By Kevin E. Noonan –
 In multiple ANDA litigations against multiple defendants, Millennium Pharmaceuticals, Inc. had several of its asserted claims held invalid for obviousness at the district court. The Federal Circuit reversed these decisions earlier this week in an opinion styled Millennium Pharmaceuticals, Inc. v. Sandoz Inc. The decision is consistent with recent trends at the Court, reviewing both district court obviousness determinations as well as those by the PTAB at the USPTO (see "Securus Technologies, Inc. v. Global Tel*Link Corp. (Fed. Cir. 2017)"; "In re Nuvasive (Fed. Cir. 2016)"), putting a greater emphasis on fact findings consistent with the ultimate legal question of obviousness in addition to fully satisfying the legal requirements of Section 103 of the Patent Statute.
In multiple ANDA litigations against multiple defendants, Millennium Pharmaceuticals, Inc. had several of its asserted claims held invalid for obviousness at the district court. The Federal Circuit reversed these decisions earlier this week in an opinion styled Millennium Pharmaceuticals, Inc. v. Sandoz Inc. The decision is consistent with recent trends at the Court, reviewing both district court obviousness determinations as well as those by the PTAB at the USPTO (see "Securus Technologies, Inc. v. Global Tel*Link Corp. (Fed. Cir. 2017)"; "In re Nuvasive (Fed. Cir. 2016)"), putting a greater emphasis on fact findings consistent with the ultimate legal question of obviousness in addition to fully satisfying the legal requirements of Section 103 of the Patent Statute.
The case involved Velcade®, Millennium's oncology product prescribed for multiple myeloma and mantle cell lymphoma and protected by U.S. Patent No. 6,713,446 ("the '446 Patent"). As described in the Federal Circuit opinion, the '446 patent is directed to D-mannitol N-(2-pyrazine)carbonyl-L-phenylalanine-L-leucine boronate (a D-mannitol ester of bortezomib):
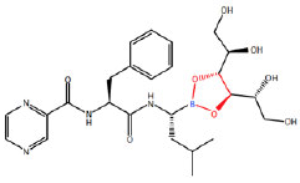 where the portion of the molecule in red identifies the ester linkage between bortezomib and the mannitol sugar. This compound is claimed in claim 20 of the '446 patent:
where the portion of the molecule in red identifies the ester linkage between bortezomib and the mannitol sugar. This compound is claimed in claim 20 of the '446 patent:
20. The lyophilized compound D-mannitol N-(2-pyrazine)carbonyl-L-phenylalanine-L-leucine boronate.
Importantly, the other invalidated claims recited methods for preparing this compound, a lyophilized embodiment thereof and reconstitution of the lyophilized form into an injectable pharmaceutical formulation. Bortezomib itself was known in the prior art; indeed, it was described and claimed in U.S. Patent No. 5,780,454 ("the '454 Patent"). Also known was the compound's activity as a proteasome inhibitor and its anticancer properties. However, it was also known to be highly unstable, insoluble in aqueous-based pharmaceutical excipients and to rapidly degrade in liquid formulations. Accordingly, no form or formulation of the compound had ever been FDA approved or marketed, despite record evidence of "extensive research efforts" ("approximately 20 different formulations") by the '454 inventors and Millennium.
A solution came from a researcher at the National Cancer Institute and the University of Kansas, as disclosed and claimed in the '446 patent (licensed by Millennium). This researcher had encountered difficulties with bortezomib similar to the Millennium researchers ("preparing approximately twenty-five different liquid formulations") and then turned to lyophilized forms. Lyophilization was a known technique that was "not intended to change the chemical structure of the active pharmaceutical ingredient." During these experiments, lyophilization in the presence of D-mannitol was performed using the sugar for its recognized properties as a bulking agent. The resulting lyophilized formulation was found to have "dramatic improvement in dissolution and stability." Presumably (in view of the Court's decision) unexpectedly, "[i]t was discovered that the reason for the dramatic improvement in dissolution and stability for this formulation was the formation of a new chemical compound during lyophilization: the claimed ester of bortezomib and mannitol."
 ANDA litigation arose when Sandoz Inc., Accord Healthcare, Inc., Actavis LLC, Mylan Laboratories Ltd., Agila Specialties Inc., Dr. Reddy's Laboratories, Ltd., Dr. Reddy's Laboratories, Inc., Sun Pharmaceutical Industries Ltd., Sun Pharma Global FZE, Glenmark Pharmaceuticals Ltd., Glenmark Generics Ltd., Glenmark Generics Inc., USA, Hospira, Inc., Wockhardt Bio Ag, and Wockhardt USA LLC (collectively referenced as "Sandoz" in the opinion) each filed ANDAs and sent Paragraph IV letters to Millennium over Velcade®. In separate litigations initiated after the first collection of lawsuits but before the District Court had made its determination in those cases, Millennium sued Teva Pharmaceuticals USA, Inc. and Apotex Corp. and Apotex Inc. based on their ANDA filings and Paragraph IV letters.
ANDA litigation arose when Sandoz Inc., Accord Healthcare, Inc., Actavis LLC, Mylan Laboratories Ltd., Agila Specialties Inc., Dr. Reddy's Laboratories, Ltd., Dr. Reddy's Laboratories, Inc., Sun Pharmaceutical Industries Ltd., Sun Pharma Global FZE, Glenmark Pharmaceuticals Ltd., Glenmark Generics Ltd., Glenmark Generics Inc., USA, Hospira, Inc., Wockhardt Bio Ag, and Wockhardt USA LLC (collectively referenced as "Sandoz" in the opinion) each filed ANDAs and sent Paragraph IV letters to Millennium over Velcade®. In separate litigations initiated after the first collection of lawsuits but before the District Court had made its determination in those cases, Millennium sued Teva Pharmaceuticals USA, Inc. and Apotex Corp. and Apotex Inc. based on their ANDA filings and Paragraph IV letters.
In the first litigations, the District Court found asserted claims 20, 31, 49, and 53 of the '446 Patent to be invalid for obviousness. The District Court held that the claims were "the inherent result of an allegedly obvious process," specifically lyophilizing bortezomib and mannitol together. Bortezomib and mannitol were both known in the art, bortezomib having anticancer properties and mannitol being useful as a bulking agent; lyophilization was known in the art; and the produced ester (admittedly resulting merely from lyophilization of bortezomib and mannitol together) was thus the inherent result. Millennium argued (unsuccessfully) that the skilled worker would not have used lyophilization with a compound with bortezomib's known (in)stability characteristics, but the District Court rejected this argument in view of testimony from Sandoz's expert that "lyophilization 'was well-known in the field of formulation' and that it was considered an obvious alternative 'when a liquid formulation provided limited success.'" Foreshadowing its decision, the panel opinion set out the factual findings the District Court had not made:
The district court did not find that the prior art taught or suggested that the claimed new compound would be formed, or taught or suggested making the claimed new compound by any method, or taught or suggested that this new compound would have the properties of stability, solubility, and dissociability that it exhibited. No reference taught or suggested reacting bortezomib with mannitol, and no reference hinted that such an esterification reaction might occur during lyophilization. No reference taught or suggested that the product of such lyophilization would be a new chemical compound that would solve the problems that had inhibited development of bortezomib in oncology.
In addition, the District Court stated that the disclosure of the '454 patent "pointed directly to mannitol" despite there being no such disclosure in this patent. The District Court apparently also disregarded inventor testimony ("and others") that the new compound was neither intended nor expected (there being "no contrary evidence" according to the Federal Circuit), finding that "the 'natural result' of a chemical procedure is inherent in the procedure, and thus the product thereof 'would have been obvious to a person of ordinary skill.'" The District Court also rejected Millennium's assertion of objective indicia (unexpected results and long-felt need) of non-obviousness.
In the later-filed actions, the District Court entered (stipulated) final judgment in favor of Teva and the Apotex entities based on collateral estoppel. The parties' appeals were consolidated before the Federal Circuit.
The Court reversed, in an opinion by Judge Newman joined by Judges Mayer and O'Malley. Giving due regard to the deference owed to the District Court on factual inquiries related to obviousness ("[r]ecognizing our obligation to give deference to a district court's greater familiarity with the record and authority to reach factual conclusions therefrom"), the panel nevertheless found that the District Court had erred in applying the Supreme Court's obviousness standard under KSR Int'l Co. v. Teleflex Inc. For the Federal Circuit:
[T]he question is whether a person of ordinary skill, seeking to remedy the known instability and insolubility and to produce an efficacious formulation of bortezomib, would obviously produce the D-mannitol ester of bortezomib, a previously unknown compound.
The Court's reasoning as to why its answer to this question differs from the District Court's is as follows. According to the opinion, there was no "teaching or suggestion" in the art regarding the claimed (novel) compound, nor was there any appreciation in the prior art that this compound would be formed during lyophilization of the combination of bortezomib and mannitol, or would have the improved stability and solubility of the compound claimed in the '446 patent. Citing post-KSR chemical obviousness precedent (including Otsuka Pharm. Co., Ltd. v. Sandoz, Inc.; Bristol-Myers Squibb Co. v. Teva Pharm. USA, Inc.; and In re Rosuvastatin Calcium Patent Litig.), the opinion sets forth its analysis that focused on whether the art would provide the skilled worker with a reason for modifying a "lead compound" in a way that would result in the claimed compound. (In this case, the parties agreed that lead compound to be bortezomib according to the opinion.) The Federal Circuit finds error in the District Court's obviousness determination because "[t]here is no teaching or suggestion in the references to produce the claimed mannitol ester," "[n]o reference shows or suggests ester formation at freeze-drying conditions, or that any such ester might solve the problems of instability and insolubility of the free acid while dissociating rapidly in the bloodstream," and "[n]o reference provides a reason to make the mannitol ester of bortezomib."
The panel conceded that lyophilization was a generally known method for pharmaceutical formulation, that bulking agents were known to be used in such lyophilizations, and that mannitol was a known bulking agent. What was unknown in the prior art was that lyophilizing these two compounds would "produce a chemical reaction and form a new chemical compound," "provide a reason to make this specific new chemical compound," or make a new compound that would provide a solution to the "previously intractable problems of bortezomib formulation" useful as an anticancer pharmaceutical. The panel states that the art cited by Sandoz to support its obviousness contentions are lacking these characteristics and that the expert testimony proffered by the Sandoz defendants does not "fill the gaps" in the art. Even references such as the '454 patent disclosing boronic acid esters of bortezomib say "nothing about the stability of any ester." Indeed, the panel agreed with Millennium's position that the skilled worker would have avoided mannitol esterification because "several different esters, each with different chemical and possibly biological properties" might result. The opinion suggests that obviousness requires the art to provide a reason for taking the particular experimental route leading to the claimed success, as well as a reasonable expectation of achieving that success. Instances, as here, where the result is unforeseen and serendipitous cannot satisfy these criteria.
As for the purported "inherency" of the outcome relied upon by the District Court in its reasoning, the panel found that the District Court had erred in agreeing with Sandoz that, because the result (the claimed bortezomib-mannitol ester) was "inevitable" it was also inherent, and therefore could not be inventive (in the archaic meaning of that term, i.e. non-obvious). The error stems from the principle that the inventor's intention is not the relevant consideration; it is whether the invention would have been objectively obvious to one of ordinary skill in the art. Here, there was no evidence that anyone "foresaw, or expected, or would have intended, the reaction between bortezomib and mannitol, or that the resulting ester would have the long-sought properties and advantages" and hence the invention was non-obvious.
Finally with regard to the District Court's obviousness determination, the Federal Circuit found that there was an insufficient consideration of the objective indicia of non-obviousness, which factors can be "independent evidence of nonobviousness," citing Ortho-McNeil Pharm., Inc. v. Mylan Labs., Inc., 520 F.3d 1358, 1365 (Fed. Cir. 2008). Regarding evidence of unexpected results, the opinion asserts that Millennium adduced expert testimony regarding the properties of the compound prepared by lyophilizing bortezomib together with mannitol (specifically, "greatly improved stability, solubility, and dissolution"). The District Court erred by determining that Millennium should have compared Velcade® with the glycerol esters of bortezomib as taught in the '454 patent. However, according to the opinion, these esters are not specifically disclosed, nor prepared, nor tested in the '454 patent, and that patent is devoid of evidence regarding stability or solubility (increased or otherwise) of bortezomib esters. The principle, from Pfizer, Inc. v. Apotex, Inc., is that "[u]nexpected results are shown in comparison to what was known, not what was unknown" and the absence in the prior art of specific disclosure regarding bortezomib esters makes bortezomib itself the closest prior art compound. In view of the known stability and solubility deficiencies of bortezomib, the mannitol ester claimed in the '446 patent has improvements in these properties not disclosed in the prior art that support a determination of non-obviousness. On the question of long-felt need, the Court found the District Court's conclusion "both perfunctory and clearly erroneous," there being "no dispute" that there was a need for anticancer treatment for multiple myeloma and the claimed compound satisfied that need. (In a footnote, the opinion countered Sandoz's argument that others failed because the '454 patent precluded them from bringing bortezomib ester formulations to market by reminding Sandoz that long-felt need and failure of others while closely related were distinct indicia and long-felt need can be established without evidence that others had failed.) The Court also considered it error for the District Court to attribute Velcade's® success to bortezomib itself.
With regard to judgment against Millennium in favor of Teva and independently the Apotex companies, all parties agreed that dismissal would be and accordingly was vacated and the case remanded, giving these generic drug makers a chance to make their invalidity cases against the asserted claims of the '446 patent.
Millennium Pharmaceuticals, Inc. v. Sandoz Inc. (Fed. Cir. 2017)
Panel: Circuit Judges Newman, Mayer, and O'Malley
Opinion by Circuit Judge Newman
 Strafford will be offering a webinar/teleconference entitled "Patents and Export Control Compliance: Managing Risk and Avoiding Unintentional Violations — Minimizing Export Control Liability in Patent Application Preparation, Development and Analysis of Innovation, and Licensing" on August 3, 2017 from 1:00 to 2:30 pm (EDT). David G. Henry of Gray Reed & McGraw and Edward J. Radlo of Radlo IP Law Group will provide guidance for patent counsel on navigating the intersection of patents and export control laws; examine the challenges facing patent owners, when and where export control issues arise, and licensing issues; and offer best practices for complying with export control laws. The webinar will review the following topics:
Strafford will be offering a webinar/teleconference entitled "Patents and Export Control Compliance: Managing Risk and Avoiding Unintentional Violations — Minimizing Export Control Liability in Patent Application Preparation, Development and Analysis of Innovation, and Licensing" on August 3, 2017 from 1:00 to 2:30 pm (EDT). David G. Henry of Gray Reed & McGraw and Edward J. Radlo of Radlo IP Law Group will provide guidance for patent counsel on navigating the intersection of patents and export control laws; examine the challenges facing patent owners, when and where export control issues arise, and licensing issues; and offer best practices for complying with export control laws. The webinar will review the following topics: