By Kevin E. Noonan –
 Last week, the Council 4 Innovation Promotion (C4IP) put on a webinar entitled "Unpacking the Proposed TRIPS Waiver Expansion: A Conversation with Gary Locke." Gary Locke (at right) was the Secretary of Commerce during the Obama Administration and later Ambassador to China, and he was joined in conversation with David Kappos, Undersecretary of Commerce and Director of the U.S. Patent and Trademark Office while Secretary Locke served as Secretary. The conversation was moderated by Andrei Iancu, himself a former Undersecretary of Commerce and Director of the U.S. Patent and Trademark Office during the Trump administration.*
Last week, the Council 4 Innovation Promotion (C4IP) put on a webinar entitled "Unpacking the Proposed TRIPS Waiver Expansion: A Conversation with Gary Locke." Gary Locke (at right) was the Secretary of Commerce during the Obama Administration and later Ambassador to China, and he was joined in conversation with David Kappos, Undersecretary of Commerce and Director of the U.S. Patent and Trademark Office while Secretary Locke served as Secretary. The conversation was moderated by Andrei Iancu, himself a former Undersecretary of Commerce and Director of the U.S. Patent and Trademark Office during the Trump administration.*
The conversation focused on the proposed expansion of the TRIPS waiver adopted as a reaction to the COVID-19 pandemic (see "The Proposed WTO IP Waiver: Just What Good Can It Do? — An Analysis"). That waiver, "limited" to vaccines against the SARS-CoV-2 virus, itself was fraught with risk that the scope of the waiver could be expanded beyond patents (see "If the Devil of the WTO IP Waiver Is in the Details, What Are the Details?"). But the new proposal expressly extends into therapeutic drugs and diagnostic assays relevant to the pandemic and accordingly poses a much greater risk to the international innovation regime.
Director Kappos began the discussion after an introduction by Director Iancu, with Ambassador Locke speaking about his involvement in the waiver issue. He noted the bipartisan opposition to the TRIPS waiver and that even the original waiver had not achieved its goal of getting vaccines to less developed countries that needed them. This was because he explained the problem was not intellectual property but rather was a confluence of logistics and the need for example for the vaccines to be refrigerated. The risk, the Ambassador stated, was that actions like this are precedent-making without the justification of improving global health or response to the pandemic. In addition, even in instances where vaccines supplies were available many of the countries used to illustrate the need for the waiver did not accept them, for historical and cultural reasons for example (Director citing the statistic of 100 million discarded doses). The waiver was particularly troubling in view of the need for patent protection to provide necessary incentives to address unmet and unsolved disease treatment needs. Ambassador Locke illustrated these concerns as follows:
The evidence is overwhelming that IP protections have not impeded people's access to treatment and did not in any way impede the development of vaccines. Drug companies have been voluntarily licensing their technology to accelerate global manufacturing and distribution of treatments. . . . Pfizer and Merck have signed hundreds of agreements to allow generic versions of their medicines to be manufactured all around the world, they've worked with the UN Medicines and Patent Pool to allow generics to be manufactured by approx. 200 companies worldwide and many other companies have entered into voluntary licensing agreements . . . . India actually threw out 100 million doses of vaccines that expired . . . [and] South Africa is trying to cancel the contract [with Johnson & Johnson] for over 10 million doses of the vaccine. And so not a single country took advantage of the waiver for IP protections . . . to increase the supply of vaccines and [that's because] so many [generic] companies from around the world . . . had agreements with companies like Moderna and Pfizer to produce generic versions, but the problem in many of these developing countries was they couldn't refrigerate it . . . [or ] had distribution problems. . . . There is no evidence that there is a problem with getting access to the medicines to treat people who come down with COVID. So, I fear we're creating a precedent that will eventually weaken the U.S. innovation economy, which is so important if we want to look at developing cures for some of the most dreaded diseases out there.
Ambassador Locke also noted the impetus for some countries in favor of waivers as being a desire to get access to the "secret sauce" of these technologies to obtain economic and competitive benefits. Director Kappos mentioned the consequence of the waiver as being a "free giveaway" of the mRNA vaccine platform, and how this was particularly troubling because as a platform it implicates more than just SARS-CoV-2 and COVID-19. Also discussed was the support from the Biden Administration (at least for the initial waiver) as showing evidence of a disconnect between treatment of medicines and biopharmaceuticals when compared with other Administration initiatives like the CHIPS Act and imposing restrictions on hi-tech materials to China and other countries.
On the bright side, the Ambassador and Director discussed rising opposition ("starting to ask the tough questions," according to Director Kappos) to the new waiver from Mexico, Switzerland, the UK, the EU, Japan, South Korea, and Singapore, but (regrettably) not the U.S., and that U.S. labor unions (a source of support for the Administration) oppose the waiver on economic grounds as a "jobs issue." More significantly noted was the December 17th target date for the proposal to be formally considered by the WTO is likely to be deferred. But despite these glimmers of silver lining both the Ambassador and the Director emphasized the continuing need to "get the message" to policymakers (both on the Hill and in the Administration) of the disincentives to innovation in medicine and the pharmaceutical industry that the waiver is certain to produce.
Director Iancu discussed with the speakers the policymaking and political realities of how a cabinet member like the Secretary of Commerce can influence the discussion in the Administration, supported by input from constituents like pharmaceutical companies and other innovation stakeholders. But "if people are silent," the Ambassador noted, then the Administration might conclude the waiver was not an issue with these constituencies. And Director Iancu noted that such waivers (although being prominent in the biopharma industry today) may be proposed for other technologies (such as climate change), with results negative to innovation in these technologies.
Director Kappos also mentioned that WTO is "wasting its time" on the waiver in view of more pressing international trade issues. Director Iancu stated that the U.S. has a leadership role to play in persuading other countries about the real IP issues in view of international pressure on WTO and World Health Organization generated by the pandemic. Ambassador Locke raised the issue of future pandemics and the need to build and maintain infrastructure to be ready for them. Director Kappos noted that IP was particularly important for biopharma innovation because they were "risky, failure-prone" technologies. And Director Iancu noted the need to achieve the long-term goal of building local capabilities, and that these will not develop unless there is adequate patent and other IP protection.
Ambassador Locke noted in conclusion that there needs to be a climate that enables R&D to address these challenges and that strong industry is required to achieve these goals, both in the U.S. and in countries like China (which he says recognizes these needs). There is no basis or reason for waiving IP rights and protections ("we cannot just give [our technology] away"), he said in bringing the discussion to a close.
It is impossible not to have a feeling of déjà vu listening to these erudite and experienced speakers discuss these issues. They are reminiscent of the arguments involved in gene patenting and more currently arguments about drug costs and the relevance of patents (actually almost none, in the popular mind almost entirely). The 2018 volcano on the Big Island and public perception of it provides a vivid analogy:
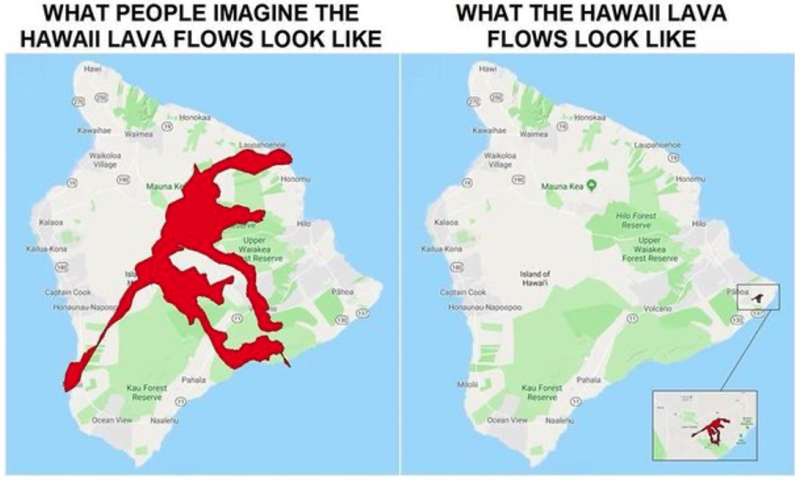 Which is not to say that the only rational response is despair at the ability for those like Ambassador Locke and Directors Kappos and Iancu to cut through the misperceptions and misinformation now rampant to explain the objective reality and the consequences of proposals like the extension of the TRIPS waiver. It is to say however than we must recognize how difficult the task may be, and also directly proportional to its importance.
Which is not to say that the only rational response is despair at the ability for those like Ambassador Locke and Directors Kappos and Iancu to cut through the misperceptions and misinformation now rampant to explain the objective reality and the consequences of proposals like the extension of the TRIPS waiver. It is to say however than we must recognize how difficult the task may be, and also directly proportional to its importance.
*Aphorisms about broken clocks and blind squirrels can be applied according to the reader's political proclivities.