By Andrew Williams —
 Late last month, while many of us were getting ready to attend the BIO International Convention in San Diego, the Patent Trial and Appeal Board ("Board") issued four related inter partes review opinions, marking what many have called the first set of opinions related to the Pharmaceutical industry. These cases, which were issued on June 20, 2014, were IPR2013-00116, IPR2013-00117, IPR2013-00118, and IPR2013-00119, and the parties were Gnosis S.P.A., Gnosis Bioresearch S.A., and Gnosis U.S.A., Inc. ("Gnosis") (Petitioner) and either Merck & Cie ("Merck") (Patent Owner in IPR2013-00117) or South Alabama Medical Science Foundation ("SAMSF") (Patent Owner in the remaining three IPRs). This post will address the Final Written Decisions related to the SAMSF patents.
Late last month, while many of us were getting ready to attend the BIO International Convention in San Diego, the Patent Trial and Appeal Board ("Board") issued four related inter partes review opinions, marking what many have called the first set of opinions related to the Pharmaceutical industry. These cases, which were issued on June 20, 2014, were IPR2013-00116, IPR2013-00117, IPR2013-00118, and IPR2013-00119, and the parties were Gnosis S.P.A., Gnosis Bioresearch S.A., and Gnosis U.S.A., Inc. ("Gnosis") (Petitioner) and either Merck & Cie ("Merck") (Patent Owner in IPR2013-00117) or South Alabama Medical Science Foundation ("SAMSF") (Patent Owner in the remaining three IPRs). This post will address the Final Written Decisions related to the SAMSF patents.
 Even though these cases were heralded as the first Pharma IPR Final Written Decisions, it is important to note a couple of things. First, even though these parties were also involved in Federal court litigation, which has been stayed pending the outcome of these cases, the accused infringer/petitioner did not file an ANDA. In fact, these cases involved compositions of natural folate used in the treatment of vitamin deficiencies. As such, these compositions are not drugs that require approval by the Food and Drug Administration. In addition, these are not composition-of-matter patents, but rather patents with claims drawn to compositions or methods of treatment. Nevertheless, these cases did center on the particular chemistry involved, and the pharmaceutical community would be remiss to ignore these decisions and the reasoning espoused by the Board.
Even though these cases were heralded as the first Pharma IPR Final Written Decisions, it is important to note a couple of things. First, even though these parties were also involved in Federal court litigation, which has been stayed pending the outcome of these cases, the accused infringer/petitioner did not file an ANDA. In fact, these cases involved compositions of natural folate used in the treatment of vitamin deficiencies. As such, these compositions are not drugs that require approval by the Food and Drug Administration. In addition, these are not composition-of-matter patents, but rather patents with claims drawn to compositions or methods of treatment. Nevertheless, these cases did center on the particular chemistry involved, and the pharmaceutical community would be remiss to ignore these decisions and the reasoning espoused by the Board.
The Technology
The technology at issue in these IPRs generally related to compositions containing reduced folates and methods of using such compositions to overcome folate deficiency. As the Board's decisions explained, the background section of the SAMSF patents explained that folate deficiency has been linked to peripheral vascular disease, various birth defects, and other disorders. These disorders can result in abnormally high blood levels of homocysteine, which is normally converted to methionine in a folate-dependent step. To overcome these conditions, folic acid can be added to commercial preparations, sometimes in combination with other vitamins. The problem is that some individuals do not adsorb folic acid readily from the intestine after oral administration. As a solution, reduced folates can be used, which are found in nature as, for example, tetrahydrofolic acid ("THFA" or "THF"), 5-methyl-tetrahydrofolic acid ("5-methyl-THFA"), and 5-formyl-tetrahydrofolic acid ("5-formyl-THFA"). The stereoisomer of 5-methyl-THFA that has the L configuration at carbon-6 was shown in the SAMSF patents as:
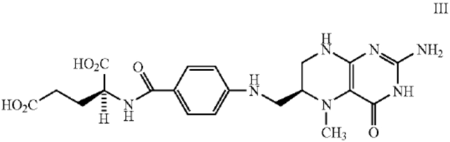
In contrast to these useful "6S isomers" found in nature, the "unnatural isomer component" can lead to adverse effects. Therefore, chirally-pure 5-formyl-(6S)-THFA has been commercially produced for use in disease therapy.
The Patents
The three patents involved in the IPRS of the three SAMSF patents were all related via continuation applications, and therefore share a specification. These patents described the use of natural isomers of reduced folates in dietary vitamin preparations, in particular 5-methyl-THFA. Claim 37 of U.S. Patent No. 5,997,915, subject of IPR2013-00116, is representative for those reviewed in that patent:
37. A method of increasing a human subject's dietary intake of folate comprising administering to the human subject a composition for human consumption comprising:
one or more natural isomers of reduced folate selected from the group consisting of (6S)-tetrahydrofolic acid, 5-methyl-(6S)-tetrahydrofolic acid, 5-formyl-(6S)-tetrahydrofolic acid, 10-formyl-(6R)-tetrahydrofolic acid, 5,10-methylene-(6R)-tetrahydrofolic acid, 5,10-methenyl-(6R)-tetrahydrofolic acid, 5-formimino-(6S)-tetrahydrofolic acid, and polyglutamyl derivatives thereof; and
a nutritional substance for human consumption being an essential nutrient preparation, the essential nutrient preparation comprising a vitamin other than ascorbic acid, wherein the vitamin is present in an amount equal to or greater than 25% of the daily requirement for the vitamin per customarily consumed quantity of said essential nutrient preparation.
All of the remaining claims under consideration required that the one or more natural isomers of reduced folate be "substantially chirally pure 5-methyl-(6S)-tetrahydrofolic acid or a polyglutamyl derivative thereof." After a Motion to Amend was granted, cancelling all but one claim of U.S. Patent No. 6,673,381 (subject of IPR2013-00118), the only remaining claim of that patent was claim 32, dependent from claim 22:
22. A method for treating and/or preventing vascular disease in a subject, said method comprising administering, to the subject, a composition which comprises:
one or more natural isomers of reduced folate selected from the group consisting of (6S)-tetrahydrofolic acid, 5-methyl-(6S)-tetrahydrofolic acid, 5-formyl-(6S)-tetrahydrofolic acid, 10-formyl-(6R)-tetrahydrofolic acid, 5,10-methylene-(6R)-tetrahydrofolic acid, 5,10-methenyl-(6R)-tetrahydrofolic acid, 5-formimino-(6S)-tetrahydrofolic acid, and polyglutamyl derivatives thereof; and
a nutritional substance selected from the group consisting of a food preparation, an essential nutrient preparation, and combinations thereof;
wherein, when the nutritional substance is a food preparation, the food preparation comprises two or more food components and each gram of said food preparation has a natural molar amount, N, of said one or more natural isomers of reduced folate, wherein N is greater or equal to zero and wherein each gram of said composition has a total molar amount, T, of said one or more natural isomers of reduced folate greater than N; wherein, when the nutritional substance is an essential nutrient preparation, the essential nutrient preparation comprises a vitamin other than ascorbic acid.
32. A method according to claim 22, wherein each of the one or more natural isomers of reduced folate is substantially chirally pure.
Similarly, after a Motion to Amend canceled all but one claim of U.S. Patent No. 7,172,778 (subject of IPR2013-0011), the only remaining claim of that patent was claims 15, dependent from claim 1:
1. A composition comprising:
one or more natural isomers of reduced folate in an amount effective for the treatment and/or prevention of vascular disease and/or in an amount effective for reducing a female's risk of having a miscarriage and/or of having a fetus with a neural tube defect, a cleft lip defect, and/or a cleft palate defect, wherein said one or more natural isomers of reduced folate is selected from the group consisting of (6S)-tetrahydrofolic acid, 5-methyl-(6S)-tetrahydrofolic acid, 5-formyl-(6S)-tetrahydrofolic acid, 10-formyl-(6R)-tetrahydrofolic acid, 5,10-methylene-(6R)-tetrahydrofolic acid, 5,10-methenyl-(6R)-tetrahydrofolic acid, 5-formimino-(6S)-tetrahydrofolic acid, and polyglutamyl derivatives thereof; and
a nutritional substance selected from the group consisting of a food preparation, an essential nutrient preparation, and combinations thereof;
wherein, when the nutritional substance is a food preparation, the food preparation comprises two or more food components and each gram of said food preparation has a natural molar amount, N, of said one or more natural isomers of reduced folate, wherein N is greater or equal to zero and wherein each gram of said composition has a total molar amount, T, of said one or more natural isomers of reduced folate greater than N; wherein, when the nutritional substance is an essential nutrient preparation, the essential nutrient preparation comprises a vitamin other than ascorbic acid.
15. A composition according to claim 1, wherein each of the one or more natural isomers of reduced folate is substantially chirally pure.
Obviousness
All of the reviewed claims of SAMSF's patents were invalidated as obvious in view of two or three cited references. The first of these references was Serfontein, European Patent Application EP 0 595 005 A1, which disclosed "a pharmaceutical preparation for lowering levels of homocysteine," which included "folate or a suitable active metabolite of folate or a substance which releases folate in vivo." The only difference between Serfontein and the claims at issue, according to the Board, was that the cited reference did not explain what "a suitable active metabolite of folate" was, nor did it provide any examples other than folate or folic acid. The second reference cited to overcome this deficiency was Marazza, U.S. Patent No. 5,194,611. Marazza was described as teaching methods for the chiral resolution of 5-methyl-THF into its (6R) and (6S) diastereomers. The final reference, Ueland (Ueland et al., Plasma Homocysteine, a Risk Factor for Vascular Disease: Plasma Levels in Health, Disease, and Drug Therapy, J. Lab. Clin. Med. 114:474-501 (1989)), was only cited against claim 15 of the '778 patent in combination with the other two references. Ueland is described as teaching homocysteine metabolism.
The sole argument made by the petitioner against all of the claims at issue was that Marazza identified chirally-pure 5-methyl-(6S)-THFA as being a naturally occurring metabolite of folate that one of skill in the art would have had reason to use as the "suitable active metabolite of folate" described in the Serfontein reference. Ueland was merely cited in the one case as identifying increased 5-methyl-THFA intracellular pools as the mechanism for reducing homocysteine. Instead of rejecting Gnosis's arguments for failing to establish why one skilled in the art would combine these two (or three) references, the Board invalidated the claims as obvious because SAMSF was unable to provide evidence why one skilled in the art would not find this combination obvious. In other words, not only was there no presumption of validity in this IPR proceedings (as expected), but the patent holder appeared to have the increased burden of proving the validity of its own patent.
SAMSF did make several arguments that the Board rejected. First, in order to explain why these references would not have been combined, SAMSF argued that one skilled in the art would not have been motivated to look to other forms of folate because of the widespread acceptance of folic acid to treat folate deficiencies. The Board responding by pointing out that a person of ordinary skill in the art would not "have avoided alternatives simply because a standard is known to be suitable and to work well." Second, SAMSF argued that folinic acid (5-formyl-THFA) was indicated for use in cancer treatment, no folic acid deficiency. In response, the Board pointed out that just because "folinic acid is not indicated for treating folate deficiency," it "does not follow . . . that folinic acid is not suitable for treating deficiency." Finally, SAMSF pointed out that the prior art taught that reduced folates were inferior to folic acid in several properties, such as bioavailability, substrate activity, disruption of folate metabolism, stability, and commercial availability. In all cases, the Board found SAMSF's arguments unpersuasive because SAMSF did not provide credible evidence why such inferior properties would have discouraged skilled artisans from using these reduced folates for dietary purposes.
SAMSF also provided evidence of objective indicia of non-obviousness, or so-called secondary considerations, based on the sales of five products: Néevo prescription prenatal vitamins, NéevoDHA® prescription prenatal vitamins, Mentanx® medical food, Cerefolin® medical food, and CerefolinNAC® medical food. The sale of these products demonstrated, according to SAMSF, commercial success, licensing, copying, long-felt but unmet need, discovering and solving an unrecognized problem, unexpected results, skepticism, and praise. The Board did not challenge these allegations, but instead found that there was no nexus between the evidence and the claimed invention. SAMSF's inventors had identified that chirally-pure 5-methyl-(6S)-THFA could be used in combination with other vitamins in dietary preparations to treat disorders related to folate deficiencies. It was the opinion of the Board that SAMSF did not identify the novel elements in the reviewed claims that would anchor them to the objective evidence. Instead, the claims were doomed because SAMSF apparently relied on the administration of 5-methyl-(6S)-THFA for its nexus, and this aspect, according to the Board, was taught by Marazza (even though Marazza did not anticipate the claims). The lesson to be learned in attempting to establish a nexus in an IPR proceeding is to not appear as though your argument rests on only part of a combination claim (even if the combination is novel).
In total, the Board found that all of the reviewed claims of these three patents invalid as obvious in view of the cited art. The fourth IPR, in which Merck was the patent holder, had a similar outcome with similar cited art. Nevertheless, because that patent was unrelated to SAMSF patents, there were some unique issues in that case that we will address at a later date. In addition, we will continue to monitor these pharma and biotech cases and provide periodic updates.

 62% of these universities are U.S. universities, with China (18%) being the foreign country with the largest number of patent-owning universities. U.S. universities having the largest number of patents are both public (the University of California system, Texas, Wisconsin, Michigan, and Illinois) and private (MIT, Stanford, Columbia and CalTech) and represent universities large and small. Undisclosed in this Report is any correlation between the number of patents and the amount of grant monies obtained by each of these universities; these results support the sentiment, voiced at the Tech Transfer Summit/North America earlier this week by many tech transfer officials that universities should concentrate on patenting fulfilling the statutory mandate to facilitate technology transfer and commercialization and less on university tech transfer offices being a profit center for their universities.
62% of these universities are U.S. universities, with China (18%) being the foreign country with the largest number of patent-owning universities. U.S. universities having the largest number of patents are both public (the University of California system, Texas, Wisconsin, Michigan, and Illinois) and private (MIT, Stanford, Columbia and CalTech) and represent universities large and small. Undisclosed in this Report is any correlation between the number of patents and the amount of grant monies obtained by each of these universities; these results support the sentiment, voiced at the Tech Transfer Summit/North America earlier this week by many tech transfer officials that universities should concentrate on patenting fulfilling the statutory mandate to facilitate technology transfer and commercialization and less on university tech transfer offices being a profit center for their universities.