By Kevin E. Noonan –
 On December 14th*, the Patent Trial and Appeal Board rendered its decision on Preliminary Motions in Interference No. 106,133 between Senior Party Sigma-Aldrich and Junior Party The Broad Institute, Harvard University, and MIT (collectively, "Broad").
On December 14th*, the Patent Trial and Appeal Board rendered its decision on Preliminary Motions in Interference No. 106,133 between Senior Party Sigma-Aldrich and Junior Party The Broad Institute, Harvard University, and MIT (collectively, "Broad").
To recap, the Board granted leave for Junior Party Broad to file its proposed Substantive Preliminary Motion No. 1 to change the Count under 37 C.F.R. §§ 41.121(a)(1)(i) and 41.208(a)(2). The justification here as in earlier interferences (Nos. 106,115 and 106,126) was to provide "a count broad enough to cover Broad's best proofs, which are directed to CRISPR-Cas9 systems using dual-molecule RNA but not including a donor polynucleotide sequence." Broad argued in the alternative a proposed Substitute Count 3:
A method comprising: introducing into, or expressing in, a eukaryotic cell having a DNA molecule,
(I) a Cas9 protein or one or more nucleotide sequences encoding the Cas9 protein;
(II) an RNA or one or more nucleotide sequences encoding the RNA, the RNA comprising: (a) a first RNA comprising a first ribonucleotide sequence and a second ribonucleotide sequence, and (b) a second RNA; and
(III) a template polynucleotide;
wherein the second RNA forms an RNA duplex with the second ribonucleotide sequence, and wherein, in the eukaryotic cell, the first ribonucleotide sequence directs the Cas9 protein to a target sequence of the DNA molecule, whereby the Cas9 cleaves both strands of the DNA molecule and the cleavage is repaired by integration of the template polynucleotide into the DNA molecule in the eukaryotic cell.
or, as in this interference as declared, claim 31 of U.S. Application No. 15/456,204; the Board notes that the first alternative portion of proposed Substitute Count 3 corresponds to Broad Application No. 16/177,403 claim 52 which has not been deemed allowable in ex parte examination.
The Board also granted Broad leave to file Contingent Preliminary Motion No. 2 to add claims in Broad applications not designated as corresponding to the Count as declared under 37 C.F.R. §§ 41.121(a)(1)(i) and 41.208(a)(2). Specifically, these included claims 1, 40, and 41 of Application No. 15/160,710; claims 74, 94, and 95 of U.S. Application No. 15/430,260; and claims 52-56 of U.S Application No. 16/177,403.
The Board also granted Broad leave to file Substantive Preliminary Motion No. 3 to designate various claims as not corresponding to Substitute Count 3 on a variety of grounds under 37 C.F.R. §§ 41.121(a)(1)(iii) and 41.208(a)(2).
The Board denied or deferred to the priority phase all other motions Broad requested.
For Senior Party Sigma-Aldrich, the Board granted leave to file Substantive Preliminary Motion No. 1 to challenge Broad's entitlement to be accorded benefit of its provisional applications for failure to provide a constructive reduction to practice of an embodiment falling within the scope of Count 1.
The Board also granted leave to file Substantive Preliminary Motion No. 2 to have the Board designate Broad's application No. 15/330,876 as not corresponding to the Count.
The Board denied or deferred to the priority phase all other motions requested by Sigma-Aldrich.
After accepting the Parties' expert witnesses and assertions regarding the level of ordinary skill in the art, the Board rendered its decisions first on Junior Party Broad's Preliminary Motions and thereafter on Senior Party Sigma-Aldrich's motions. No motions were granted; the Board denied Broad's Substantive Motions No. 1 and 3 and dismissed as moot Broad's Contingent Motion No. 2, denied Sigma-Aldrich's Substantive Motion No. 1. In addition, the Board dismissed as moot Sigma-Aldrich's Miscellaneous Motion No. 2 and Broad's Miscellaneous Motion No. 4 to exclude evidence as moot in view of its denial of the parties' substantive motions.
Regarding the Board's reasoning for denying Broad's Substantive Preliminary Motion No. 1, the Board agreed with Sigma-Aldrich that Broad failed to comply with the provisions of the Board's grant of leave to file the motion, and specifically that portion of the grant where the Board restricted the designation of one of Broad's claims as part of a McKelvey Count (a hybrid Count reciting in the alternative a representative claim of each party) to a claim that had been granted or allowed, which was not the case for the claim Broad designated (claim 52 of the Broad '403 application). The basis for this decision was that the Examiner in the '403 prosecution expressly stated on the record that "no agreement had been reached" as to the patentability of this claim at the time Broad filed its Motion, inter alia, because prosecution had been suspended (although the claim was deemed allowable on March 7, 2022). Broad filed its brief on December 3, 2021 and thus this claim had not been deemed allowable when the Motion as filed. In addition to this procedural deficiency, the Board agreed with Sigma-Aldrich that proposed Count 3 was excessively broad and introduced changes in Count 1 as declared "without any apparent reason." This flaw extended to the Count not being restricted to "Type II CRISPR-Cas9" (because this term was not recited) and recites "first RNA" and "second RNA" species rather than crRNA and tracrRNA as recited in Count 1 as declared. The Board further denies Broad's request to substitute because "Broad fails to present persuasive arguments that Count 1 is not limited to a single patentable invention" as well as Broad citing Sigma-Aldrich arguments made with regard to applications for which the Board states "it is not clear that these arguments are specifically relevant to the issues Broad raises in this motion." Finally, the Board denies Broad's Substantive Motion No. 1 because while Broad argues the Count as declared unfairly excludes Broad's best proofs because it includes a Broad claim that is not the broadest claim involved in the interference" Broad did not take advantage of the opportunity to propose a new count without these purported deficiencies by submitting a proposed Count No. 3 that did not correspond to a granted or at least allowed claim.
Regarding Broad's Contingent Preliminary Motion No. 2, the Board dismissed this claim as moot based on its denial of Broad's Substantive Preliminary Motion No. 1.
The Board considered Broad Substantive Preliminary Motion No. 3 in view of similar motions made in other interferences (for example Interference No. 106,115); there the Board held that disclosing two or more nucleus localization signal (NLS) sequenced rendered the claims as corresponding to the Count and on this basis denied Broad's motion to have claims designated as not corresponding to the Count as declared (despite Sigma-Aldrich providing no opposition to this Motion) based on issue preclusion. Specifically, the Board held that these requirements of A.B. Dick Co. v. Burroughs Corp., 713 F.2d 700, 702 (Fed. Cir. 1983) were satisfied:
(1) the issue is identical to one decided in the first action; (2) the issue was actually litigated in the first action; (3) resolution of the issue was essential to a final judgment in the first action; and (4) plaintiff had a full and fair opportunity to litigate the issue in the first action.
The fact that Broad ultimately prevailed in the priority phase of the '115 interference did not rebut the Board's conclusion, the opinion stating that "we see no reason why Broad should continue to get additional bites of the apple merely because it ultimately prevailed in an earlier proceeding" and finding the status of this decision on appeal was also insufficient unless the Federal Circuit produces a basis (i.e., reversal) to do so. And with regard to Broad's argument that claims reciting chimeric Cas9 proteins, including such proteins fused to heterologous protein domains should fall outside the scope of the Count the Board summarily held that they were unpersuaded (after setting forth Broad's arguments in detail and even Sigma's argument from Interference No 106,132 on this issue).
Turning to Sigma-Aldrich Substantive Preliminary Motion No. 1, the Board rejected Sigma's argument that Broad Provisional Application No. 61/736,527 (designated "P1" and filed December 12, 2012) did not provide a constructive reduction to practice (i.e., lacked adequate written description and enabling disclosure) for one embodiment falling within the scope of Count 1 as declared. Specifically, the Board disagreed with Sigma's assertion that a constructive reduction to practice required P1 to demonstrate differences in gene expression and not just double-stranded CRISPR cleavage of a target genomic DNA sequence recognized by the crRNA portion of the CRISPR-Cas9 complex introduced into a eukaryotic cell. This decision is based in part on the Board's consideration of expert testimony from each Party's expert as well as portions of the P1 specification identified by each Party in support of their arguments. And the Board rejects Sigma's argument that the term "altering" gene expression is limited to exclude cleaving a genomic DNA sequence per se and relies on Paragraph 38 of the P1 specification for a broader meaning of the phrase:
[T]extbook discussion of the basic cellular processes involved in gene expression (e.g., transcription, translation, and splicing) [and] is unenlightening here because the claim limitation is directed to the cell's "express[ion]" of the gene product, not the DNA molecule's "encod[ing]" process (i.e., transcription, translation, or splicing).
Based on this disclosure, the Board held that "the preponderance of the evidence supports a broader interpretation wherein the alteration of any aspect of gene expression meets the limitation in the Broad portion of Count 1 of "whereby express of the at least one gene product is altered."
The Board further cites Broad's assertion of disclosure of example of homology-directed recombinant to replace "a 2000-nucleotide section of the EMXI gene with a homologous template that includes a 12-base pair sequence changing the stop codon of the gene to sequences for two restriction enzymes (HindIII and NheI) and, thus, altering the expression of at least one gene product using a CRISPR-Cas9 system in a eukaryotic cell" in support of this construction (as illustrated by Figures 4C-4E of the P1 specification:
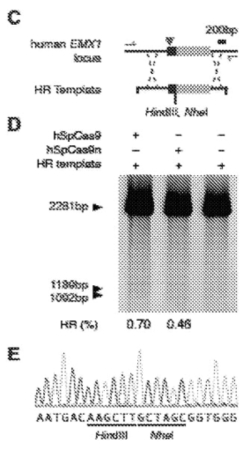 Illustrating how an expert witness can produce unpersuasive testimony, the Board states with regard to her support for Sigma's argument that this disclosure is insufficient to show alteration of gene expression that:
Illustrating how an expert witness can produce unpersuasive testimony, the Board states with regard to her support for Sigma's argument that this disclosure is insufficient to show alteration of gene expression that:
Neither Sigma nor Dr. Cannon explains why sequence data showing the absence of a stop codon would not have indicated to one of ordinary skill in the art that the resulting protein would contain additional amino acids.
and
Dr. Cannon does not testify that one of ordinary skill in the art would not have understood that the sequencing information in Figure 4E would produce a protein longer than the unaltered emx1 gene would produce.
On this argument, the Board was persuaded by Broad's expert that this disclosure satisfies the requirement that the CRISPR-Cas9 complex disclosed in Broad's P1 priority document demonstrates a system wherein expression of at least one gene product is altered" as required by Count 1.
Finally, the Board easily rejects Sigma's argument that the P1 disclosure of Cas9 embodiments comprising an unspecified number of NLS sequences does not provide constructive reduction to practice of embodiments comprising one NLS sequence, saying "Because the Broad portion of Count 1 includes an embodiment without a limitation on the number of NLS or even if an NLS is present on the Cas9 protein, we are not persuaded that even if Broad P1 does not describe a CRISPR-Cas9 system with a Cas9 protein having only one linked NLS, as Sigma argues."
On the basis of these Party arguments, the Board denied Sigma-Aldrich's Substantive Preliminary Motion No. 1.
And as with the priority phase in co-pending Interference No. 106,126, No. 106,127, and No. 106,132 (see "PTAB Suspends ToolGen Interferences" and "PTAB Redeclares Interference No. 106,132 and Suspends Priority Phase Proceedings"), the Board suspended the priority phase of this interference until the appeal in Interference No. 106,115 has been decided by the Federal Circuit.
*Our apologies for the delay in posting on these decisions.
 Fitch Even will be offering a webinar entitled "Full Disclosure About Non-Disclosure Agreements" on January 31, 2023 from 12:00 pm to 1:00 pm (ET). Steven G. Parmelee and Michael R. Anderson of Fitch Even will address the following topics:
Fitch Even will be offering a webinar entitled "Full Disclosure About Non-Disclosure Agreements" on January 31, 2023 from 12:00 pm to 1:00 pm (ET). Steven G. Parmelee and Michael R. Anderson of Fitch Even will address the following topics:







