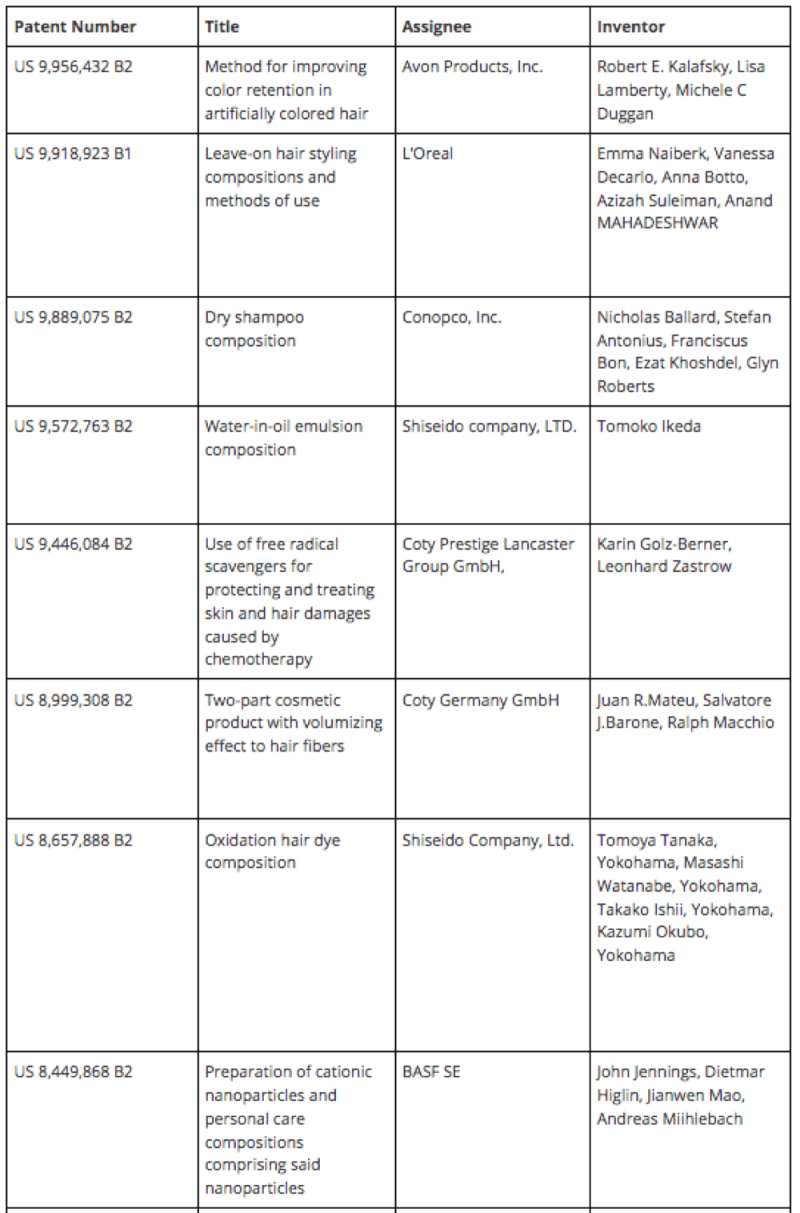
By Bryan Helwig —
 About Court Report: Each week we will report briefly on recently filed biotech and pharma cases, and a few interesting cases will be selected for periodic monitoring.
About Court Report: Each week we will report briefly on recently filed biotech and pharma cases, and a few interesting cases will be selected for periodic monitoring.
Fresenius Kabi USA, LLC v. Eurohealth International Sarl
1-18-cv-00835; filed June 1, 2018 in the District Court of Delaware
• Plaintiffs: Fresenius Kabi USA, LLC and Fresenius Kabi Deutschland GmbH
• Defendant: Eurohealth International Sarl
Claim: Infringement of U.S. Patent Nos.
• 9,248,229: ″Packaging System for Oxygen-Sensitive Drugs″
• 9,731,082: ″Drug Container″
Synopsis: Fresnius claims infringement of the ′082 and ′229 patents. Fresenius owns all rights, title, and interest in the '082 and '229 patents. Fresenius is the holder of NDA No. 019034 for Dilaudid® Injection. Eurohealth submitted amendments to ANDA No. 202159 to engage in the commercial manufacture, use, importation, offer for sale, or sale of generic Deluadid® Injection.
View the complaint here.
Cipla Ltd. v. Eli Lilly and Company
1-18-cv-01671; filed June 1, 2018 in the Southern District of Indiana, Indianapolis Division
• Plaintiffs: CIPLA Ltd. and CIPLA USA, Inc.
• Defendants: Eli Lilly and Company and ICOS Corp.
Claim: Infringement of U.S. Patent Nos.
• 6,821,975: ″Beta-carboline drug products″
• 7,182,958: ″β-Carboline Pharmaceutical Compositions″
Synopsis: Cipla seeks a declaratory judgment that the ′975 and ′958 patents are invalid. ICOS is the assignee of the ′975 and ′958 patents. Lilly is the exclusive licensee of both patents. The patents are associated with submitted NDA No. 022332. Cipla was not the first generic drug manufacturer to file an ANDA directed to tadalafil tablets in the 20 mg dosage strength. Cipla sent Lilly and ICOS notice of Cipla's Paragraph IV certification with ANDA No. 210255. Cipla's Notice Letter initiated a 45-day statutory period during which Defendants did not file an action against Cipla for infringement of the '975 or '958 patents. Cipla believes the patents are an impediment to entry into the market. As a subsequent ANDA filer, Cipla's ANDA for 20 mg tadalafil tablets cannot be approved by the FDA until the first ANDA filer's 180-day exclusivity period is either forfeited or runs out. As of the date of this Complaint, the '975 and '958 patents are the only two remaining unexpired patents listed on the Orange Book for NDA No. 022332. As such, there are no patents barring the first ANDA filer from entering the market. Additionally, no first ANDA filer has entered the market. Cipla seeks a declaratory judgment to allow entry into the market.
View the complaint here.
Astellas Parma Inc. v. Cipla Ltd.
1-18-cv-00844; filed June 5, 2018 in the District of Delaware
• Plaintiffs: Astellas Pharma Inc.; Astellas Pharma U.S., Inc.; Astellas Ireland Co., Ltd.; and Astellas Pharma Europe Ltd.
• Defendant: Cipla Ltd.
Claim: Infringement of U.S. Patent No.
• 6,017,927: ″Quinuclidine derivatives and medicinal composition thereof″
Synopsis: Astellas claims infringement of the ′927 patent. Astellas Pharma U.S., Inc. holds approved NDA No. 21518 for VESIcare® tablets in 5 mg and 10 mg strength tablet dosage forms, which contain the active ingredient solifenacin succinate. VESIcare® is indicated for treatment of overactive bladder with symptoms of urge urinary incontinence, urgency, and urinary frequency. Cipla submitted ANDA No. 209839 for generic solifenacin succinate 5 mg and 10 mg tablets, as a pharmaceutical composition in an oral dosage form for the treatment of overactive bladder prior to the expiration of the '927 patent. In response, Plaintiffs filed for patent infringement.
View the complaint here.
Biozone Laboratories, Inc. v. Next Step Laboratories Corp.
1-18-cv-03296; filed June 5, 2018 in the Eastern District of New York (Brooklyn)
• Plaintiff: Biozone Laboratories, Inc.
• Defendants: Next Step Laboratories Corp. and Richard Rigg
Claim: Infringement of U.S. Patent Nos.
• 6,610,322: ″Self forming, themodynamically stable liposomes and their applications″
• 6,958,160: ″Self forming, thermodynamically stable liposomes and their applications″
• 6,495,596: ″Compounds and methods for inhibition of phospholipase A2 and cyclooxygenase-2″
• 6,998,421: ″Compounds and methods for inhibition of phospholipase A2 and cyclooxygenase–2″
• 7,150,883: ″Self forming, thermodynamically stable liposomes and their applications″
• 7,718,190: ″Self forming, thermodynamically stable liposomes and their applications
Synopsis: BioZone claims infringement of patents ′322, ′160, ′596, ′421, ′883, and ′190. The Patents-in-Suit cover BioZone’s proprietary QuSome® and Inflacin® proprietary technologies. Plaintiffs claim Defendants directly and indirectly infringe, by offering to sell QuSomes Products according to the claims of the patents-in-suit.
View the complaint here.
H. Lundbeck A/S v. MSN Laboratories Private Ltd.
1-18-cv-00853; filed June 7, 2018 in the District of Delaware (Wilmington)
• Plaintiffs: H. Lundbeck A/S, Takeda Pharmaceutical Co. Ltd., Takeda Pharmaceuticals U.S.A., Inc., Takeda Pharmaceuticals International AG, and Takeda Pharmaceuticals America, Inc.
• Defendants: MSN Laboratories Private Ltd., MSN Pharmaceuticals, Inc., and MSN Pharmachem Private Ltd.
Claim: Infringement of U.S. Patent No:
• 9,861,630: ″1-[2-(2,4-dimethylphenylsulfanyl)-phenyl]piperazine as a compound with combined serotonin reuptake, 5-HT.sub.3 and 5-HT.sub.1A activity for the treatment of cognitive impairment″
Synopsis: Plaintiffs claim infringement of the ′630 patent. Plaintiff H. Lundbeck is the assignee and owner of the ′630 patent. Lundbeck granted Takeda Japan an exclusive license to the ′630 patent. Takeda Japan issued sublicenses to its subsidiary Plaintiffs. Takeda USA is the holder of New Drug Application ("NDA") No. 204447 for TRINTELLIX® tablets (5 mg, 10 mg, 15 mg, and 20 mg dosage strengths) containing the active ingredient vortioxetine hydrobromide for the treatment of Major Depressive Disorder. Defendants have submitted ANDA No. 211101 to FDA, or caused ANDA No. 211101 to be submitted to FDA, under 21 U.S.C. § 355(j), in order to obtain approval to engage in the commercial manufacture, use, or sale of vortioxetine hydrobromide tablets as purported generic versions of TRINTELLIX® tablets prior to the expiration of the ’630 Patent.
View the complaint here.
Purdue Pharma LP v. Ascent Pharmaceuticals, Inc.
1-18-cv-00855; filed June 7, 2018 in the District Court of Delaware (Wilmington)
• Plaintiffs: Purdue Pharma L.P., Purdue Pharmaceuticals L.P., P.F. Laboratories, Inc., Rhodes Technologies, and Grünenthal GmbH
• Defendant: Ascent Pharmaceuticals, Inc.
Claim: Infringement of U.S. Patent Nos:
• 8,309,060: ″Abuse-proofed dosage form″
• 9,060,976: ″Pharmaceutical formulation containing gelling agent″
• 9,073,933: ″Oxycodone hydrochloride having less than 25 PPM 14-hydroxycodeinone″
• 9,149,533: ″Tamper resistant pharmaceutical formulations″
• 9,522,919: ″Oxycodone compositions″
• 9,675,610: ″Abuse-proofed dosage form″
• 9,861,582: ″Pharmaceutical formulation containing gelling agent″
Synopsis: Plaintiffs claim infringement of the ′060, ′976, ′933, ′533, ′919, ′610, and ′582 patents. Plaintiffs have approved NDA No. 022272 in the 10 mg, 15 mg, 20 mg, 30 mg, 60 mg and 80 mg dosage strengths. Defendant filed ANDA No. 211178 seeking approval to engage in the commercial manufacture, use, or sale of Defendant's ANDA Products, a generic product based on the Reference Listed Drug OxyContin®, which is the subject of approved NDA No. 022272. Plaintiffs sued for patent infringement.
View the complaint here.
Teijin Ltd. v. MSN Laboratories Private Ltd.
1-18-cv-00881; filed June 7, 2018 in the District Court of Delaware (Wilmington)
• Plaintiffs: Teijin Ltd., Teijin Pharma Ltd., and Takeda Pharmaceuticals U.S.A., Inc.
• Defendants: MSN Laboratories Private Ltd. and MSN Pharmaceuticals, Inc.
Claim: Infringement of U.S. Patent Nos:
• 7,361,676: ′Solid preparation containing single crystal form″
• 8,372,872: ″Methods for concomitant treatment of theophylline and febuxostat″
• 9,107,912: ″Methods for concomitant treatment of theophylline and febuxostat″
Synopsis: Plaintiffs claim infringement of the ′676, ′872, and ′912 patents. Plaintiffs own the patents noted. In addition, Takeda holds NDA No. 21-856 for oral tablets containing 40 mg or 80 mg of the active ingredient febuxostat. Takeda markets and sells these tablets in the United States under the brand name Uloric®. MSN certified in ANDA No. 210461 that the claims of 7 the patents-in-suit are invalid, unenforceable, or would not be infringed by the commercial manufacture, use, offer for sale, or sale of MSN’s Generic Products. Plaintiff claims the Defendant infringes the claims of the ′676, ′872, and ′912 patents.
View the complaint here.
 June 25-27, 2018 – Summit on Biosimilars (American Conference Institute) – New York, NY
June 25-27, 2018 – Summit on Biosimilars (American Conference Institute) – New York, NY