Absent Description of Representative Species to Support Entire Genus, Functionally Defined Genus Claim Lacked Adequate Written Description
By Donald Zuhn —
 Last week, in AbbVie Deutschland GmbH v. Janssen Biotech, Inc., the Federal Circuit affirmed judgments by the District Court for the District of Massachusetts in infringement and interference actions involving AbbVie Deutschland GmbH & Co., KG; AbbVie Bioresearch Center, Inc.; and AbbVie Biotechnology Ltd. ("AbbVie") and Janssen Biotech, Inc. and Centocor Biologics, LLC ("Centocor").
Last week, in AbbVie Deutschland GmbH v. Janssen Biotech, Inc., the Federal Circuit affirmed judgments by the District Court for the District of Massachusetts in infringement and interference actions involving AbbVie Deutschland GmbH & Co., KG; AbbVie Bioresearch Center, Inc.; and AbbVie Biotechnology Ltd. ("AbbVie") and Janssen Biotech, Inc. and Centocor Biologics, LLC ("Centocor").
In the infringement action, AbbVie had filed suit against Centocor in the District of Massachusetts for infringement of claims 29, 30, 32, and 64 of U.S. Patent 6,914,128 and claim 11 of U.S. Patent 7,504,485. Following a trial on validity, a jury determined that the asserted claims were invalid for lack of written description, lack of enablement, and for obviousness. The District Court denied AbbVie's post-trial motions for judgment as a matter of law and a new trial.
 In the inference action, Centocor had sought review under 35 U.S.C. § 146 of a determination by the Board of Patent Appeals and Interferences in an interference between Centocor's U.S. Patent Application 10/912,994 and AbbVie's '128 patent. The Board awarded priority to AbbVie and determined that the contested claims of the '128 patent were not invalid for obviousness.
In the inference action, Centocor had sought review under 35 U.S.C. § 146 of a determination by the Board of Patent Appeals and Interferences in an interference between Centocor's U.S. Patent Application 10/912,994 and AbbVie's '128 patent. The Board awarded priority to AbbVie and determined that the contested claims of the '128 patent were not invalid for obviousness.
In an opinion by Judge Lourie, joined by Judge Chen with Judge O'Malley concurring in the judgment, the Federal Circuit determined that the Board's decision in the interference proceeding lacked finality for purposes of collateral estoppel, the evidence supported the jury verdict that the claims lacked adequate written description, and there was no reversible error in the District Court's evidentiary rulings or jury instructions relating to written description that warranted a new trial. The Federal Circuit therefore affirmed the District Court's judgments of invalidity in both the infringement and interference actions.
The '128 and '485 patents are directed to fully human antibodies that bind to and neutralize the activity of human interleukin 12 (IL-12). The patents, which share the same written description, disclose the amino acid sequences of nearly 300 antibodies having a range of IL-12 binding affinities. The antibodies described in the patents were developed using phage display, with AbbVie initially identifying an antibody, Joe-9, that had low affinity for IL-12. To improve the IL-12 affinity of Joe-9, AbbVie introduced mutations into the CDR sequences of that antibody, identifying an improved antibody, Y61. Site directed mutagenesis was then employed to generate additional antibodies from Y61, with one antibody, J695, showing a significant increase in IL-12 binding and neutralizing activity. Because all of the antibodies described in the patents were derived from Joe-9, all of the antibodies have VH3 heavy chains and Lambda light chains like Joe-9. The antibodies described in the '128 and '485 patents also share at least 90% amino acid sequence similarity in their variable regions; with more than 200 of the described antibodies differing from Y61 at a single amino acid residue and sharing 99.5% similarity in the variable regions.
The claims of the '128 and '485 patents that were at issue in the appeals define the claimed antibodies by their function — i.e., IL-12 binding and neutralizing characteristics — rather than by structure. Representative claim 29 of the '128 patent recites:
29. A neutralizing isolated human antibody, or antigen-binding portion thereof that binds to human IL-12 and disassociates from human IL-12 with a koff rate constant of 1×10-2 s-1 or less, as determined by surface plasmon resonance.
Centocor markets a human IL-12 neutralizing antibody under the brand name Stelara®. Centocor's IL-12 antibody, which was developed using transgenic mice technology, has VH5 type heavy chains and Kappa type light chains, and shares about 50% sequence similarity in the variable regions as compared to Joe-9.
After filing the '994 application, which is directed to IL-12 antibodies, Centocor provoked an interference with AbbVie's '128 patent. During the interference, Centocor filed motions on the issues of obviousness and priority. Following the Board's decision to award priority to AbbVie and finding the '128 patent not invalid for obviousness, AbbVie filed suit against Centocor for infringement of the '128 and '458 patents, and Centocor filed two actions in the District Court for the District of Colombia seeking judicial review of the Board's interference decisions under § 146, as well as a declaratory judgment of noninfringement and invalidity of the '128 and '485 patents. Those actions were transferred to Massachusetts and consolidated with AbbVie's infringement action.
At trial, the District Court denied AbbVie's motion of summary judgment that Centocor was collaterally estopped from challenging the validity of the '128 patent because it had failed to invalidate the patent during the interference proceeding. After construing the claims, the District Court entered summary judgment that Centocor had infringed the asserted claims of the '128 and '458 patents, and the issue of validity was tried before a jury. Centocor challenged the validity of the '128 and '458 patents on the basis of written description, enablement, obviousness, and anticipation, arguing that the antibodies described in the patents were not representative of other members of the functionally claimed genus, which included Centocor's antibody. The differences between AbbVie's Joe-9 and J685 antibodies and Centocor's Sterlara-brand antibody were presented in Federal Circuit opinion as follows:
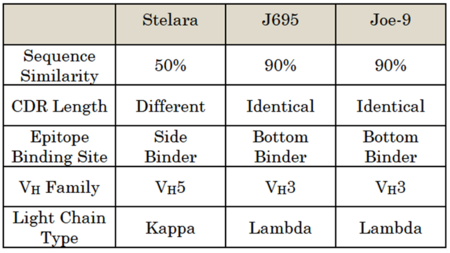
The jury determined that the asserted claims of the '128 and '458 patents were invalid for lack of adequate written description, lack of enablement, and obviousness. AbbVie then moved for JMOL on the grounds on which it lost, or for a new trial alleging that the District Court erred with respect to its evidentiary rulings and jury instructions. The District Court denied both motions and entered final judgment, and AbbVie appealed. On appeal, AbbVie challenged the District Court's denial of (1) AbbVie's motion for summary judgment that Centocor was collaterally estopped from raising invalidity defenses in the infringement action after the interference proceeding, (2) AbbVie's motion for JMOL on the issues of written description and enablement, and (3) AbbVie's motion for a new trial based on alleged errors in the District Court's evidentiary rulings and jury instructions.
With respect to the collateral estoppel issue, the Federal Circuit determined that Centocor was not estopped from raising invalidity defenses in the infringement action because the Board's priority and nonobviousness decisions lacked finality for purposes of collateral estoppel. The Court explained that because "a party seeking review of a decision of the Board under § 146 may 'shor[e] up evidentiary gaps' in the agency record by presenting live testimony, which could not be presented to the PTO," Board decisions reviewed under § 146 are not binding final judgments precluding a losing party from litigating the same or related issues in a parallel proceeding. In particular:
[Centocor was] entitled to present new evidence at least with respect to the issues of priority and obviousness in the underlying interference action. The factual record in the interference action was therefore open as to those issues, subject to a de novo determination by the district court. Consequently, the Board's priority and nonobviousness decisions lacked the requisite finality for purposes of collateral estoppel.
With respect to AbbVie's motion for a new trial, the Federal Circuit determined that the District Court did not abuse its discretion in excluding additional testimony from AbbVie's expert regarding the USPTO's reasoning for concluding that the '128 and '458 patents satisfied the written description requirement. The Court noted that "[i]n view of the record as a whole, including the substantial evidence of structural differences between Stelara and the Joe-9 antibodies and the fact that the jury considered the number of structurally similar antibodies disclosed in AbbVie's patents," the District Court did not abuse its discretion in excluding the additional testimony of AbbVie's expert. As for the District Court's jury instructions, the opinion refers to AbbVie's argument that the instructions were prejudicial "as a last ditch argument," concluding that "[t]aken as a whole, the jury instruction reasonably apprised the jury on weighing evidence relevant to the written description issue."
Finally, with respect to AbbVie's motion for JMOL on the issues of written description and enablement, the Federal Circuit determined that substantial evidence supported the jury's verdict of invalidity for lack of adequate written description of the claimed genus and affirmed the District Court's denial of JMOL on that issue. The opinion began its analysis of the issue by noting that "the requirement for an adequate written description serves a different purpose from that of the claims," citing Ariad Pharm., Inc. v. Eli Lilly & Co., 598 F.3d 1336, 1347 (Fed. Cir. 2010), for the proposition that "[c]laims define and circumscribe, the written description discloses and teaches." The Court explains that:
When a patent claims a genus using functional language to define a desired result, "the specification must demonstrate that the applicant has made a generic invention that achieves the claimed result and do so by showing that the applicant has invented species sufficient to support a claim to the functionally-defined genus." We have held that "a sufficient description of a genus . . . requires the disclosure of either a representative number of species falling within the scope of the genus or structural features common to the members of the genus so that one of skill in the art can 'visualize or recognize' the members of the genus" [citations omitted].
According to the opinion, in the instant appeals "[t]he question . . . is whether the ['128 and '458] patents sufficiently otherwise describe representative species to support the entire genus." In answering this question, the opinion analogizes the claimed genus to a plot of land, stating that:
[I]f the disclosed species only abide in a corner of the genus, one has not described the genus sufficiently to show that the inventor invented, or had possession of, the genus. He only described a portion of it. That is the case here.
While cautioning against "tak[ing] the analogy of a plot of land too far in thinking of written description issues because, even if one builds a house only in one corner of the plot, one may still own the whole plot," the opinion continues by explaining that:
One describes a plot of land by its furthest coordinates, in effect drawing a perimeter fence around it. That may be akin to the function of patent claims to particularly point out and distinctly circumscribe the outer boundaries of a claimed invention. With the written description of a genus, however, merely drawing a fence around a perceived genus is not a description of the genus.
Returning to the appeals at hand (and IL-12 antibodies), the opinion states that "[h]ere, the jury heard ample evidence that AbbVie's patents only describe one type of structurally similar antibodies and that those antibodies are not representative of the full variety or scope of the genus," noting that all of the antibodies described in the patents were derived from Joe-9, have VH3 heavy chains and Lambda light chains like Joe-9, and share at least 90% amino acid sequence similarity in their variable regions, and that more than 200 of the antibodies differ from Y61 at a single amino acid residue. In contrast, Centocor's antibody, which is encompassed by the scope of the claimed genus, has VH5 type heavy chains and Kappa type light chains, and shares about 50% sequence similarity in the variable regions as compared to Joe-9.
While acknowledging that "[i]t is true that AbbVie's patents need not describe the allegedly infringing Stelara in exact terms," the opinion states that "the patents must at least describe some species representative of antibodies that are structurally similar to Stelara." Suggesting that "[f]unctionally defined genus claims can be inherently vulnerable to invalidity challenge for lack of written description support, especially in technology fields that are highly unpredictable, where it is difficult to establish a correlation between structure and function for the whole genus or to predict what would be covered by the functionally claimed genus," the opinion also indicates that "[i]t is true that functionally defined claims can meet the written description requirement if a reasonable structure-function correlation is established, whether by the inventor as described in the specification or known in the art at the time of the filing date." However:
[T]he record here does not indicate such an established correlation. Instead, AbbVie used a trial and error approach to modify individual amino acids in order to improve the IL-12 binding affinity. Moreover, the '128 and '485 patents do not describe any common structural features of the claimed antibodies. The asserted claims attempt to claim every fully human IL-12 antibody that would achieve a desired result, i.e., high binding affinity and neutralizing activity, and cover an antibody as different as Stelara, whereas the patents do not describe representative examples to support the full scope of the claims.
Judge Lourie therefore concluded that substantial evidence supported the jury verdict of invalidity for lack of an adequate written description of the claimed genus, and affirmed the District Court's denial of JMOL on that issue.
In an opinion concurring in judgment, Judge O'Malley noted that she would have affirmed the District Court's judgment invalidating the asserted claims in the '128 and '485 patents for obviousness, pointing out that this was "a finding from which AbbVie does not appeal." Judge O'Malley explained that:
[B]ecause AbbVie did not appeal the district court's finding of obviousness, our decision on the jury instruction issue controls the outcome of this case. If we find no prejudicial error in the challenged jury instruction, the finding of obviousness stands and the patent is invalid. Alternatively, if we hold that the jury instruction is erroneous, AbbVie is entitled to a new trial on all validity issues.
With respect to the jury instruction, Judge O'Malley writes (quoting Judge Lourie) that "'[t]aken as a whole, the jury instruction reasonably apprised the jury on weighing evidence relevant to' both the written description and obviousness findings." As for Judge Lourie's discussion of the written description issue, Judge O'Malley "express[es] no opinion regarding the thoughtful written description analysis in the majority opinion," adding that "I simply do not think it necessary or dispositive to the outcome of this case."
In summary, the Federal Circuit found that Centocor was not collaterally estopped from raising invalidity defenses in the infringement action, that substantial evidence supported the jury verdict that the asserted claims were invalid for lack of an adequate written description, and that the District Court did not abuse its discretion in denying AbbVie's motion for a new trial with respect to the jury's written description verdict. The Federal Circuit therefore affirmed the District Court's judgments in both the infringement and interference actions.
AbbVie Deutschland GmbH v. Janssen Biotech, Inc. (Fed. Cir. 2014)
Panel: Circuit Judges Lourie, O'Malley, and Chen
Opinion by Circuit Judge Lourie; concurring opinion by Circuit Judge O'Malley
 In a post from yesterday entitled "Help Improve our AIA Trial Proceedings," Michelle Lee, Deputy Under Secretary of Commerce for Intellectual Property and Deputy Director of the U.S. Patent and Trademark Office, announced on the Director's Forum blog that the USPTO is seeking feedback on Patent Trial and Appeal Board (PTAB) trial proceedings established by the Leahy-Smith America Invents Act (AIA). This request follows on the heels of the AIA Trial Roundtables that were conducted earlier this year, as reported here. One of the stated goals of these roundtables was to obtain feedback on how the trial proceedings might be improved. Now, Ms. Lee explained, the USPTO would like to receive formal comments from the public on a variety of topics related to the PTAB, "including claim construction, amendment practice, and management of concurrent proceedings before the office." In addition to requesting constructive criticism, the USPTO would like to know what it is doing right. "We have a world class patent system," Ms. Lee explained, "and this is a golden opportunity to make it even better." The deadline for submission of comments is September 16, 2014, the two-year anniversary of the PTAB.
In a post from yesterday entitled "Help Improve our AIA Trial Proceedings," Michelle Lee, Deputy Under Secretary of Commerce for Intellectual Property and Deputy Director of the U.S. Patent and Trademark Office, announced on the Director's Forum blog that the USPTO is seeking feedback on Patent Trial and Appeal Board (PTAB) trial proceedings established by the Leahy-Smith America Invents Act (AIA). This request follows on the heels of the AIA Trial Roundtables that were conducted earlier this year, as reported here. One of the stated goals of these roundtables was to obtain feedback on how the trial proceedings might be improved. Now, Ms. Lee explained, the USPTO would like to receive formal comments from the public on a variety of topics related to the PTAB, "including claim construction, amendment practice, and management of concurrent proceedings before the office." In addition to requesting constructive criticism, the USPTO would like to know what it is doing right. "We have a world class patent system," Ms. Lee explained, "and this is a golden opportunity to make it even better." The deadline for submission of comments is September 16, 2014, the two-year anniversary of the PTAB.












