By Kevin E. Noonan —
 In April, the White House released a policy paper what was styled the "National Bioeconomy Blueprint." Its presumptions are based on the idea that the portion of the economy "fueled by research and innovation in the biological sciences" is a "large and rapidly growing segment of the world economy that provides substantial public benefit." This has caused innovation in the biological sciences to become a priority of the Obama Administration, promising not only economic development but to "live longer, healthier lives, reduce our dependence on oil, address key environmental challenges, transform manufacturing processes, and increase the productivity and scope of the agricultural sector while growing new jobs and industries." Successes of the bioeconomy touted by the Blueprint include $76 billion in revenues from genetically engineered crops and $100 billion in revenues from "industrial biotechnology" (including "fuels, materials, chemicals, and industrial enzymes derived from genetically modified systems"). The Blueprint identifies genetic engineering technology, DNA sequence analysis and "automated high-throughput manipulations of biomolecules" as "foundational technologies," while identifying "synthetic biology," proteomics and bio-informatics as "emerging technologies."
In April, the White House released a policy paper what was styled the "National Bioeconomy Blueprint." Its presumptions are based on the idea that the portion of the economy "fueled by research and innovation in the biological sciences" is a "large and rapidly growing segment of the world economy that provides substantial public benefit." This has caused innovation in the biological sciences to become a priority of the Obama Administration, promising not only economic development but to "live longer, healthier lives, reduce our dependence on oil, address key environmental challenges, transform manufacturing processes, and increase the productivity and scope of the agricultural sector while growing new jobs and industries." Successes of the bioeconomy touted by the Blueprint include $76 billion in revenues from genetically engineered crops and $100 billion in revenues from "industrial biotechnology" (including "fuels, materials, chemicals, and industrial enzymes derived from genetically modified systems"). The Blueprint identifies genetic engineering technology, DNA sequence analysis and "automated high-throughput manipulations of biomolecules" as "foundational technologies," while identifying "synthetic biology," proteomics and bio-informatics as "emerging technologies."
The result of the "bioeconomy" becoming an Administration "priority" is a directive (Executive Memorandum (M-10-30)) to all Federal agencies to "support research to establish the foundations for a 21st century bioeconomy," an effort that the Blueprint says has made "significant early progress." While these efforts also "raise important ethical and security issues that are also top priorities for the Administration," discussions of these concerns are expressly deemed to be outside the scope of the Blueprint itself.
The first chapter of the Blueprint sets out the broad goals, citing the National Research Council's 2009 report, A New Biology for the 21st Century, for a recognition of the importance of biological research for addressing societal needs for foods, fuel, and medicines (echoing BIO's motto of "Healing, Fueling and Feeding the World"). The Blueprint enunciates "key elements" of the recommended national effort, including:
• a full spectrum of basic and applied R&D activities performed by academic, government, and private sectors
• public-private partnerships
• a supportive commercialization system for bioinventions
• innovative regulatory policies that reflect government awareness of needs for and impediments to progress
• a skilled and creative workforce
• public support for technological advances
• flexibility to accommodate the evolving needs, discoveries, and challenges
The second chapter enumerates several examples of efforts to achieve the goals.
The Blueprint sets forth "five strategic objectives" for achieving the promised bioeconomy:
1. Support R&D investments that will provide the foundation for the future U.S. bioeconomy.
This objective includes "[c]oordinated, integrated R&D efforts will help strategically shape the national bioeconomy R&D agenda"; "[e]xpand[ing] and [d]eveloping essential bioeconomy technologies"; "[i]ntegrat[ing] approaches across fields"; and "[i]mplement[ing] Improved Funding Mechanisms." In this effort, "the President has called for agencies to identify strategic R&D investments, as well as increase the use of flexible funding mechanisms to improve program efficiency and provide the best opportunity to enhance economic growth." The rationale is that "[a] robust biological/biomedical R&D enterprise, backed by government, foundations, and for profit investments, is necessary to produce the new knowledge, ideas, and foundational technologies required to develop products and services that support businesses and industries and help create jobs."
Much of what is cited in the Blueprint are efforts like the NIH Center for Regenerative Medicine, to accelerate the development and testing of new clinical protocols using iPS-cell-based treatments, and the use of FDA data on drug products clinical data ("[t]he FDA currently houses one of the largest known repositories of clinical data, including safety, efficacy, and performance information, and an increasing amount of post-market safety surveillance data"). While technology-based, such efforts do raise their own questions, such as whether translational activities are the best use of NIH resources and the extent to which clinical and other data submitted in support of regulatory approval can be expropriated by the government for its own, alternative uses without raising constitutional concerns. The Blueprint also cites several biofuels efforts by Advanced Research Projects Agency-Energy, Department of Agriculture (USDA) and Department of Energy's (DOE) Biomass Program, USDA-DOE Plant Feedstock Genomics for Bioenergy Program, National Science Foundation's (NSF) Engineering Directorate Sustainable Energy Pathways (SEP) effort, as well as government programs supporting biomanufacturing.
2. Facilitate the transition of bioinventions from research lab to market, including an increased focus on translational and regulatory sciences.
This objective includes "[a] dedicated commitment to translational efforts will accelerate movement of bioinventions out of laboratories and into markets, specifically: [a]ccelerat[ing] progress to market; enhance[ing] entrepreneurship at eniversities; and utilize[ing] federal procurement authority." The Blueprint identifies programs intended to help start-ups negotiate the "valley of death" of investment, including the Startup America initiative, specifically naming "five areas highlighted for action: unlocking access to capital; connecting mentors with entrepreneurs; reducing regulatory barrier; tax relief; and other economic incentives for small businesses"; the National Institute of Standards and Technology (NIST) Hollings Manufacturing Extension Partnership (MEP); the National Innovation Marketplace; BioInnovation Initiative; Executive Order 13514, Federal Leadership in Environmental, Energy, and Economic Performance; reauthorization of the Small Business Innovation Research (SBIR) and Small Business Technology Transfer (STTR) programs; translational biomedicine efforts such as the National Center for Advancing Translational Sciences (NCATS), established in December 2011, and cooperative programs between FDA and NIH. In addition, the Blueprint advocates enhanced entrepreneurial activities in universities and improved technology transfer, which includes exclusive licensing of NIH-developed innovation, "to companies developing drugs, vaccines, or therapeutics from NIH-patented or patent-pending technologies." But the Bleuprint also contains statements like "[t]he America Invents Act provides entrepreneurs the tools they need to obtain patents more quickly and to defend them against litigation challenges, both at lower costs" which reduces a reader's confidence that the Administration has accurately assessed the important components of bio-innovation.
3. Develop and reform regulations to reduce barriers, increase the speed and predictability of regulatory processes, and reduce costs while protecting human and environmental health.
This objective includes "[i]mproved regulatory processes [that] will help rapidly and safely achieve the promise of the future bioeconomy, specifically: improve[ing] regulatory processes and regulations; and collaborat[ing] with stakeholders."
This initiative is actually directed to "reducing regulatory barriers": specifically, regulations that "have become inadequate or unnecessarily restrictive because technology and its associated products and services, as well as our national interests, have evolved and regulations may not have kept pace." The Blueprint also cites Executive Orders, Improving Regulation and Regulatory Review and Regulation and Independent Regulatory Agencies and revisions to FDA regulations that "increase transparency, consistency, and predictability of the regulatory processes and help drive medical product innovation forward." Specific regulatory changes are directed to medical devices and regulation for "emerging technologies."
4. Update training programs and align academic institution incentives with student training for national workforce needs.
This objective includes the directive that "Federal agencies should take steps to ensure that the future bioeconomy has a sustainable and appropriately-trained workforce," specifically enumerating "employer-educator partnerships" and "reengineer[ing] training programs." A large portion of this objective is directed towards an emphasis on minority-serving institutions (including community colleges).
5. Identify and support opportunities for the development of public-private partnerships and precompetitive collaborations — where competitors pool resources, knowledge, and expertise to learn from successes and failures.
This objective includes the directive that "Federal agencies should provide incentives for public-private partnerships and precompetitive collaborations to benefit the bioeconomy broadly, specifically directed to "catalyz[ing] public-private partnerships." "Models" for collaboration are listed as:
• Open-source initiatives
• Industry consortia for process innovation
• Discovery-enabling consortia
• Public–private consortia for
knowledge creation
• Prizes
• Innovation incubators
• Industry complementors
• Virtual pharmaceutical companies
Citing "Overview of Precompetitive Collaboration for Institute of Medicine Workshop."
This portion of the Blueprint sets out public-private partnerships for "rescuing" or repurposing" abandoned pharmaceutical compounds and smarter drug design.
The next portion of the Blueprint sets forth purported "successes" of the Administration's efforts to stimulate the bioeconomy, which include:
• "Broad Spectrum Anti-Viral: Researchers at the Massachusetts Institute of Technology's Lincoln Laboratory reported in 2011 creation of a broad-spectrum antiviral technology that selectively kills any virus-infected cell but does not harm uninfected cells." ("Broad-Spectrum Antiviral Therapeutics")
• "Diesel from CO2: Through photosynthesis, plants, algae, and some bacteria use the energy of sunlight to convert CO2 into a variety of organic compounds needed for growth and survival A Massachusetts-based company re-engineered photosynthetic organisms to synthesize, from sunlight and CO2, molecules that form the chemical basis of diesel fuel."
• "Designing Biological Systems for Next- Generation Biomanufacturing: Synthetic biology is enabling scientists to rapidly design organisms that can be used in the production of renewable chemicals, biofuels, renewable specialty and fine chemicals, food ingredients, and health-care products. Bioacrylic acid heralds the advent of synthetic-biology-enabled manufacturing: acrylic acid ingredients are used to make adhesives stronger, paints more durable, and diapers more absorbent, and today petroleum-based acrylic is an $8 billion global market" ("Current Uses of Synthetic Biology")
• "Allergen-Free Peanuts: Researchers at a number of institutions in the United States have made inroads towards eliminating or inactivating allergenic proteins in peanuts. If successful, these approaches could lead to significant health benefits for Americans and economic opportunities for the peanut industry."
• "Biodegradable Plastics from Biomass: A major commercial "polylactic acid" bioplastic is already made today from cellulose However, the ability to replace petroleum-based plastics with this bioproduct is constrained by the limited availability of the specific cellulosic source material. To address this limitation, USDA scientists discovered a bacterium that can ferment a broader range of cellulosic biomass materials into polylactic acid, enabling commercial production on a much larger scale. With an estimated $375 billion market for chemical, plastic, and rubber products based on petroleum, this represents a substantial bioeconomy opportunity." ("Sustainable Polymers")
• "Biosensor Pollution Monitoring: Biosensors are devices composed of a biological sensing component linked
to a signaling component, working together to reveal the presence of an element, molecule, or organism of interest. In 2011, a team at the Virginia Institute of Marine Science reported creation of a portable biosensor that could detect marine pollutants, including oil, much faster and more cheaply than current technologies. If deployed near oil facilities, such sensors could provide early warning of spills and leaks and track dispersal patterns in real time." ("Near real-time, on-site, quantitative analysis of PAHs in the aqueous environment using an antibody-based biosensor")
More naïve (or perhaps idealist) portions of the Blueprint posit "trends" in the bioeconomy such as "sharing:"
Non-traditional research collaborations that feature the
sharing of information, resources, and capabilities are
transforming the bioeconomy. Precompetitive collabora
tions — where "competitors" partner and pool resources — are growing as partners seek new ways to leverage constrained resources and surmount shared problems. Partnerships for innovation are increasingly observed as a response to changing economic and technological conditions.
In agriculture, as the public and private sectors seek increased information for the bioeconomy on potential crop characteristics, there is increased sharing, both domestically and internationally, of genetic information. The sharing of genetic information enhances U.S. agricultural competitiveness for food, energy, chemical production in plants, and other biobased-product crop species.
In the health sector, precompetitive collaborations are having significant impacts in clinical-trial design and biomarker discovery, among other areas. Combined industrial R&D has contributed to transformative progress despite major challenges such as increased management costs due to "outside" collaborations and the need to develop effective communication networks across companies.
Similarly idealistic (or perhaps fantastical) portions of the Bleuprint include the idea for "using the power of prizes to drive innovation," specifically with regard to enactment of the "America COMPETES Reauthorization Act[,] granting all Federal agencies broad authority to conduct prize competitions as called for by the President" in December 2011 ("Implementation of Federal Prize Authority: Progress Report").
The Blueprint contains some important information, such as the "30-year decline in new molecular entities per dollar spent on R&D":
 Figure 1. 30-year decline in new molecular entities per dollar spent on R&D There has been a 30-year decline in pharmaceutical industry productivity, as measured by new molecular entities per dollar spent
on R&D, normalized to 5-year rolling average of 1970
to 1975 While R&D costs have increased 50-fold during this time period, the output of investigational new drug candidates and new drug application products has stayed flat. Figure Source: IOM (Institute of Medicine) 2010 Extending the spectrum of precompetitive collaboration in oncology research: Workshop summary Washington, DC: The National Academies Press.
Figure 1. 30-year decline in new molecular entities per dollar spent on R&D There has been a 30-year decline in pharmaceutical industry productivity, as measured by new molecular entities per dollar spent
on R&D, normalized to 5-year rolling average of 1970
to 1975 While R&D costs have increased 50-fold during this time period, the output of investigational new drug candidates and new drug application products has stayed flat. Figure Source: IOM (Institute of Medicine) 2010 Extending the spectrum of precompetitive collaboration in oncology research: Workshop summary Washington, DC: The National Academies Press.
The Blueprint interprets this data, by asserting that "[o]ne issue that government and industry must address is that increased investment in research has not resulted in a concomitant increase in approved drugs. Figure 1 shows that the number of FDA-approved new molecular entities has not kept pace with the pharmaceutical sector's spending over time have cited internal drug pipeline productivity declines, delays in application review caused by both industry and regulators, and a decreasing number of tractable medical challenges as reasons for this decline in productivity. Advances in translational and regulatory sciences, improvements in the transfer of technology from the public to the private sector, regulatory process improvements, Federal workforce enhancements, and innovative public-private partnerships have potential to help overcome these challenges."
On the other hand, the Blueprint identifies certain sectors of the bioeconomy that are rapidly advancing, such as genomic sequencing as illustrated in the following Figure:
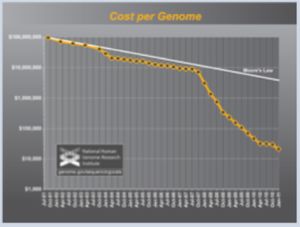
Figure 2. "Cost per Genome" — the cost of sequencing a human-sized genome Data from 2001 through October 2007 represent the costs of generating DNA sequence using first generation sequencing technology. Beginning in January 2008, the data represent the costs of generating DNA sequence using 'second-generation' (or 'next-generation') sequencing platforms. The change in instruments represents the rapid evolution of DNA sequencing technologies that has occurred in recent years. www genome gov/sequencingcosts.
The Blueprint asserts that "[w]hile the sequencing of the first human genome took 13 years and cost $2.7 billion, researchers can now sequence a human genome for a fraction of that cost (~$7,700) and within two weeks' time" and considers the implications of these rapid increases in speed and decreases in cost for "whole-genome" human genomic sequencing.
The Blueprint concludes:
The Administration has made great strides in harnessing biological research innovations to address national challenges in health, energy, food, environment, and manufacturing via the commercial economy. But there is much more to be done. The Administration plans to explore additional creative ideas for promoting U. S. leadership in the bioeconomy, such as innovative financing for translational research, increasing the impact of the SBIR program, and improving the ability of faculty and students to move from "idea to IPO," and will continue to accept public input by email at bioeconomy@ostp gov.
By strategically shaping future R&D investments, improving commercialization of bioinventions, updating workforce training programs for new bioeconomy careers, reforming regulatory processes, and building new bioeconomy public-private partnerships, the Administration will help stimulate the growth of a high-wage, high-skill sector while improving the lives of all Americans.
Politics aside, publication of the Blueprint indicates that the Obama Administration recognizes the importance of the bioeconomy to the nation's economy as a whole. Provided that the government is receptive to the input and information from all stakeholders (and not just government policy wonks, academics or think tank, armchair entrepreneurs, the Blueprint may actually provide a roadmap for progress.
 The U.S. Patent and Trademark Office has announced a revised agenda for the next biotechnology/ chemical/pharmaceutical (BCP) customer partnership meeting to be held on June 6, 2012. New to the agenda is an afternoon session on the Recommendation for the Disclosure of Sequence Listings using XML (Proposed ST.26). The time slots for a number of sessions have also been changed. The revised agenda is as follows:
The U.S. Patent and Trademark Office has announced a revised agenda for the next biotechnology/ chemical/pharmaceutical (BCP) customer partnership meeting to be held on June 6, 2012. New to the agenda is an afternoon session on the Recommendation for the Disclosure of Sequence Listings using XML (Proposed ST.26). The time slots for a number of sessions have also been changed. The revised agenda is as follows:














