By Donald Zuhn —
 Last Friday, the U.S. Patent and Trademark Office held a four-hour long forum to receive public feedback on the Myriad-Mayo Guidance, which was issued by the Office on March 4. According to the Office's Guidance webpage, the forum was intended to provide an opportunity for stakeholders to present their interpretation of the impact of Supreme Court precedent on the complex legal and technical issues involved in subject matter eligibility analyses during patent examination.
Last Friday, the U.S. Patent and Trademark Office held a four-hour long forum to receive public feedback on the Myriad-Mayo Guidance, which was issued by the Office on March 4. According to the Office's Guidance webpage, the forum was intended to provide an opportunity for stakeholders to present their interpretation of the impact of Supreme Court precedent on the complex legal and technical issues involved in subject matter eligibility analyses during patent examination.
On Monday, we reported on the opening remarks provided by Drew Hirshfeld, Deputy Commissioner for Patent Examination Policy, and the overview of the Guidance provided by Raul Tamayo, Senior Legal Advisor for the Office of Patent Legal Administration. Yesterday, we reported on the first of three groups of public presentations. Today, we report on the second group of public presentations. This group of presenters consisted of Courtenay C. Brinckerhoff of Foley & Lardner LLP; Dr. Leslie Fischer of Novartis Pharmaceuticals Corp.; and Dr. Warren D. Woessner of Schwegman Lundberg & Woessner, P.A.
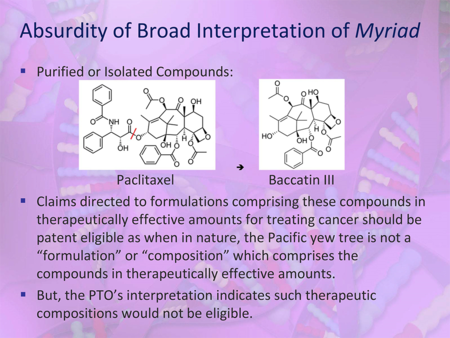
 Like prior presenters Dr. Sauer and Dr. Sonnenfeld, Ms. Brinckerhoff (at left) began her presentation by contending that the Guidance "[e]xtends Myriad to subject matter that was not before the Court and/or that the Court expressly stated that it was not addressing," including for example, compositions of matter, methods of treatment, and methods of manufacture. Noting that the Supreme Court in Myriad and Mayo "warned against overly-broad applications of the subject matter eligibility exceptions [because] too broad an interpretation of this exclusionary principle could eviscerate patent law," Ms. Brinckerhoff declared that the Guidance "threatens to do just that — eviscerate patent law and stifle investment and innovation." In support of her argument, she cited a Managing Intellectual Property article by Sherry Knowles that relied on a Journal of Natural Products paper by Dr. David J. Newman and Dr. Gordon M. Cragg entitled "Natural Products as Sources of New Drugs over the 30 Years from 1981 to 2010," (see "Sherry Knowles 'Speaks Truth to Power' on the PTO's § 101 Guidelines"). As shown in a slide from Ms. Brinckerhoff's presentation, 71% of the 1,355 drugs approved between 1981 and 2010 would fall outside the scope of the Guidance.
Like prior presenters Dr. Sauer and Dr. Sonnenfeld, Ms. Brinckerhoff (at left) began her presentation by contending that the Guidance "[e]xtends Myriad to subject matter that was not before the Court and/or that the Court expressly stated that it was not addressing," including for example, compositions of matter, methods of treatment, and methods of manufacture. Noting that the Supreme Court in Myriad and Mayo "warned against overly-broad applications of the subject matter eligibility exceptions [because] too broad an interpretation of this exclusionary principle could eviscerate patent law," Ms. Brinckerhoff declared that the Guidance "threatens to do just that — eviscerate patent law and stifle investment and innovation." In support of her argument, she cited a Managing Intellectual Property article by Sherry Knowles that relied on a Journal of Natural Products paper by Dr. David J. Newman and Dr. Gordon M. Cragg entitled "Natural Products as Sources of New Drugs over the 30 Years from 1981 to 2010," (see "Sherry Knowles 'Speaks Truth to Power' on the PTO's § 101 Guidelines"). As shown in a slide from Ms. Brinckerhoff's presentation, 71% of the 1,355 drugs approved between 1981 and 2010 would fall outside the scope of the Guidance.

According to Ms. Brinckerhoff, the Office erred in two respects when crafting the Guidance. In particular, she argued that the Office relied on an overly narrow interpretation of Funk Bros. that resulted in an overly broad application of that case, and the Office parsed claim elements rather than considering claims as a whole. With respect to the Office's interpretation of Funk Bros., Ms. Brinckerhoff explained that the Guidance finds a composition comprising a combination of components to be non-eligible unless at least one of the components is not a natural product (as in the training slides example of pomelo juice) or unless the components have a specific physical interrelationship (as in the Guidance's firework example).
Pointing to a passage from Funk Bros. (see slide below), Ms. Brinckerhoff remarked that while she sees "mention after mention of function and utility" in the citation, the Office instead "sees that same passage as requiring a markedly different structure." Ms. Brinckerhoff also reminded the Office (as did a number of presenters) that Chakrabarty spoke of "markedly different characteristics," and not "markedly different structure."

Contending that the Supreme Court did not question the eligibility of pharmaceutical compositions in either Myriad or Mayo, Ms. Brinckerhoff suggested an alternative test for compositions based on a proper reading of Funk Bros. (as set forth in the following slide from her presentation).

Ms. Brinckerhoff next inquired as to why method of treatment claims were subject to eligibility scrutiny as neither Mayo nor Myriad undermined the eligibility of such claims, and she indicated that she didn't understand why these types of claims were being rejected by examiners. She concluded her presentation by noting that applicants had few options for dealing with the Guidance. She asked whether applicants would need to take every adverse case to the Patent Trial and Appeal Board, and because the appeal route could take 3-5 years, inquired as to whether the Office would offer an expedited route to get a PTAB decision.
A copy of Ms. Brinckerhoff's presentation can be found here.
 The second presenter, Dr. Leslie Fischer (at right), argued that the problem with the Guidance was that no consideration is given to the function of claimed subject matter. Instead, "subject matter comprising only natural products must have a marked structural difference from the corresponding natural product in order to be patent eligible" (emphasis in presentation). According to Dr. Fischer, the solution would be to use the following test for patent eligibility:
The second presenter, Dr. Leslie Fischer (at right), argued that the problem with the Guidance was that no consideration is given to the function of claimed subject matter. Instead, "subject matter comprising only natural products must have a marked structural difference from the corresponding natural product in order to be patent eligible" (emphasis in presentation). According to Dr. Fischer, the solution would be to use the following test for patent eligibility:
Whether the claimed subject matter, as a whole, has:
(1) a physical difference (e.g., structure, form, purity, etc.) relative to the natural product(s); and
(2) a different function or use relative to the natural product(s).
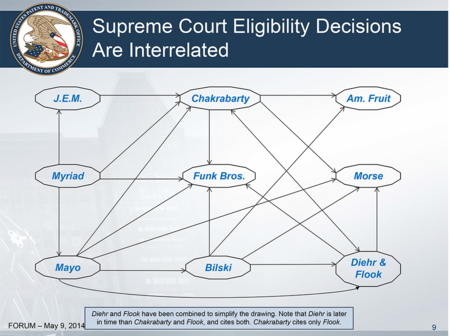
Dr. Fischer also noted that the Office's "web" of relevant cases (as shown in a slide from Mr. Tamayo's presentation) overlooked a few important decisions (which she indicated in her revised web). These additional cases "tell us that function is important."
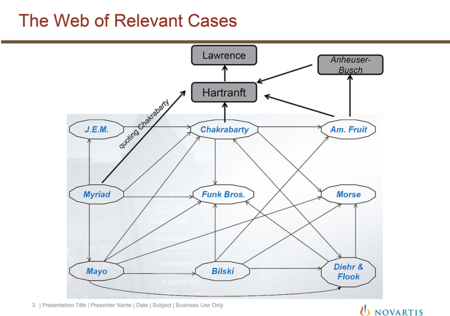
According to Dr. Fischer, the tariff cases Hartranft v. Wiegmann, 121 U.S. 609 (1877), and Lawrence v. Allen, 48 U.S. 785 (1849), indicate that a manufacture requires a new or distinctive form, character, name, or use. Noting that Chakrabarty cites Hartranft in finding that the claimed bacteria in that case constituted "a product of human ingenuity 'having a distinctive name, character [and] use,'" Dr. Fischer concluded that "[t]he consideration of functional changes as relevant to patent eligibility is in accordance with historical tar[]iff and patent cases, which consider whether products derived from nature have different functions and uses" (emphasis in presentation). Dr. Fischer also noted that the Myriad Court discussed Chakrabarty (and that case's citation to Hartranft) and Funk Bros. in similar terms, and concluded that "Myriad's use of Chakrabarty and Funk is perfectly compatible with considering whether a functional change can convert an otherwise 'natural product' into patent eligible subject matter." Thus, she declared that "Myriad does not tell us to throw away function."
Dr. Fischer further suggested that Myriad's claims were not directed to discrete chemical compositions, but rather were directed to information. She explained that the Myriad Court entwined DNA's functional aspect (i.e., information) with DNA's molecular structure (i.e., structure). In her view, "Myriad addresses a product, that, according to the Court, does not have a different function or structure from that which is found in nature." Dr. Fischer contended that the Myriad Court saw the characteristics of structure (sequence) and function (information) as inseparable, and argued that "Myriad has very limited applicability — [applying] only to subject matter in which molecular structure and function completely collapse, e.g., DNA." Dr. Fischer concluded her presentation by asking why the Office had decided to use the most damaging interpretation of Myriad, when it did not have to, and by reminding the Office that "for natural products, function matters."
A copy of Dr. Fischer's presentation can be found here.
 Noting that the Guidance "require[s] both that the claimed compound be 'non‐naturally occurring' and 'markedly different in structure from naturally occurring products' to be patent‐eligible," the final presenter in the second group, Dr. Woessner (at left), pointed out that "[n]either Myriad, Mayo nor Funk Bros. define 'natural product', much less a 'markedly structurally different, non‐naturally occurring product.'" Dr. Woessner next reminded forum attendees that while the Supreme Court vacated In re Bergy, 596 F.2d 952 (CCPA 1979) (finding pure culture of bacterium producing antibiotic not to be a natural product) when it took up Chakrabarty, Bergy has nevertheless been extensively cited as precedent ever since.
Noting that the Guidance "require[s] both that the claimed compound be 'non‐naturally occurring' and 'markedly different in structure from naturally occurring products' to be patent‐eligible," the final presenter in the second group, Dr. Woessner (at left), pointed out that "[n]either Myriad, Mayo nor Funk Bros. define 'natural product', much less a 'markedly structurally different, non‐naturally occurring product.'" Dr. Woessner next reminded forum attendees that while the Supreme Court vacated In re Bergy, 596 F.2d 952 (CCPA 1979) (finding pure culture of bacterium producing antibiotic not to be a natural product) when it took up Chakrabarty, Bergy has nevertheless been extensively cited as precedent ever since.

Noting that Chakrabarty contains no language disparaging Bergy or requiring structural alterations, Dr. Woessner contended that the claimed subject matter of Bergy would satisfy the test in Chakrabarty. Dr. Woessner concluded his presentation by declaring that neither Myriad nor Mayo provide a sufficient rationale for departing from about a century of practice that culminated in Bergy. According to Dr. Woessner, the Guidance has been "disruptive, regressive and should be withdrawn." He also reminded the Office that the first version of the written description guidelines were "taken back for a major overhaul after a public outcry just like this."
A copy of Dr. Woessner's presentation can be found here.
Before moving on to the third group of presentations, representatives from the Office fielded questions and received additional comments from attendees. One attendee remarked that the Office had taken "a step backwards" and "gone way overboard" with the Guidance, and warned that "you will have your head handed to you" once the courts had a chance to consider the Guidance. Mr. Hirshfeld noted that the Office was "exploring" the structural versus functional issue that had been raised by a number of presenters, but pointed out that the Office could not ignore Funk Bros., which he believed constrained what the Office could do. Nevertheless, he asked attendees if there was "a way between Funk and Chakrabarty"? June Cohan, a Legal Advisor with the USPTO's Office of Patent Legal Administration who presented on the Guidance at the biotechnology/chemical/pharmaceutical (BCP) customer partnership meeting and at the BIO's 2014 Spring Intellectual Property Counsels Committee (IPCC) Conference, stated that she was particularly concerned about discerning function that is inherent from function that was "added" to the claimed subject matter by the applicant.
In the final post in this series, we will examine the comments made by the third group of public presenters as well as the open participation/question and answer session that concluded the forum.
For additional information regarding this topic, please see:
• "USPTO Holds Forum on Subject Matter Eligibility — Part II," May 14, 2014
• "Guest Post: How to Patent Grapefruit Juice — The New USPTO Guidance for Patent Eligible Subject Matter Is Both Sticky and Sour," May 13, 2014
• "USPTO Holds Forum on Subject Matter Eligibility — Part I," May 12, 2014
• "USPTO Tries to Address Public Misunderstandings Regarding Myriad-Mayo Guidance," April 16, 2014
• "USPTO Issues Guidance for Analyzing Subject Matter Eligibility of Claims Reciting Laws of Nature/Natural Principles, Natural Phenomena or Natural Products," March 4, 2014