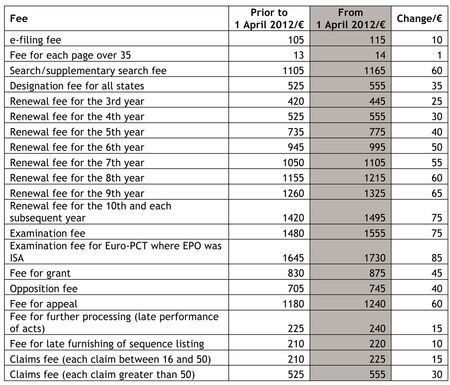
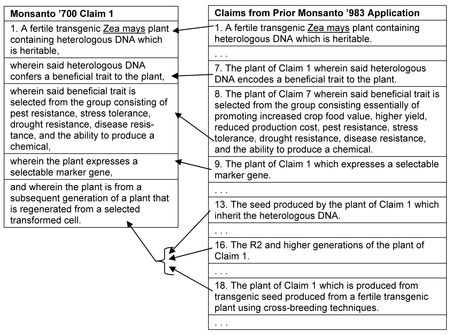
By Kevin E. Noonan —

On Friday, Judge Naomi Reice Buchwald presiding in the Southern District of New York granted Monsanto's motion to dismiss a case brought by the Public Patent Foundation (PubPat) on behalf of "farmers and seed businesses, both organic and non-organic, as well as related membership organizations." Organic Seed Growers and Trade Assn. et al. v. Monsanto Co. This puts to an end (for now) PubPat's latest effort to influence U.S. patent policy through the courts (the other instance being, most (in)famously, Association for Molecular Pathology v. U.S. Patent and Trademark Office ("the Myriad case"), although here PubPat was not aided by the American Civil Liberties Union). The complaint named almost seventy plaintiffs, and the patents at issue were U.S. Patent Nos. 5,322,938 (expired 6/21/2011), 5,532,605 (expired 7/28/2008), 5,362,865 (expired 11/8/2011), 5,378,619 (expired 1/3/2012), 5,424,412 (will expire 6/13/2012), 5,463,175 (will expire 10/31/2012), 5,530,196, 5,554,798, 5,593,874, 5,641,876, 5,659,122, 5,717,084, 5,728,925, 5,750,871, 5,859,347, 6,025,545, 6,040,497, 6,051,753, 6,083,878, 6,753,463, and 6,825,400, and U.S. Reissue Patent Nos. RE38825 and RE39247.
Monsanto asked the Court to dismiss the action for failure to allege a "case or controversy" under the Declaratory Judgments Act as required under Article III of the Constitution. The District Court began its opinion granting this motion by noting that a court's jurisdiction cannot exceed this Article III requirement under the Declaratory Judgment Act, citing Prasco, LLC v. Medicis Pharm. Corp., 537 F.3d 1329, 1335 (Fed. Cir. 2008). The Supreme Court's test for determining whether a sufficient case or controversy has been plead requires a plaintiff to establish that "the facts alleged, under all the circumstances, show that there is a substantial controversy, between parties having adverse legal interests, of sufficient immediacy and reality to warrant the issuance of a declaratory judgment," citing (without apparent irony) Association for Molecular Pathology v. U.S. Patent & Trademark Office ("AMP"), 653 F.3d 1329, 1342-43 (Fed. Cir. 2011) (quoting MedImmune, Inc. v. Genentech, Inc., 549 U.S. 118, 127 (2007)). The Court noted that this test has been "refined" by the Federal Circuit to require "an injury in fact traceable to the patentee," which only exists if plaintiffs have alleged "both (1) an affirmative act by the patentee related to the enforcement of his patent rights, and (2) meaningful preparation to conduct potentially infringing activity." Id. at 1343 (citing SanDisk Corp. v. STMicroelectronics, Inc., 480 F.3d 1372, 1380-81 (Fed. Cir. 2007).
The opinion notes (in footnote 5) that:
[P]laintiffs attempt to circumvent this test first by arguing that MedImmune confers per se standing on any plaintiff who has purchased a license to a patent. (Pls.' Mem. 18.) The argument evinces a lack of understanding of MedImmune's holding, which was that purchasing a patent license does not preclude jurisdiction where the purchase has been coerced. See 549 U.S. at 129-31. Plaintiffs, relying on their misreading of MedImmune, next endeavor to extend the argument and contend that standing should be conferred on all parties who are able to purchase a license, whether or not they have done so. (Pls.' Mem. 19.) Accepting this suggestion would functionally eliminate the case or controversy requirement and should thus be rejected out of hand.

The Court explained that the first prong of the Federal Circuit's test ensures that the parties have "adverse legal interests," which is established "where a patentee asserts rights under a patent based on certain identified ongoing or planned activity of another party, and where that party contends that it has the right to engage in the accused activity without license." SanDisk, 480 F.3d at 1381. (The Court recognized that "it is possible that the first prong of the test may be satisfied upon some lesser showing.") In any case, there must be "an underlying legal cause of action that the declaratory defendant could have brought or threatened to bring, if not for the fact that the declaratory plaintiff had preempted it," because otherwise "any adverse economic interest that the declaratory plaintiff may have against the declaratory defendant is not a legally cognizable interest sufficient to confer declaratory judgment jurisdiction," citing Creative Compounds, LLC v. Starmark Labs., 651 F.3d 1303, 1316 (Fed. Cir. 2011). The "mere existence of a potentially adverse patent does not cause an injury [or] create an imminent risk of an injury; absent action by the patentee, a potential [infringer] is legally free to market its product in the face of an adversely-held patent," citing Prasco, 537 F.3d at 1338.
As to the second prong, the Federal Circuit's test requires that the declaratory judgment plaintiff has taken "significant, concrete steps to conduct infringing activity"; otherwise, "the dispute is neither 'immediate' nor 'real' and the requirements for justiciability have not been met," citing Cat Tech LLC v. TubeMaster, Inc., 528 F.3d 871, 880 (Fed. Cir. 2008). The Court notes that "the greater the length of time before potentially infringing activity is expected to occur" is "significant" and impacts the immediacy of the cause of action.
Here, the Court held that plaintiffs failed to assert a "case or controversy" that supported Article III jurisdiction. The facts supporting the Court's conclusion were that there was no evidence that Monsanto "ever demanded royalty payments from plaintiffs, identified any of plaintiffs' conduct as potentially infringing, or even initiated any contact with plaintiffs whatsoever." Plaintiffs only support for there being a "case or controversy," according to the Court, was "(1) defendants' pattern of enforcing their patent rights against non-plaintiff farmers through litigation or threats of litigation; (2) plaintiffs' assertion of the 'implicit threat' in defendants' statement that it is not their policy to enforce their patent rights against farmers whose crops inadvertently acquire trace amounts of patented seeds or traits; and (3) defendants' refusal to provide plaintiffs with a blanket covenant not to sue."
These reasons were not sufficient in the Court's opinion. The first reason, that Monsanto has a pattern of enforcing its patent rights through litigation or threat of litigation, was rejected by the Court because it did not include evidence that Monsanto had exercised its patent rights against the plaintiffs. While conceding that suits against third parties could support declaratory judgment jurisdiction, the Court noted that these circumstances required that suits against such third parties must be "sufficiently similar" to one that a patentee "may bring" against the plaintiffs (put another way, against "similarly situated parties"). The reason, according to the opinion, is that "a fear of future harm that is only subjective is not an injury or threat of injury caused by the defendant that can be the basis of an Article III case or controversy," citing Prasco, 537 F.3d at 1338. This is evaluated objectively, wherein "it is the reality of the threat of injury that is relevant to the standing inquiry, not the plaintiff's subjective apprehensions." Id. at 1338-39 and Hewlett-Packard Co. v. Acceleron LLC, 587 F.3d 1358, 1363 (Fed. Cir. 2009). Specifically, the Court determined that the 144 patent infringement suits filed by Monsanto between 1997 and 2010 were not excessive, comparing the frequency of thirteen lawsuits a year with the "approximately two million" farms in America. In addition, these suits were not against "similarly situated plaintiffs" in the Court's view, since the declaratory judgment plaintiffs here averred that they had no interest in having their seed "contaminated by transgenic seed"; the defendants in Monsanto's other actions were farmers who infringed Monsanto's patents by replanting seeds or breaching their contractual obligations. There was "no evidence that defendants have commenced litigation against anyone standing in similar stead to plaintiffs and thus the evidence did not support the 'affirmative acts' prong of the Federal Circuit's test for declaratory judgment jurisdiction" according to the opinion. And any allegations by plaintiffs that Monsanto had threatened "similarly situated" farmers who wished to avoid inadvertent use of Monsanto's patented seed were, according to the Court, "equally lame" because "not one single plaintiff claims to have been so threatened."

The Court also rejected plaintiffs' argument that Monsanto's actions contained an "implicit" threat of litigation. This allegation stemmed from Monsanto's statements. In response to a post-filing letter from PubPat demanding a covenant not to sue in order to dismiss the lawsuit, and similar statements on Monsanto's website. The "purportedly threatening languages reads (in its entirety)":
It has never been, nor will it be[,] Monsanto policy to exercise its patent rights where trace amounts of our patented seeds or traits are present in [a] farmer's fields as a result of inadvertent means.
The Court found plaintiffs' characterization of this language as threatening to be "objectively unreasonable." This determination is based on plaintiffs' avowed intention to not "use or sell transgenic seeds," and that plaintiffs' intention was "specifically communicated" to Monsanto. If these intentions were true, "the presence of patented traits in plaintiffs' seeds could only be inadvertent," according to the Court, and the possibility that plaintiffs could somehow "find themselves unknowingly utilizing it in significant quantities strains credulity." The Court finds the "plain meaning" of Monsanto's statement to be clear, and that the Court would not adopt plaintiffs' "deliberate misreading" thereof. Further:
Indeed, plaintiffs' letter to defendants seems to have been nothing more than an attempt to create a controversy where none exists. This effort to convert a statement that defendants have no intention of bringing suit into grounds for maintaining a case, if accepted, would disincentivize patentees from ever attempting to provide comfort to those whom they do not intend to sue, behavior which should be countenanced and encouraged. In contrast, plaintiffs' argument is baseless and their tactics not to be tolerated.
Remarkably, the Court noted that these statements were made after filing the initial complaint, in view of the fact that it is impermissible for the Court to consider such conduct in evaluating whether declaratory judgment jurisdiction exists. The policy reason for this rule is that it would "invite a declaratory judgment plaintiff in a patent case to file suit at the earliest moment it conceives of any potential benefit to doing so" in an attempt to "draw an infringement suit in response (thereby retroactively establishing jurisdiction over their first-filed declaratory judgment suit)," said the Court, citing Innovative Therapies, Inc. v. Kinetic Concepts, Inc., 599 F.3d 1377, 1384 (Fed. Cir. 2010). This is exactly what plaintiffs are attempting here, according to the opinion, which is "a further reason to discount their argument."
Finally, the Court deigned not to credit any effect of Monsanto's refusal to execute a covenant not to sue in plaintiffs' favor. Calling it a request for a "blanket waiver," the Court found it "unsurprising" that Monsanto declined plaintiffs' invitation to enter into such an agreement. Plaintiffs' argument was "groundless and their tactics unacceptable," the Court held. Monsanto's refusal to enter into the covenant was but one factor to be considered under Federal Circuit precedent and here "does not meaningfully add to plaintiffs' case." According to the opinion, "plaintiffs' letter was clearly intended to be used as a prop in this litigation, and the failure to sign a covenant not to sue borders on the wholly irrelevant."
The Court also found no evidence of the required "preparatory conduct" needed under the second prong of the Federal Circuit test. In this case, plaintiffs contended that there was no requirement for them to take affirmative action to infringe, since "defendants' patented seeds will spread with no action on plaintiffs' part and are self-replicating." Regardless of the accuracy of these allegations, the Court said that the "potential infringement " must be "a matter of immediate concern," and that plaintiffs' allegations fail in this regard. The opinion notes that not only have plaintiffs not alleged that any of them have "actually grown or sold contaminated seed," they have maintained their intention to "specifically avoid any such use." They do, however, allege that they "could . . . be accused of patent infringement" due to contamination of their seed with Monsanto's seed. The Court characterized this allegation as the sort of "intangible worry, unanchored in time" that the Federal Circuit has found "insufficient to support an 'actual or imminent' injury for standing without any specification of when the some day will be," citing AMP, 653 F.3d at 1346. Addressing plaintiffs' allegation that they are suffering injury because some of them have stopped planting certain crops due to fear of a lawsuit, the Court called this an injury of their own making that did not support declaratory judgment jurisdiction.
Finally, considering all the circumstances, the Court granted Monsanto's motion to dismiss, calling plaintiffs' allegations "diaphanous" and the complaint "a transparent effort to create a controversy where none exists."