By John Cravero —
 About the PTAB Life Sciences Report: Each month we will report on developments at the PTAB involving life sciences patents.
About the PTAB Life Sciences Report: Each month we will report on developments at the PTAB involving life sciences patents.
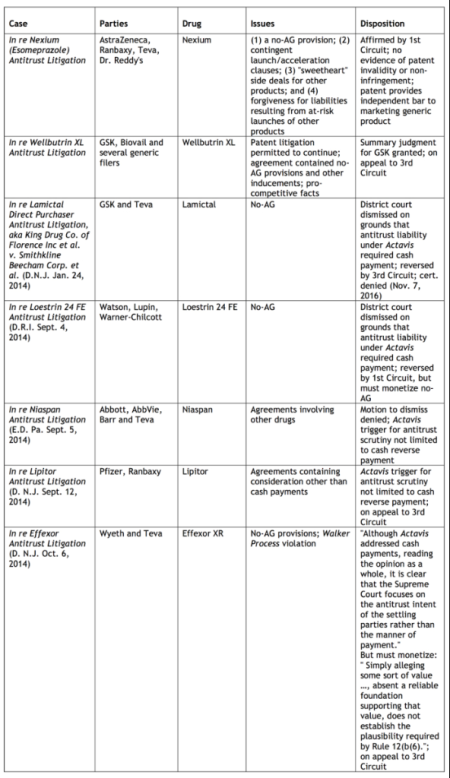
Celltrion, Inc. v Genentech, Inc.
PTAB Petition: IPR2017-01121; filed March 21, 2017.
Patent at Issue: U.S. Patent No. 7,846,441 ("Treatment with anti-ErbB2 antibodies," issued December 7, 2010) claims a method for the treatment of a human patient with a malignant progressing tumor or cancer characterized by overexpression of ErbB2 receptor, comprising administering a combination of an intact antibody which binds to epitope 4D5 within the ErbB2 extracellular domain sequence and a taxoid, in the absence of an anthracycline derivative, to the human patient in an amount effective to extend the time to disease progression in said human patient, without increase in overall severe adverse events.
Petitioners Celltrion, Inc., Celltrion Healthcare Co. Ltd., and Teva Pharmaceuticals International GmbH are challenging the '441 patent on one ground as being obvious under 35 U.S.C. § 103(a). View the petition here.
Related Matters: According to the petition, the '441 patent is involved in inter partes review IPR2017-00731 (Hospira, Inc.; filed 01/20/2017; pending). Petitioner concurrently filed a petition for inter partes review of related U.S. Patent No. 7,892,549 (IPR2017-01122; filed 03/21/2017; pending), which is also the subject of IPR2017-00737 (Hospira, Inc.; filed 01/20/2017; pending) and IPR2017-00739 (Hospira, Inc.; filed 01/20/2017; pending).
Celltrion, Inc. v Genentech, Inc.
PTAB Petition: IPR2017-01122; filed March 21, 2017.
Patent at Issue: U.S. Patent No. 7,892,549 ("Treatment with anti-ErbB2 antibodies," issued February 22, 2011) claims a method for the treatment of a human patient with breast cancer that overexpresses ErbB2 receptor, comprising administering a combination of an antibody that binds ErbB2, a taxoid, and a further growth inhibitory agent to the human patient in an amount effective to extend the time to disease progression in the human patient, wherein the antibody binds to epitope 4D5 within the ErbB2 extracellular domain sequence.
Petitioners Celltrion, Inc., Celltrion Healthcare Co. Ltd., and Teva Pharmaceuticals International GmbH are challenging the '549 patent on one ground as being obvious under 35 U.S.C. § 103(a). View the petition here.
Related Matters: According to the petition, the '549 patent is involved in inter partes reviews IPR2017-00737 (Hospira, Inc.; filed 01/20/2017; pending) and IPR2017-00739 (Hospira, Inc.; filed 01/20/2017; pending). Petitioner concurrently filed a petition for inter partes review of related U.S. Patent No. 7,846,441 (IPR2017-01123; filed; pending), which is also the subject of IPR2017-00731 (Hospira, Inc.; filed 01/20/2017; pending).
Sanofi-Aventis U.S. LLC. v Immunex Corp.
PTAB Petition: IPR2017-01129; filed March 23, 2017.
Patent at Issue: U.S. Patent No. 8,679,487 ("Anti-interleukin-4 receptor antibodies," issued March 25, 2014) claims an isolated human antibody that competes with a reference antibody for binding to human IL-4 interleukin-4 (IL-4) receptor.
Petitioners Sanofi-Aventis U.S. LLC., Genzyme Corp., and Regeneron Pharmaceuticals, Inc. are challenging the '487 patent on one ground as being anticipated under 35 U.S.C. § 102(b). View the petition here.
Related Matters: According to the petition, the '487 patent is involved in the following litigation: Sanofi-Aventis U.S. LLC et al. v. Amgen Inc. et al., 17-cv-10465 (D. Ma.).
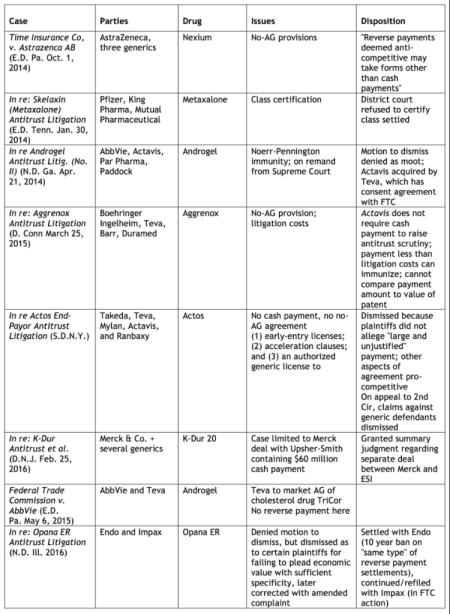
Fisher & Paykel Healthcare Ltd. v. Resmed Ltd.
PTAB Petition: IPR2017-00061; filed October 12, 2016.
PTAB Trial Instituted; entered March 23, 2017.
Patent at Issue: U.S. Patent No. 9,119,931 ("Mask system," issued September 1, 2015) claims a mask system, comprising: (i) a shroud module; wherein the shroud module includes headgear connectors adapted to removably attach to respective headgear straps of headgear; and (ii) a cushion module, comprising: a rigid or semi-rigid frame defining a breathing chamber; and a cushion to form a seal with the patient's face in a nasal bridge region, a cheek region and a lower lip/chin region of the patient's face, wherein the cushion is constructed of a first, relatively soft, elastomeric material and the frame is constructed of a second material that is more rigid than the cushion, wherein the shroud module and the cushion module are configured to be removably and non-rotatably coupleable to one another; and wherein the frame includes a protruding vent arrangement having a plurality of holes, wherein the shroud module includes a first opening to accommodate said protruding vent arrangement, and further wherein the shroud module includes a second opening positioned to align with a frame opening of the frame leading to the breathing chamber.
Petitioner Fisher & Paykel Healthcare Ltd. is challenging the '931 patent on eleven grounds as being obvious under 35 U.S.C. § 103(a). View the petition here. Administrative Patent Judges Richard E. Rice (author), Barry L. Grossman, and James J. Mayberry issued a decision instituting inter partes review of whether claims 23–78, 80–82, and 84–86 are obvious under 35 U.S.C. § 103(a) over Ogden, Lovell, and Gunaratnam; and whether claims 79 and 83 are obvious under 35 U.S.C. § 103(a) over Ogden, Lovell, Gunaratnam, and Geist.
Related Matters: According to the petition, the '931 patent is involved in U.S. International Trade Commission Investigation No. 337–TA–1022. The '931 patent is also the subject of the following litigation: Fisher & Paykel Healthcare Ltd. v. ResMed Corp., Case No. 3:16-cv- 02068 (S.D. Cal.). Petitioner has filed three additional petitions seeking inter partes review of the '931 patent (IPR2017-00062 (Fisher & Paykel Healthcare Ltd.; filed 10/12/2016; Instituted 03/31/2017); IPR2017-00064 (Fisher & Paykel Healthcare Ltd.; filed 10/12/2016; Institution denied 04/11/2017); and IPR2017-00065 (Fisher & Paykel Healthcare Ltd.; filed 10/12/2016; Institution denied 04/11/2017).
Medtronic Xomed, Inc. v. Resmed Ltd.
PTAB Petition: IPR2016-01847; filed September 19, 2016.
PTAB Trial Instituted; entered March 23, 2017.
Patent at Issue: U.S. Patent No. 8,467,844 ("Electrode for prolonged monitoring of laryngeal electromyography," issued September 1, 2015) claims a device for use in monitoring electrical signals during laryngeal electromyography comprising: an endotracheal tube having a retention balloon at or adjacent a distal end thereof, said tube having on its outer surface one or more electrically conductive electrode plates applied proximal of the balloon directly to the surface of the tube, without the inclusion of a carrier film between the tube surface and the electrode plates, said tube having on its surface electrically conductive traces connected to or integral with the electrode plates, the traces applied directly to the tube surface and running along the length of the endotracheal tube to a proximal end thereof, conductive pads connected to or integral with the conductive traces, the pads applied directly to the tube surface at the proximal end of the endotracheal tube, and electrical leads connected to the pads, said leads adapted to connect to monitoring equipment, the electrically conductive traces covered by an insulating material along their length from a point adjacent the electrode plates to a point adjacent the conductive pads wherein a first of said electrode plates is located proximal of the balloon and positioned to contact the vocal cords when placed within the trachea and a second electrode plate is located further proximal thereof and positioned to contact the tongue when the first electrode plate is positioned to contact the vocal cords.
Petitioner Medtronic Xomed, Inc. is challenging the '844 patent on two grounds as being obvious under 35 U.S.C. § 103(a). View the petition here. Administrative Patent Judges Meredith C. Petravick (author), William V. Saindon, and Richard E. Rice issued a decision instituting inter partes review of whether claims 1–7 are obvious under 35 U.S.C. § 103 over Kartush, Topsakal, Cook, and Hon; and whether claims 1–7 are obvious under 35 U.S.C. § 103 over Goldstone, Teves, Cook, and Hon.
Related Matters: According to the petition, the '844 patent is the subject of the following litigation: Neurovision Med. Prods. v. Medtronic Pub. Ltd. Co., Case No. 2:16-cv- 00127 (E.D. Tex.). Also, according to the petition, related U.S. Patent No. 8,634,894 B2 ("the '894 patent"), which claims priority to the '844 patent, is the subject of pending inter partes reviews IPR2016-01405 (Medtronic Xomed, Inc.; filed 07/11/2016; instituted 12/29/2016; pending); IPR2016-01406 (Medtronic Xomed, Inc.; filed 07/11/2016; instituted 12/29/2016; pending); and IPR2017-00456 (Medtronic Xomed, Inc.; filed 12/09/2017; pending). Additionally, the '894 patent was the subject of IPR2015- 00502 (NuVasive, Inc.; filed 12/24/2104; instituted 07/16/2015; terminated due to settlement 07/29/2015).
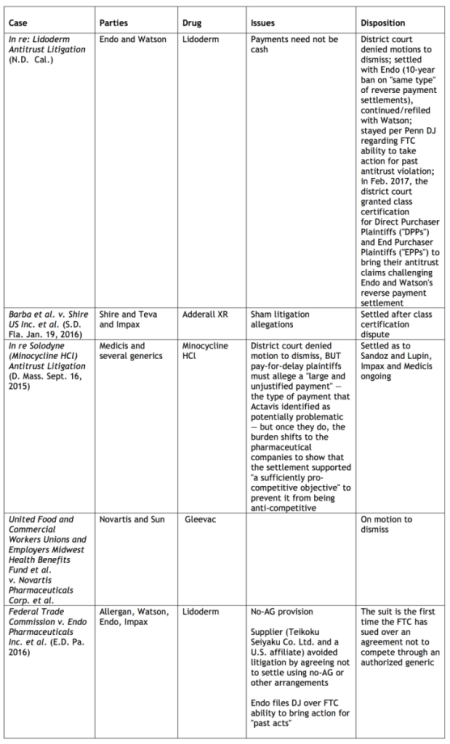
Natus Medical Inc. v. Nox Medical Ehf
PTAB Petition: IPR2016-01822; filed September 15, 2016.
PTAB Trial Instituted; entered March 23, 2017.
Patent at Issue: U.S. Patent No. 9,059,532 ("Biometric belt connector," issued June 16, 2015) claims an electrode belt and a belt connector for electrically connecting a conductor of the electrode belt to a male portion of a snap connector electrode connected to a biometric device, the belt connector comprising: a molded plastic frame including a receiving hole having radial flexibility, the receiving hole being configured to function as a female snap button fastener for receiving and fastening the frame to a protrusion of the male portion of the snap connector electrode, a fastener configured to fasten the frame to a first end of said electrode belt, and an engaging member adjacent to said receiving hole, the engaging member engaging the conductor of the electrode belt by the conductor passing through the receiving hole while being wrapped around the engaging member, such that when the male portion of the snap connector electrode penetrates the receiving hole, the conductor is forced into physical contact with at least a lateral surface of the male portion of the snap connector electrode, wherein radial flexibility of said receiving hole is achieved by one or more slot extending from said hole, and wherein said receiving hole and one or more slot are formed by at least one elongated member having flexibility transverse to its longitudinal axis, thus imparting flexibility to the width of the hole.
Petitioners Natus Medical Inc., Natus Neurology Inc., Embla Systems LLC, and Embla Systems Ltd. are challenging the '532 patent on eight grounds as being anticipated under 35 U.S.C. § 102(b) (ground 1) or obvious under 35 U.S.C. § 103(a) (grounds 2-8). View the petition here. Administrative Patent Judges Erica A. Franklin, Susan L. C. Mitchell (author), and Amanda F. Wieker issued a decision instituting inter partes review of whether claims 1–9 and 13 are obvious under 35 U.S.C. § 103(a) over the McIntire and Kristbjarnarson or Linville in view of Archer, Caldecott, Uehara, Abizaid, or Orewiler.
Related Matters: According to the petition, the '532 patent is the subject of the following litigation: Nox Medical Ehf. v. Natus Neurlogy Inc., 15-709 (D. Del.).
Reactive Surfaces LTD., LLP v. Toyota Motor Corp.
PTAB Petition: IPR2016-01914; filed September 30, 2016.
PTAB Trial Instituted; entered March 23, 2017.
Patent at Issue: U.S. Patent No. 8,394,618 ("Lipase-containing polymeric coatings for the facilitated removal of fingerprints," issued March 12, 2013) claims a method of facilitating the removal of a fingerprint on a substrate or a coating comprising: providing a substrate or a coating; associating a lipase with said substrate or said coating such that said lipase is capable of enzymatically degrading a component of a fingerprint, and facilitating the removal of a fingerprint by vaporization from the lipase associated substrate or coating when contacted by a fingerprint.
Petitioner Reactive Surfaces Ltd., LLP is challenging the '618 patent on eight grounds as obvious under 35 U.S.C. § 103(a). View the petition here. Administrative Patent Judges Christopher M. Kaiser (author), Jeffrey W. Abraham, and Michelle N. Ankenbrand issued a decision instituting inter partes review of whether claims 1–9 and 13 are obvious under 35 U.S.C. § 103(a) over McIntire and Kristbjarnarson or Linville in view of Archer, Caldecott, Uehara, Abizaid, or Orewiler.
Related Matters: According to the petition, there are no judicial or administrative matters that involve the '618 patent.