo, 535 U.S. at 739 (quoting Warner-Jenkinson v. Hilton Davis Chem. Co., 520 U.S. 17, 32 n.6 (1997)).
Judge Moore also visits the opinion of the Solicitor General (who had a lively twenty minutes under Judge Moore's questioning (and open skepticism) regarding whether the Justice Department's position was a change in policy that had split the government). Kindly, she notes that:
Certainly the magic microscope has curb appeal — its child-like simplicity an apparent virtue. The magic microscope, however, would not see the claimed DNA molecules at issue in this case. An isolated DNA molecule has different chemical bonds as compared to the "unisolated" sequence in the chromosome (the ends are different). In short, the claimed molecules cannot be seen in nature through the magic microscope. While you may be able to see the order of DNA nucleotides in the chromosome, the isolated fragment of DNA is a different molecule. It may be that the microscope can also break and form chemical bonds to yield the claimed isolated DNA. Even so, the microscope must make some decisions: should the isolated DNA begin and end in a phosphate? a hydrogen? a hydroxyl? a methyl group? an acyl group? These decisions might be obvious to a person of ordinary skill in the art, but they are not inherent to the unisolated sequence as part of the chromosome. Creating the claimed isolated DNA sequences therefore results in a distinctly unnatural molecule.
Her conclusion is simply that "[t]he magic microscope test simply does not work the way the government claims," and moreover, is itself contrary to Supreme Court precedent:
While the magic microscope creates a bright line rule, it presents a poorly defined question: can we "see" the claimed molecule, or something fairly similar, in nature? Even if the scientific imprecision of the test were excusable, the government also asks us to do away with Chakrabarty's flexible inquiry as to whether the invention, as claimed, has "markedly different characteristics from any found in nature" which result in "the potential for significant utility." Id. at 310. Indeed, the bright line magic microscope test actually appears to be contrary to Funk Brothers, since the combination of bacteria in that case was a "new and different composition of non-inhibitive strains," 333 U.S. at 130-31, and therefore not actually present in nature. There may be additional nuance in the government's argument that accounts for this inconsistency, but under my understanding of the magic microscope test, the combination in Funk Brothers would be patentable subject matter.
Judge Moore also sees inconsistencies in the government's profferred test, noting that according to the government's brief "[a] chemical alteration of a bioactive molecule to improve absorption by the body . . . would likely satisfy section 101." United States Amicus Br. 31 n.8. But she sees the isolated DNA molecules here being "the result of a 'chemical alteration of a bioactive molecule' that leads to different properties, including a dramatic reduction in size." In the same way, the hypothetical "chemical alteration" posited by the government produces a (patent-eligible) molecule because it has "improved absorption properties," the isolated DNA claimed by Myriad changes the properties of the gene in the chromosomal DNA. "This is not '[m]erely sorting the proverbial wheat from the chaff,' as the government contends, id., but the creation of new DNA molecules with distinct properties and additional utility, including the ability to be used as a primer in genetic testing."
Judge Moore, in her concurring opinion (as she was at oral argument), is concerned by the "apparent lack of awareness about the impact of the proposed test." Although the Solicitor General claims that the magic microscope represents "a very limited position," Judge Moore disagrees. She recognizes that there is neither a legal nor a scientific basis to limit the application of the government's "magic microscope" to isolated DNA molecules; she posits that "Louis Pasteur's 1873 claim to 'Yeast, free from organic germs of disease, as an article of manufacture' runs afoul of the magic microscope since the microscope could zoom in to see that yeast free from contaminants,'" and that other "isolated naturally occurring molecules" (including adrenaline, vitamin B-12, and prostaglandins," as well as "smaller fragments of naturally occurring molecules, for example claims to truncated proteins (see, e.g., U.S. Patent No. 4,762,914, entitled "Truncated Protein of Interleukin-1") that have been "long considered patentable subject matter" would fail the government's test.
In addition to these practical concerns, Judge Moore contends that "[t]he government's new test fundamentally changes more than a century of precedent and Patent Office practice in the pharmaceutical and biotechnology arena" being "a purely mechanical inquiry that fails to account for the possibility that chemical changes to the isolated DNA sequences at issue, as compared to their natural state, could result in markedly different uses." As a result, adopting the government's position would "call[] into question the validity of an unknown number of patents and claims and upsets the settled expectations of some of our most innovative industries," which she refuses to considern a "very limited position." She also recognizes that the administration could change U.S. patent policy and not affect any existing patent rights, but that the government is asking the court to retroactively invalidate ("destroy existing property rights") thousands of granted patents containing claims to isolated DNA molecules.
Judge Moore believes that the proper place to debate such a drastic change in the law is Congress, and she refuses to "ignore the years of Patent Office practice and the accompanying expectations that practice created within the industry." She refuses to do so, despite the "temptation" to rule on matters that "raise substantial moral and ethical issues related to awarding a property right to isolated portions of human DNA — the very thing that makes us humans, and not chimpanzees" because to do so would "usurp Congress's constitutionally mandated authority to promote science and useful arts." Such "[j]udicial restraint is particularly important here because an entire industry developed in the decades since the Patent Office first granted patents to isolated DNA. Disturbing the biotechnology industry's settled expectations now risks impeding, not promoting, innovation." Continuing in this vein, Judge Moore finishes her concurrence by remarking that "[u]nsettling the expectations of the biotechnology industry now, based on nothing more than unsupported supposition, strikes me as far more likely to impede the progress of science and useful arts than advance it. Given the complicated technology and conflicting incentives at issue here, any change must come from Congress" (noting the several times legislation has been introduced limiting the scope of 101 to exclude patenting genetic material, and that Congress has passed none of them). And she closes her opinion by citing to one of the lions of patent law in the 20th Century, saying that "[t]his case typifies an observation by the late Chief Judge Markey, our first Chief Judge, that "[o]nly God works from nothing. Men must work with old elements." Fromson v. Advance Offset Plate, Inc., 755 F.2d 1549, 1556 n.3 (Fed. Cir. 1985) (quotation, citations omitted). Judge Moore concludes:
Human DNA is, for better or worse, one of the old elements bequeathed to men to use in their work. The patents in this case revealed a new molecular understanding about ourselves; "the inventions most benefiting mankind are those that 'push back the frontiers of chemistry, physics, and the like.'" Chakrabarty, 447 U.S. at 316 (quoting Great A. & P. Tea Co. v. Supermarket Corp., 340 U.S. 147, 154 (1950)). We cannot, after decades of patents and judicial precedent, now call human DNA fruit from the poisonous tree, and punish those inquisitive enough to investigate, isolate, and patent it. "Our task . . . is the narrow one of determining what Congress meant by the words it used in the statute; once that is done our powers are exhausted." Id. at 318. This inquiry does not have moral, ethical, or theological components. Cf. id. at 316-17 ("[W]e are without competence to entertain" arguments about "the grave risks" generated by genetic research.). The patents in this case might well deserve to be excluded from the patent system, but that is a debate for Congress to resolve. I therefore decline to extend the "laws of nature" exception to include isolated DNA sequences.
Association for Molecular Pathology v. U.S. Patent and Trademark Office (Fed. Cir. 2011)
Panel: Circuit Judges Lourie, Bryson, and Moore
Opinion for the court by Circuit Judge Lourie; opinion concurring in part by Circuit Judge Moore; opinion concurring in part and dissenting in part by Circuit Judge Bryson
 Medicis has entered into multiple agreements with Lupin over the acne drug Solodyn, allowing the generic drugmaker to enter the market prior to the expiration of Medicis' patents. In a separate agreement, Medicis agreed to join Lupin in a drug development collaboration worth an initial $20 million to the generic company.
Medicis has entered into multiple agreements with Lupin over the acne drug Solodyn, allowing the generic drugmaker to enter the market prior to the expiration of Medicis' patents. In a separate agreement, Medicis agreed to join Lupin in a drug development collaboration worth an initial $20 million to the generic company. On July 21, Judge J. Frederick Motz signed off on a consent judgment that permanently enjoined Lupin from selling generic Solodyn except under the terms of a separate licensing agreement. The licensing agreement allows Lupin to sell a generic version of Solodyn in various different doses prior to the expiration of the patents. Under the agreement, Lupin can move ahead with 45-, 90-, and 135-milligram doses in November, the 65- and 115-milligram doses in February 2018, and the 55-, 80-, and 105-milligram doses in February 2019. Under certain conditions that were not disclosed, Lupin could begin making the generic earlier.
On July 21, Judge J. Frederick Motz signed off on a consent judgment that permanently enjoined Lupin from selling generic Solodyn except under the terms of a separate licensing agreement. The licensing agreement allows Lupin to sell a generic version of Solodyn in various different doses prior to the expiration of the patents. Under the agreement, Lupin can move ahead with 45-, 90-, and 135-milligram doses in November, the 65- and 115-milligram doses in February 2018, and the 55-, 80-, and 105-milligram doses in February 2019. Under certain conditions that were not disclosed, Lupin could begin making the generic earlier. A $577 million award secured by Asahi against Actelion has been cut by $70 million. In its suit, Asahi claimed that Actelion bought CoTherix in order to undermine a licensing deal over the hypertension treatment Fasudil.
A $577 million award secured by Asahi against Actelion has been cut by $70 million. In its suit, Asahi claimed that Actelion bought CoTherix in order to undermine a licensing deal over the hypertension treatment Fasudil. Asahi brought suit in 2008 in the Superior Court of the State of California, County of San Mateo, accusing Actelion of acquiring CoTherix and then interfering with the latter's deal with Asahi to develop Fasudil. CoTherix terminated the licensing deal for Fasudil in 2007 after being acquired by Actelion. Asahi alleged that Actelion's acquisition of CoTherix was an attempt to protect its monopoly in the market for treating pulmonary arterial hypertension. Actelion maintained that there was no commitment between Asahi and CoTherix, noting that the licensing agreement explicitly stated that there was no partnership. During a three-month trial, Asahi argued that Actelion expected a major loss in market share if competitive drugs were introduced, for Acetlion's drug Tracleer had caused 3,427 patient deaths that had gone unreported to the FDA. Actelion argued that the decision not to develop Fasudil was made due to numerous safety concerns from doctors and medical authorities as to the drug's potential side effects on the kidney and the liver. In May 2011, the jury found that Actelion and CoTherix had acted with malice in interfering with Asahi's 2006 agreement with CoTherix to develop and commercialize Fasudil, and awarded Asahi $550 million in damages for interrupting the licensing agreement.
Asahi brought suit in 2008 in the Superior Court of the State of California, County of San Mateo, accusing Actelion of acquiring CoTherix and then interfering with the latter's deal with Asahi to develop Fasudil. CoTherix terminated the licensing deal for Fasudil in 2007 after being acquired by Actelion. Asahi alleged that Actelion's acquisition of CoTherix was an attempt to protect its monopoly in the market for treating pulmonary arterial hypertension. Actelion maintained that there was no commitment between Asahi and CoTherix, noting that the licensing agreement explicitly stated that there was no partnership. During a three-month trial, Asahi argued that Actelion expected a major loss in market share if competitive drugs were introduced, for Acetlion's drug Tracleer had caused 3,427 patient deaths that had gone unreported to the FDA. Actelion argued that the decision not to develop Fasudil was made due to numerous safety concerns from doctors and medical authorities as to the drug's potential side effects on the kidney and the liver. In May 2011, the jury found that Actelion and CoTherix had acted with malice in interfering with Asahi's 2006 agreement with CoTherix to develop and commercialize Fasudil, and awarded Asahi $550 million in damages for interrupting the licensing agreement. Following the damage award verdict, Actelion asked Judge Marie S. Weiner to offset a $78.4 million arbitration payment CoTherix made in 2009 from the jury verdict. Judge Weiner ordered a $70.4 million offset. Actelion also contested part of the verdict, asking the judge for an election between lost profit and development costs. The $577 million jury award consisted of $360 million in lost profits, $187 million in development costs, and $30 million in punitive damages. Judge Weiner rejected the motion on procedural grounds, telling Actelion that it must wait until judgment before filing its motion. Actelion has stated its intention to appeal the jury verdict in due course following entry of judgment, expected after August 8, and rulings on further post-trial motions.
Following the damage award verdict, Actelion asked Judge Marie S. Weiner to offset a $78.4 million arbitration payment CoTherix made in 2009 from the jury verdict. Judge Weiner ordered a $70.4 million offset. Actelion also contested part of the verdict, asking the judge for an election between lost profit and development costs. The $577 million jury award consisted of $360 million in lost profits, $187 million in development costs, and $30 million in punitive damages. Judge Weiner rejected the motion on procedural grounds, telling Actelion that it must wait until judgment before filing its motion. Actelion has stated its intention to appeal the jury verdict in due course following entry of judgment, expected after August 8, and rulings on further post-trial motions. Pfizer could be wrapping up its infringement lawsuits over the migraine drug Relpax, as it reached a settlement agreement with Apotex, the deal with Apotex following a settlement Pfizwer reached with Teva earlier this month. Pfizer filed suit against Apotex on July 9, 2010 in the District Court of the Southern District of New York, after Apotex filed an ANDA to manufacture a generic form of eletripian hydrobromide, which Pfizer markets as Relpax. Pfizer claimed that Apotex's generic would infringe U.S. Patent No. 6,110,940. Pfizer sought a judgment of infringement and a permanent injunction against Apotex, along with attorneys' fees and costs. Apotex asserted that its generic version did not infringe the '940 patent because it uses a different form of eletripian hydrobromide. In an answer and counterclaim filed in September, Apotex requested a declaratory judgment of invalidity and noninfringement of the '940 patent.
Pfizer could be wrapping up its infringement lawsuits over the migraine drug Relpax, as it reached a settlement agreement with Apotex, the deal with Apotex following a settlement Pfizwer reached with Teva earlier this month. Pfizer filed suit against Apotex on July 9, 2010 in the District Court of the Southern District of New York, after Apotex filed an ANDA to manufacture a generic form of eletripian hydrobromide, which Pfizer markets as Relpax. Pfizer claimed that Apotex's generic would infringe U.S. Patent No. 6,110,940. Pfizer sought a judgment of infringement and a permanent injunction against Apotex, along with attorneys' fees and costs. Apotex asserted that its generic version did not infringe the '940 patent because it uses a different form of eletripian hydrobromide. In an answer and counterclaim filed in September, Apotex requested a declaratory judgment of invalidity and noninfringement of the '940 patent. On July 26, Pfizer's attorneys filed a letter with Judge Lewis A. Kaplan, informing him that the companies had reached a settlement. According to the letter, the parties sought to avoid further judicial involvement, and achieve the same result as with Teva. Terms of Pfizer's settlement with Teva were not disclosed. Pfizer also asked Judge Kaplan to extend discovery deadlines in the case, in order to provide adequate time for the parties to close on a final settlement agreement.
On July 26, Pfizer's attorneys filed a letter with Judge Lewis A. Kaplan, informing him that the companies had reached a settlement. According to the letter, the parties sought to avoid further judicial involvement, and achieve the same result as with Teva. Terms of Pfizer's settlement with Teva were not disclosed. Pfizer also asked Judge Kaplan to extend discovery deadlines in the case, in order to provide adequate time for the parties to close on a final settlement agreement.