By Kevin E. Noonan —
 The Federal Circuit used its decision that the District Court erred in certain of its claim construction determinations to reverse a jury award of greater than $100 million, but left intact large portions of the District Court's claim construction and remanded for a retrial consisting mainly of reconsideration of the proper scope of damages.
The Federal Circuit used its decision that the District Court erred in certain of its claim construction determinations to reverse a jury award of greater than $100 million, but left intact large portions of the District Court's claim construction and remanded for a retrial consisting mainly of reconsideration of the proper scope of damages.
The lawsuit involved U.S. Patent No. 5,727,554, which claimed an apparatus used to reduce damage to normal tissue during radiation therapy by producing a synchronization of radiation administration with patient movement such as breathing. The patent specification discloses the use of both natural (e.g., moles) or artificial (reflective surfaces) "fiducial" markings on patient skin used by the claimed apparatus to detect movement. Defendant Varian produces radiation equipment, including embodiments thereof comprising the accused infringing device, the "Real-Time Position Management ("RPM") Respiratory Gating System ("RPM System")" as an add-on option to their radiation equipment. The infringing device uses infrared camera to detect patient movement associated with reflective markers and synchronizes radiation treatment.
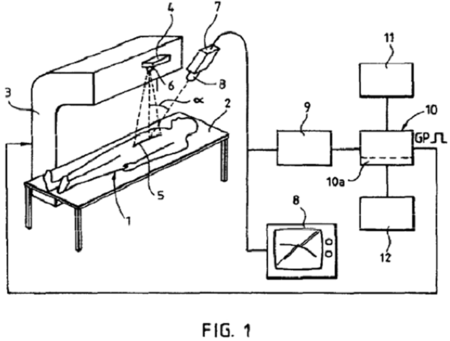
An earlier case between these parties was dismissed for lack of standing and appealed while this case was brought; during appeal of the first case (in which the Court reversed the decision to dismiss) Varian instituted an ex parte reexamination limited to claims 20-22 of the '554 patent; during the reexamination, Pittsburgh added claims 23-38. The ex parte reexamination was ultimately decided in favor of Pittsburgh.
Claims 20 and 22 are representative of the claims at issue:
20. Apparatus responsive to movement of a patient positioned on a patient positioning assembly, said apparatus comprising:
camera means generating digital image signals representative of an image of said patient; and
processing means comprising means determining movement of said patient from said digital image signals, including movement associated with breathing by said patient, and gating means generating gating signals synchronized with said movement associated with breathing by said patient.
22. The apparatus of claim 20 adapted for use during treatment of said patient with a radiation beam generated by a beam generator, wherein said gating means comprises means generating said gating signals synchronized to actuate said beam generator in synchronism with patient breathing.
The District Court determined that the radiation-generating portion of a claimed machine should be part of the infringing article (a decision having importance to the extent of the damages assessed). The parties stipulated that the phrase "means determining movement of said patient" is a means-plus- function term, but the parties differed on whether the disclosed structure corresponding to the means was "a computer processor programmed to perform a two-step algorithm[,] as Pitt contended, or a thirty-step algorithm, as Varian contended." The court-appointed special master weighed in at trial, recommending that the correct structure was "[a] computer processor programmed as a patient motion detector that (1) identifies at least one fiducial from the image signals; and (2) tracks its movement; and equivalents" thereof.
Claim 38 was added during the reexamination:
38. The apparatus of claim 20, further comprising a beam generator configured to provide computer-controlled multi-beam conformal dynamic radio therapy for said patient, wherein said gating signals are synchronized to actuate said beam generator in synchronism with patient breathing.
According to the Special Master, this claim encompassed:
[A] beam generator configured to provide radiation therapy under computer control. The radiation therapy treatment involves using radiation beams corresponding to the shape of the target. The beam generator is configured to be repeatedly re-positioned to irradiate the target with multiple treatment beams, each from a different direction.
The District Court granted summary judgment to Pittsburgh on infringement of claims 20, 21, 25, 26 and 36 by Varian devices comprising the RPM System itself; infringement of claims 22 and 38 by embodiments comprising radiation beam generator; and that Varian's actions satisfied the objective prong of the Court's willfulness assessment.
The District Court presided over a trifurcated trial on willfulness (the jury finding subjective willfulness, damages (awarding royalties for past damages), and invalidity. The total award entered by the Court was greater that $100 million.
The Federal Circuit, in an opinion by Judge O'Malley joined by Judge Lourie, affirmed in part (claim construction of claim 20), reversed in part (claim construction of claim 22 and the Court's determination that infringement was willful) vacated the overall damages award but affirmed the damages assessed for infringement of claim 38, and remanded to the trial court. Judge Dyk dissented.
The panel majority's reversal of the willfulness award was based on its determination that Varian's invalidity positions, while not sufficient to satisfy the clear and convincing evidence requirement, were not objectively baseless, and its vacature of damages with regard to infringement of claim 22 was because the Court found that damages should only be assessed on the RPM system itself. The Court construed claim 20 to recite means supported in the specification by disclosure of a two-step algorithm, the two steps comprising "first identifying fiducials and then tracking those fiducials." The panel rejected Varian's reliance on drawings in support of its claim construction position as being "merely implementations of the algorithm" that can be applied as needed. According to the opinion:
The district court properly located the disclosure of an algorithm that covered what was necessary to perform the claimed function of detecting patient movement and nothing more. Varian's attempt to pick and choose which steps it deems necessary by synthesizing steps from disparate portions of the written description is too limiting. The algorithm need only include what is necessary to perform the claimed function.
Under this construction the Court also rejected Varian's non-infringement arguments regarding claim 20: Varian argued that its RPM System only tracked the fiducials, not patient movement itself and thus did not meet the claim limitation of "determin[ing] movement of the patient from the digital image signals of the patient." The panel disagreed, using Varian's own argument:
Varian's infringement argument is belied by its own admission. Varian admits that the RPM System camera captures patient images to identify the location of the fiducials and only then eliminates the image of the patient. In other words, the RPM System uses the image of the patient and then processes that image to identify and extract from it the portions representing the fiducials.
The opinion also noted that the District Court had found that the phrase "image of said patient" in claim 20 encompassed "both images of fiducials and images of the patient image."
The Federal Circuit panel reversed the jury's willfulness determination because Varian's invalidity arguments not objectively baseless — the panel found that the cited prior art patent related to solving same problem used a "mark" such as a laser line as the reference, projected on patient and "continually tracked" during radiation treatment. In this regard, the panel noted that the PTO had initially rejected the claims over this reference during reexamination based on this reference. The opinion notes that this rejection is evidence that the assertion of an invalidity defense, and infringement based on the likelihood that the '554 patent claims were invalid in view of the reference was not objectively unreasonable. The Court also found that the District Court did not provide sufficient explanation for its contrary conclusion in view of the expert evidence presented at trial.
The Court's assessment of the damages award contained a determination that the District Court had erred in construing some of the claims as having a scope that encompassed some of Varian's accused infringing devices. Specifically, the panel reviewed the basis for the damages award, which were a 10.5% royalty on RPM System sales (for infringement of claim 20) and a 1.5% award on integrated systems comprising the RPM System and the radiation beam device (for infringement of claims 22 and 38). Varian did not specifically object to the damages award relating to the RPM System itself, except to contend that it does not infringe. On the other hand, Varian did ask the Court to vacate the damages award for infringement of claims 22 and 38, for three reasons. First, Varian argued that damages for the period during which the '554 patent was undergoing reexamination were improper because the limitation relating to inclusion of the radiation source were not in the claim as presented in the reexamination. Second, Varian argued that the District Court improperly calculated those damages. And third, Varian argued (briefly according to the panel) that the award violates 35 U.S.C. § 284.
With regard to claim 22, the panel agreed with Varian that the claim term "adapted for use during treatment of said patient with a radiation beam generated by a beam generator" did not include the beam generator within the scope of the claim and, thus, it was error for the jury to find infringement and award damages for sales of Varian's devices comprising the combination of a radiation beam generator and the RPM System. Claim 38, on the other hand, did include the beam generator as an affirmative limitation. In this instance, the Court rejected Varian's argument that the policy concerns enunciated in Garretson v. Clark, 111 U.S. 120 (1884), as relied upon under Lucent Techs., Inc. v. Gateway, Inc., 580 F.3d 1301, 1337 (Fed. Cir. 2009), required that the entire market value rule ("EMVR") be applied to the damages calculation. The Court based this decision on the inclusion of the beam generator as a claimed component within the scope of the claim. "As such," according to the Court, "Varian's argument fails because Pitt is not attempting to include the value of unpatented features within its royalty base. The beam generator is incorporated into the linear accelerator in claim 38; it is the combination apparatus that is claimed." In this regard, the Court discussed the proper application of the factors enunciated in Georgia–Pacific Corp. v. U.S. Plywood Corp., 318 F. Supp. 1116, 1120 (S.D.N.Y. 1970), to distinguish between a patented invention that merely adds "incremental value to the conventional element(s)" and an invention that "adds significant value to conventional elements." At trial, the jury heard expert testimony and received evidence from Varian itself that supported the value of the combination of Varian's beam generator and the RPM System as recited in claim 38. As a result, the Court determined that:
Put simply, the evidence the jury heard regarding the Georgia-Pacific factors and how those factors might impact any value added by the RPM System to the combination devices claimed in claim 38, the evidence and argument it heard regarding the need to guard against application of an unduly high royalty rate on sales of linear accelerators so as to not award Pitt for what it did not invent, and the fact that the jury was instructed on all the Georgia-Pacific factors and was instructed that it was free to award a zero percent royalty on the linear accelerators, were sufficient to guard against any unduly excessive damages award in this case. On this record, the Georgia-Pacific factors were sufficient to safeguard against over compensation for the infringing combination sales at issue.
After distinguishing various pre-Georgia Pacific decisions from various Circuit Courts of Appeal, the opinion "emphasize[d] the fact- and record-specific nature of [the] holding," particularly the extent to which Varian's own arguments and reliance of certain Georgia Pacific factors rendered inconsistent their arguments on appeal that were not persuasive and the Court affirmed the damages awarded by the jury in recompense of Varian's infringement of claim 38.
Judge Dyk dissented, based on his differing opinion on the proper construction of claim 20 and 22 and 38 depending therefrom. In Judge Dyk's view, claim 20, being a means-plus-function claim, must be supported by express disclosure in the specification of structural embodiments of the recited means. Here, because Judge Dyk appreciates that the recited function is performed by a computer, in his view the only proper support for a computer means requires disclosure of the computer program that executes the means. Judge Dyk believes that the computer means disclosed in the specification does not perform all the steps necessary to practice the claimed method, and thus that the claims as construed by the majority would be invalid for failure to be supported by a specification that satisfies the written description requirement of 35 U.S.C. § 112(a). For Judge Dyk, this indicates that there was an error in claim construction below and he would remand for reconsideration of infringement under the correct claim construction.
University of Pittsburgh v. Varian Medical Systems, Inc. (Fed. Cir. 2014)
Nonprecedential disposition
Panel: Circuit Judges Lourie, Dyk, and O'Malley
Opinion by Circuit Judge O'Malley; dissenting opinion by Circuit Judge Dyk
 About Court Report: Each week we will report briefly on recently filed biotech and pharma cases.
About Court Report: Each week we will report briefly on recently filed biotech and pharma cases.