By Kevin E. Noonan –
 It came as something of a shock in May, 2021 when the Biden administration voiced support for waiver of intellectual property protection mandated under the TRIPS agreement for COVID-19 vaccines (see "Biden Administration Supports Waiver of IP Protection for COVID-19 Vaccines"). The waiver had originally been proposed to the World Trade Organization (WTO) by India and South Africa; in that incarnation a much broader swath of intellectual property protections had been proposed (encompassing diagnostics and therapeutic drugs related to the pandemic). When eventually imposed under the WTO's June 17, 2022 Ministerial Decision, the waiver was limited to vaccines with further consideration of waivers having broader scope postponed (until later this year, in fact).
It came as something of a shock in May, 2021 when the Biden administration voiced support for waiver of intellectual property protection mandated under the TRIPS agreement for COVID-19 vaccines (see "Biden Administration Supports Waiver of IP Protection for COVID-19 Vaccines"). The waiver had originally been proposed to the World Trade Organization (WTO) by India and South Africa; in that incarnation a much broader swath of intellectual property protections had been proposed (encompassing diagnostics and therapeutic drugs related to the pandemic). When eventually imposed under the WTO's June 17, 2022 Ministerial Decision, the waiver was limited to vaccines with further consideration of waivers having broader scope postponed (until later this year, in fact).
In anticipation of negotiations over expanding the IP waiver, the U.S. Trade Representative, Ambassador Katherine Tai sent a letter on December 16, 2022 to the International Trade Commission requesting the ITC to prepare a Report under section 332(g) of the Tariff Act of 1930 on the needs and justification for further expansion of the IP waiver. The Trade Representative asked that the ITC's Report be a product of market and other research within the Commission's expertise and also reflect the views of stakeholders and the public, to be garnered from public hearings and solicited comments from "foreign governments, non- governmental health advocates, organizations such as MPP and Foundation for Innovative New Diagnostics(FIND), and diagnostic and therapeutic manufacturers." The letter expressly disclaims any request for the Report to "draw any policy conclusions" and asked for the Report to be provided no later than October 17, 2023.
The ITC (Chairman David S. Johanson, Rhonda K. Schmidtlein, Jason E. Kearns, Randolph J. Stayin, and Amy A. Karpel) issued its almost 500-page Report, entitled "COVID-19 Diagnostics and Therapeutics: Supply, Demand, and TRIPS Agreement Flexibilities," on the deadline date (Ambassador's Tai's December 16, 2022 letter can be found as Appendix A to the Report). Containing as a prelude a list of abbreviations and glossary (necessary and advantageous for a report from any government agency or commission), which also identifies various companies, organizations, agencies in the U.S. and abroad, regulations, and proposals, the Report provides a succinct Executive Summary. Contained in this Summary are the sentiments that the COVID-19 pandemic had caused almost 7 million deaths worldwide and that there was a possibility (espoused by some) that IP could "act as a barrier to access, particularly in developing countries." Several highlights of this summary are:
• The TRIPS Agreement sets minimum standards for the protection and enforcement of IP rights. It also includes flexibilities (although there were differences of opinion regarding the scope of those flexibilities).
• Determining a definitive scope of what products are covered by the terms "diagnostics" and "therapeutics" as they pertain to COVID-19 and what constitutes relevant COVID-19 diagnostics and therapeutics covered by patents is complicated and subject to interpretation.
• The development and commercialization of COVID-19 diagnostics and therapeutics occurred on an extremely compressed timeline. Manufacturing of diagnostics and therapeutics involves multiple stages, each of which requires careful attention to detail and strict quality control measures. The two fields of diagnostics and therapeutics are composed of different producers, inputs, know-how, and so on (recognizing the unique characteristics of COVID-19 and thus suggesting caution in applying this recent history to future pandemics, but the commonalities such as diagnostics being marketed sooner than therapeutics). Data points: As of summer 2023, China (UMIC) reportedly had the highest number of manufacturers of COVID-19 diagnostics (247), and India (LMIC) had the largest number of reported therapeutics manufacturers (56).
• A variety of advantages and challenges are associated with the use of voluntary licenses to provide access to IP associated with COVID-19 therapeutics and compulsory licenses to access COVID-19 therapeutics and other patented pharmaceutical products (wherein neither licensing type was used during the pandemic).
• The availability of supplies to meet global demand for COVID-19 diagnostics and therapeutics has been a moving target throughout the pandemic (market demand or need? Varied over the time course of the pandemic).
• The disparity among countries of different income groups is wide in terms of access and availability to COVID-19 diagnostics and therapeutics (regarding government procurements: 80% HIC; 14% UMICs, 5% LMICs, none by LICs but multilateral organizations help in LICs. Recognition that this is multivariable, depending on "access to IP, prices and affordability, regulatory approvals, healthcare infrastructure, and the healthcare priorities of governments").
• Academic literature on the effects of patent protection, compulsory licenses, and the MPP is limited and would benefit from additional research ("From the available evidence, patent protection is generally found to be more beneficial to innovation in the health sector for developed countries and less so for developing countries.")
The Executive Summary also sets forth both the source of the information considered by the Commission (from academic and governmental literature, public and private sources, more than 120 interviews with stakeholders internationally, public hearings held March 29-20, 2023, and over 300 written submissions) and gaps in that information, which vary by country and region ("greatly"), in particular with regard to production costs. Also noted were gaps in information regarding IP-related agreements, including compulsory licenses and the existence of "very little price transparency" (which raises the question of the factual basis for the purported of the necessity for the waiver in the first place). Nor was there a great deal of information on consumption of COVID-19 related diagnostic or therapeutic moieties outside high income countries.
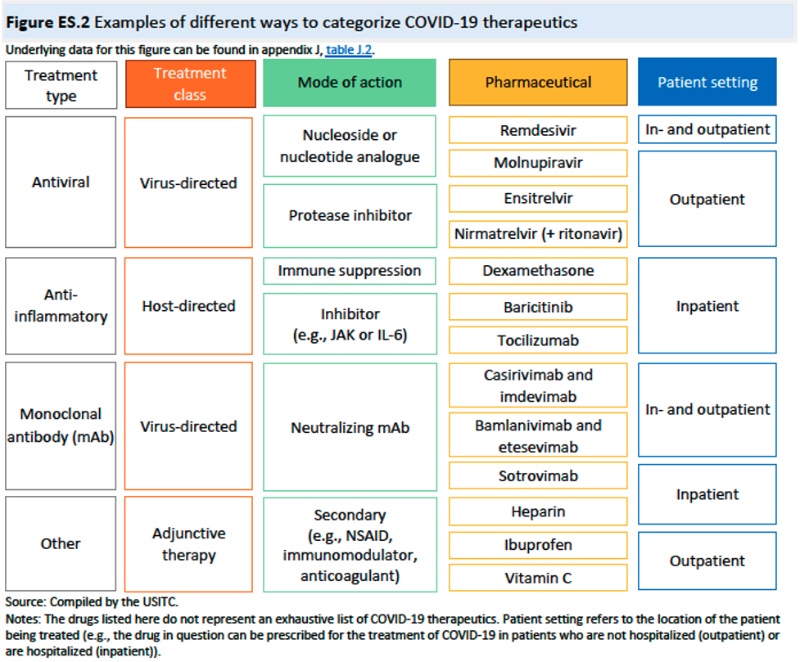
The Executive Summary also contains a brief review of IP and the TRIPS agreement, including an even briefer history of WTO grant of compulsory licenses in limited instances for pharmaceutical products. This portion of the Report identifies IP protections for COVID-19 related diagnostics and therapeutics (and expiry of the foundation IP on diagnostic methods) ("the best evidence that patents did not act as a primary barrier to the production of these tests is that many new producers and products, including in developing countries, came online relatively quickly"). The results in the Report include a "broad and varied" (and indeterminate) universe of the diagnostics that include "(1) tests used to diagnose an active infection of the SARS-CoV-2 virus, (2) tests that measure an adaptive immune response to the virus, and (3) tests used in the management of patients with COVID-19" and ancillary products (such as swabs, etc.). Therapeutic entities likewise include the drugs and delivery vehicles (syringes, etc.) for administering them. This portion of the Summary is devoid of any recognition or discussion of vaccines, in view of the mandate from the Trade Representative and the existing vaccine waiver. The Report provides schematic representations of how diagnostics and therapeutics were characterized:

With regard to supply chain issues (which in some ways were even more critical post-pandemic), the Report recognizes the multinational, interdependent nature of the pharmaceutical industry (see Cott et al., "How Pfizer Makes Its Covid-19 Vaccine," New York Times, April 28, 2021), and the complexities created by this situation. As of June/July 2023, the Report estimates there were 900 diagnostics manufacturers and 221 therapeutics manufacturers.
This Summary of the Report also addresses the source of COVID-related IP, which include R&D agreements, manufacturing partnerships, and licensing agreements (only R&D agreements were informative for diagnostics technology). While small- and medium-sized entities (SME) were responsible for development of new COVID therapeutics, larger pharmaceutical companies and the Federal government supplied economic and development support (and, according to the Report, was a source of calls for the resulting products and IP to be broadly and more cheaply available and those contracts be more transparent). Voluntary licenses provide one source of IP, which would disclose not only the patented invention but also technical information, know-how, and trade secrets the disclosure of which would undoubtedly raise resistance to this disclosure. While geographical restrictions are discussed, the Report also somewhat unrealistically posits royalty-free licenses as a norm. One advantage of such licenses noted in the Report was somewhat more fulsome disclosure than was obtained from compulsory licenses. Also mentioned with approval was disclosure though the Medicines Patent Pool ("a United Nations-backed public health organization"). So-called "upper-middle-income countries," defined by the World Bank as those economies with incomes per capita between $4,256 and $13,205, were not included in voluntary licenses for four COVID-19 therapeutics.
Turning to compulsory licenses, the Report notes that while the subject and provisions for and under which compulsory licenses for COVID-related IP was the subject of the 2022 Ministerial decision, no compulsory licenses to patents related to COVID-19 vaccines have been granted (although such licenses to IP relating to COVID-19 therapeutics have been granted in Hungary, Israel, and Russia). The Report briefly sets forth purported advantages and real challenges that arise with using compulsory licenses, including for advantages reduced price, development of domestic manufacturing capacity, and "leverage" for negotiating with rightsholders. The Report recognizes the limitation (for now) that compulsory licenses involve solely patents and do not grant access to know-how and trade secrets (which the Report notes can be particularly important for biologic drugs). The Report acknowledges that compulsory licenses can be granted in the context of lack of availability of a qualified manufacturer and result in economic and political pressure and legal challenges under domestic law or TRIPS (although the latter may be something of a paper tiger in the current political climate).
The Report particularly acknowledges issues in "least developed countries" (LDC) as classified by the United Nations. Bangladesh, Laos, Myanmar, and Paraguay are provided as exemplars for taking advantage of the exceptions to TRIPS requirements related to IP, which in the COVID context has been used as a pretext for producing generic versions of nirmatrelvir (+ ritonavir), remdesivir, and baricitinib and exporting these drugs to countries without patent protection for them.
With regard to the availability and consumption of COVID-19 diagnostics and therapeutics, the Report assesses both parameters through the lens of price, actual purchase, donations, and consumption of diagnostics and therapeutics, as illustrated in this schematic for therapeutics:

For diagnostics, the data reviewed (albeit subject to inconsistencies in different countries, from different manufacturers, and different times) indicate government procurement and distribution of test kits (in the U.S. pledged to provide $1 billion in kits free of charge). Multilateral organizations made tests broadly available to middle- and low-income countries during the pandemic, which have reduced substantially by mid-2023, as the pandemic seems to have waned and WHO has ended the public health emergency.
For therapeutics, the Report found very little price transparency but notes a tiered-pricing regime depending on income level, with low-income countries paying substantially less. Except for high income countries like the U.S. and Europe COVID therapeutics are obtained/procured through donations or purchase from governments (79% of 98 million treatment courses) and multinational organizations. Pfizer, Eli Lilly, and Merck have donated COVID therapeutics to low- and middle-income countries. Consistent with these data the Report states that "many consider the prices of COVID-19 therapeutics to be a significant barrier to access for many" countries (other than high income countries, of course), illustrating the challenge of therapeutic cost with Pfizer's nirmatrelvir (+ ritonavir) treatment, which at $250 per treatment course is about half of the average per capita healthcare expenditure across all upper-middle income countries. Coupled with this disincentive the waning of the pandemic has caused a deep reduction in demand for these drugs. Even when demand was higher regulatory approvals in each country caused delays, and many countries have pre-existing public health demands (HIV/AIDS, malaria, and tuberculosis are given as examples) that limited availability of COVID-19 drugs.
The Report next sets forth synopses of the views of "interested persons" on "(1) IP protection, R&D, and jobs; (2) the TRIPS Agreement and access to medicine; and (3) the TRIPS Agreement and COVID-19 diagnostics and therapeutics" obtained for eight topics in each of these categories. Not unexpectedly, the Report asserts that while some respondents attested to the beneficial effect of IP on development of technologies critical to addressing COVID-19 and future pandemics, others expressed the belief that extending the 2022 Ministerial Decision would not have a deleterious effect on the supply of diagnostics or therapeutics. Likewise respondents could be split into those who believe the IP protections in TRIPS facilitated access to medicines (including COVID-related diagnostics and therapeutics by incentivizing investment) while others thought the purported increased cost had the opposite effect. The pros and cons of compulsory licensing were also addressed and some respondents asserted that further clarification on the "flexibilities" in IP protection would be a useful pathway towards improving access to medicines worldwide, particularly in low- and middle-income countries. Respondents were in agreement, however, that "waiving or weakening IP rights on COVID-19 diagnostics and therapeutics would reduce incentives to invest in the R&D of tests and treatments for future pandemics." Nevertheless, respondents split reliably on lines wherein some advocated that IP protection was necessary to be prepare for future pandemic (providing incentives and financial rewards for innovation) while others stated that the 2022 Ministerial Decision should be extended to encompass future drug development. The former respondent group warned that "an extension of the 2022 Ministerial Decision to COVID-19 diagnostics and therapeutics would cover an overly broad set of diagnostics and therapeutics and reduce incentives for companies to look for COVID-19 applications for their products." Respondents also submitted statistics on the effect of the Decision on employment in 26 countries (including the U.S.) but these statistics included jobs unrelated to COVID-19 diagnostics and therapeutics and thus were of limited value.
At the request of the Trade Representative the Report contains a "literature review" on four topics: innovation in the health sector and access to medicines in relation to patent protection; the effects of compulsory licensing; and the effects of the Medicines Patent Pool (this topic having received limited academic study that focused in the main on HIV/AIDS drugs). These reviews found little consensus in the current academic literature on these topics, the Report characterizing them as "limited" and that would "benefit from additional research on these topics," due in part to "limited data availability" and "difficulty identifying the causal effect" of IP on these topics.
The remainder of the Report comprises the following sections.
Chapter 1: Introduction: explaining that the Report was in response to a letter dated December 16, 2022 from the U.S. Trade Representative asking the ITC to perform the study in response, in part, to the June 17, 2022 Ministerial Decision providing a waiver of certain TRIPS protections for intellectual property in response to a joint communique from India and South Africa. The scope and intention of the December 2022 letter indicated that the ITC's Report would be used by the U.S. before the WTO in its deliberations regarding whether to extend the IP waiver to include COVID-related diagnostics and therapeutics. In making these determinations, economies for WTO members were set forth according to this schema:

(where HIC = high-income countries; UMIC = upper-middle-income countries; LMIC = lower-middle-income countries; LIC = low-income countries). This section provides cumulative information that as of August 2023 the COVID pandemic had produced 769 million confirmed cases and almost 7 million deaths.
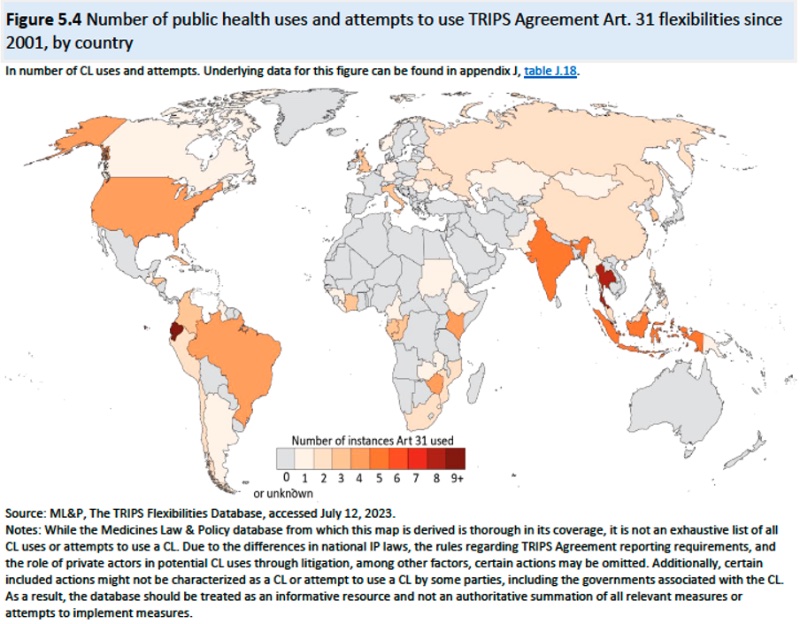
Chapter 2: Background on IP and Regulations: providing background information on TRIPS and mandated IP protections required thereby. Notably, this section discloses that the "flexibilities" regarding IP protection of TRIPS Articles 31 and 31bis and the Doha Declaration (2001) have resulted in "172 instances (122 compulsory licenses; 46 extended transition implementation period until 2033 for LDCs; 3 exceptions to patent rights under TRIPS Article 30; and 1 instance of parallel importation) in which governments used or attempted to use TRIPS Agreement flexibilities for public health purposes as of May 2023. And modifications to the Doha declaration in 2003 permitted compulsory licenses to extend to production for export as well as domestic applications. This Chapter also sets forth the particulars of the 2022 Ministerial Declaration (in particular mentioning that the original proposal included waiver of "undisclosed information" meaning trade secrets), in apparent contradiction to the provisions of TRIPS Article 39.3. Finally in this regard, this Chapter sets forth information from both those in favor and against the 2022 Ministerial Declaration that IP rights did little or nothing to inhibit diagnostic testing for COVID-19 during the pandemic, due to expiration of most applicable IP.
The Report also discloses landscape searches performed by WIPO and the USPTO (824 COVID-19 diagnostic-specific published patent filings at the USPTO between December 2019 and the end of March 2023, in which 47% of relevant patent families were filed in China) directed to diagnostics. On the other hand, WIPO reported 4,787 applications filed between January 2020 and September 2022 directed to COVID-19 therapeutics.
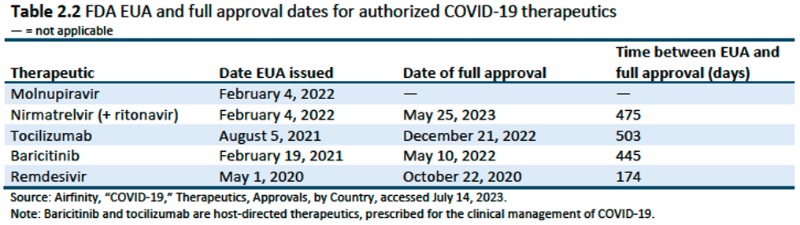
This Chapter concludes with an overview of the drug regulatory landscape worldwide (in each individual country and WHO) and the status of regulatory approval for COVID-19 related diagnostics and therapeutics. Five COVID therapeutics have been granted prequalification by WHO: tocilizumab, dexamethasone, molnupiravir, remdesivir, and nirmatrelvir (+ ritonavir), and FDA has authorized 278 molecular diagnostic tests and 64 antigen tests under EUAs as of July 2023.

Chapter 3: Definitions and the Universe or COVID-19 Diagnostics and Therapeutics (vaccines were not included in the Trade Representative's Request letter): specifically sets forth "the range of definitions for diagnostics and therapeutics in the medical field; (2) identifies and defines the universe of existing COVID-19 diagnostics and therapeutics covered by patents, as well as diagnostics and therapeutics in development; and (3) provides a broad overview of relevant COVID-19 diagnostics and therapeutics," acknowledging that these definitions are more complex than identifying vaccines, at least with regard to functional properties. These data are illustrated in several tables in the Chapter.


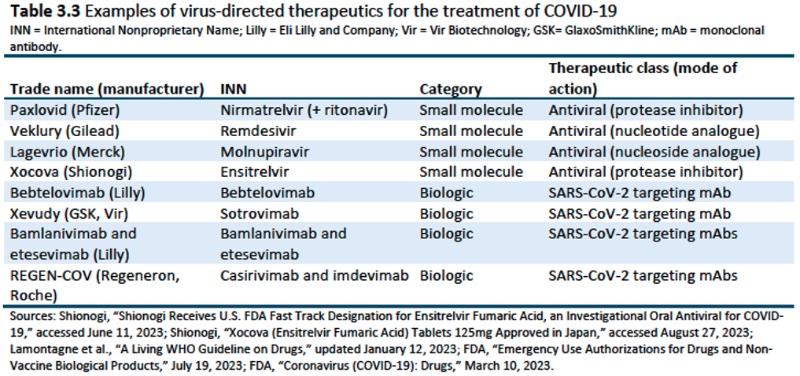

Chapter 4: COVID-19 Diagnostics and Therapeutics Manufacturing Supply Chain and Trade: provides information "describing the development, production, and trade of COVID-19 diagnostics and therapeutics; discussing the regulations needed to bring these goods to the global market; and providing an overview of the key producing countries and firms operating in the supply chain." Diagnostics were limited to PCR and rapid antigen tests, while therapeutics were limited to virus-directed drugs on the market during the pandemic (January 2020 to July 2023), including both small molecule and biologic drugs. The Report acknowledges the limitations in the data for a variety of reasons.
Chapter 5: Approaches to Access the Intellectual Property Associated with COVID-19 Diagnostics and Therapeutics: This Chapter is focused more on therapeutics than diagnostics and addresses ways to accesses COVID-related IP, including "research and development (R&D) agreements, manufacturing partnerships, bilateral license agreements (BLAs) between firms, and license agreements involving the Medicines Patent Pool (MPP), a United Nations (UN)-backed public health organization" established in 2010 (these agreements comprising voluntary licensing). The consequences and benefits ("advantages and challenges") of compulsory licenses are also discussed. Statistics provided include that "87 percent of COVID-19 therapeutic R&D programs in the United States, and 75 percent of products in the global clinical development pipeline, originate from U.S.-based small- and medium-sized enterprises (SMEs)," according to the Biotechnology Innovation Organization (BIO). Also mentioned is the involvement of the Federal government in producing COVID-related therapeutics. Specific BLA agreements (to the extent they are public) discussed include Gilead/remdesivir, Merck/molnupiravir, and Eli Lilly/baricitinib, particularly as a means to avoid compulsory licenses and regulatory delays. Finally, the Chapter contains an extensive discussion of the advantages of using the MPP for voluntary licensing, stating:
Rather than negotiating individual license agreements directly with producers, licensors authorize the MPP to sublicense their technologies to third parties. In turn, the MPP solicits applications for sublicense agreements, reviews prospective producers, and handles all aspects of implementation of the sublicense agreements with selected companies.
(although conceding that academic research on the MPP is limited and what exists is focused on HIV/AIDS drugs). Merck is noted as the first company to enter into an MPP license in October, 2021 for molnupiravir (having the advantage that sublicenses come with a regulatory waiver), followed by Pfizer for nirmatrelvir (+ ritonavir) in November 2021, and Shionogi for ensitrelvir in October 2022. Examples of these MMP licenses are set forth in the following table:
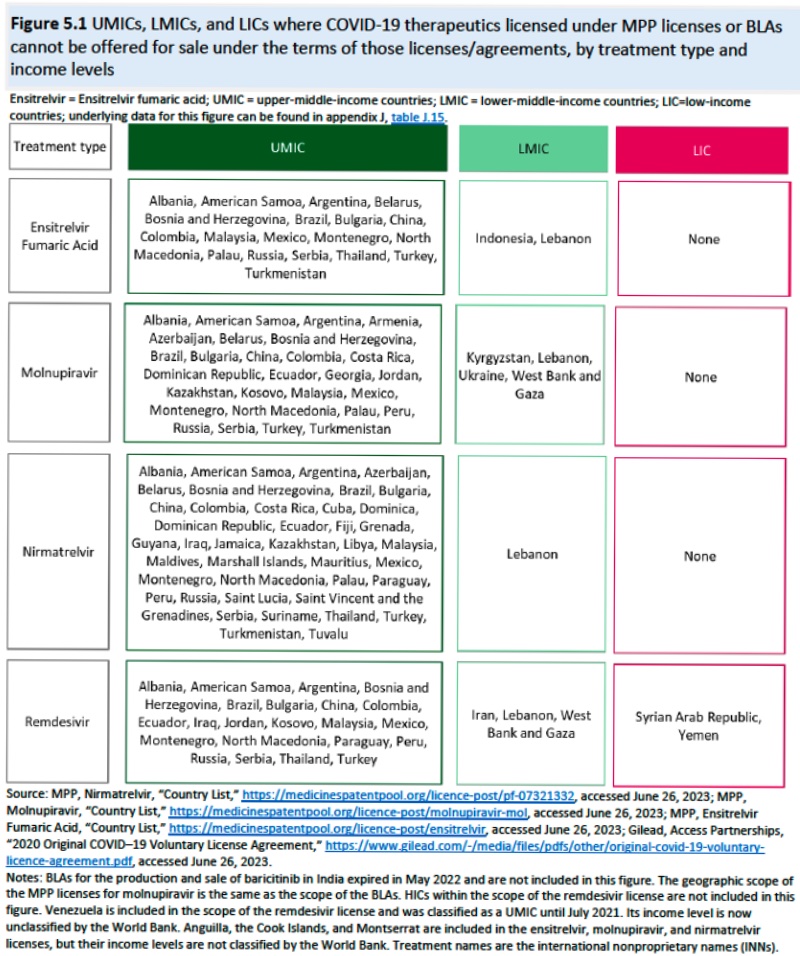
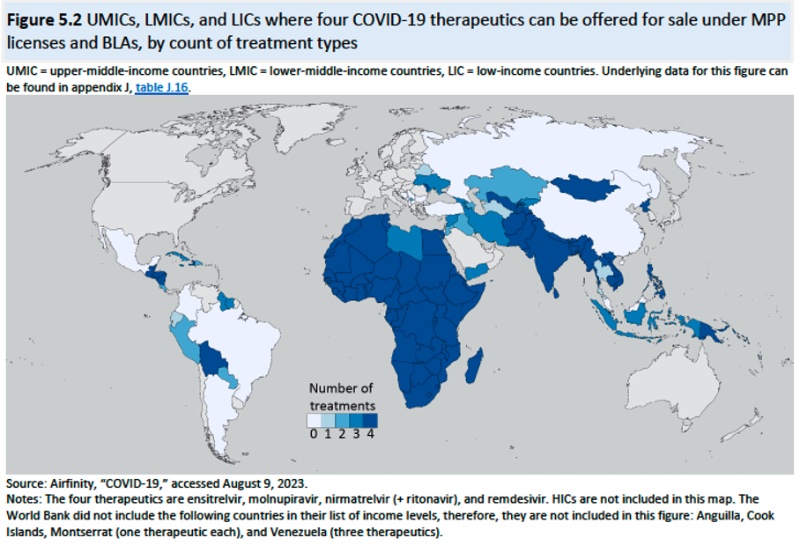
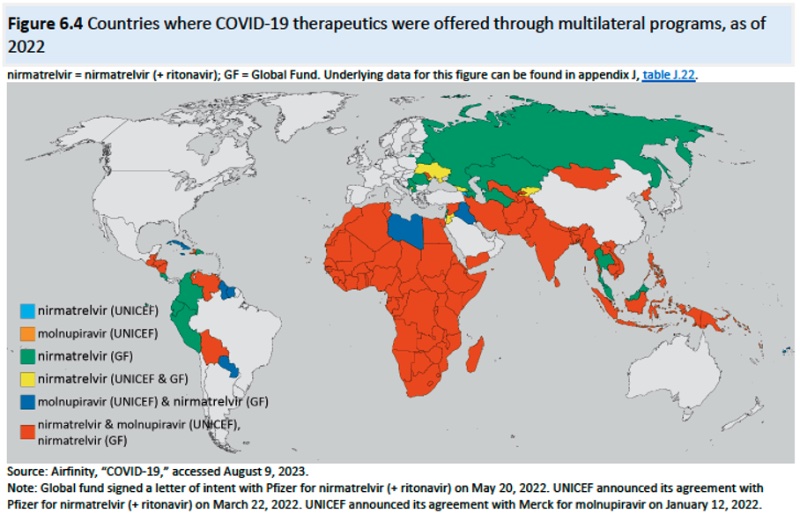
And the geographic distribution of MPP licenses shown in this diagram:
The Report also describes compulsory licenses which have not been used with COVID vaccines or diagnostics but have been imposed on COVID therapeutics. According to the Report, there have been approximately 121 attempts to issue a public health compulsory license (including 4 by the U.S.) since 2001, but only about 75% of threatened compulsory licenses were imposed, as illustrated by this diagram:
Advantages of such licenses include lowering drug prices, providing leverage in negotiations, and in some instances improving domestic manufacturing capacity. Of course, compulsory licensing has also resulted in retaliation against countries imposing them, including reducing investment or withholding drug products from the national market. Compulsory licenses are also imperfect in having the desired effect because they are limited to patents and cannot expropriate know-how necessary to produce a drug product. Specific instances of compulsory licensing discussed in the Report include Zambia, India, Brazil, Ecuador, Malaysia, South Africa, Thailand, and Canada (in this instance for importing HIV/AIDS drugs to Rwanda). And the effects of compulsory licenses on innovation are disputed by researchers according to the Report.
Chapter 6: Availability and Consumption of COVID-19 Diagnostics and Therapeutics: This Chapter provides the Trade Representative with information regarding the necessity (or lack of it) of a COVID waiver extension based on global needs. This discussion is presented before the backdrop of fundamental disagreements between proponents and opponents of the waiver over the causes and even the existence of global deficiencies in providing COVID-19 diagnostics and therapeutics, arguing insufficient demand or insufficient (affordable) supply, with evidence on both sides of the debate. Needs assessments are limited almost entirely to therapeutics, there being no analogous studies or reports for diagnostics. The Report provides statistics relating to prices, purchases (including governmental purchases and multilateral programs and private sector purchases), donations, and consumption for diagnostics and therapeutics as well as factors affecting each of these statistics (such as "last mile delivery" effects). Regulatory authorizations (and needs therefor) as well as the effects of competing healthcare priorities in many countries is also discussed in the Report.
Chapter 7: Views of Interested Persons: This Chapter provides synopses and examples of public input requested by the Trade Representative. Providing a cautionary warning the Report states that "[t]he summaries of public views provided in this chapter do not attempt to assess, analyze, or draw conclusions about these views" and that "this chapter often uses language as provided by participants to avoid changing the meaning of their submitted views" and thus "some wording used in this chapter may not match wording used in other chapters of this report." The structure of the Report in this chapter is as follows:
IP protection, R&D, and jobs
• The relationship between IP protection and corporate R&D expenditures, taking into account other expenditures, such as share buybacks, dividends, and marketing
• The location of jobs associated with the manufacturing of diagnostics and therapeutics, including in the United States
The TRIPS Agreement and access to medicine
• Whether and how existing TRIPS rules and flexibilities can be deployed to improve access to medicines
• Successes and challenges in using existing TRIPS flexibilities
• To what extent further clarifications of existing TRIPS flexibilities would be useful in improving access to medicines
The TRIPS Agreement and COVID-19 diagnostics and therapeutics
• How the TRIPS Agreement promotes innovation in and/or limits access to COVID-19 diagnostics and therapeutics
• The extent to which products not yet on the market, or new uses for existing products, could be affected by an extension of the Ministerial Decision to COVID-19 diagnostics and therapeutics
• The relevance, if any, of the fact that diagnostic and therapeutic products used with respect to COVID-19 may also have application to other diseases
Appendix D of the Report contains the positions and policy statements of witnesses at the public hearings
The Report is a wealth of information and data (albeit wherein some of the data is incomplete) and abides by the request by the Trade Representative not to espouse any particular course of action or outcome. Accordingly, the Report can be expected to be a useful tool during negotiations. It is the reality, however, that whatever course the Administration takes will, in the absence of fact- or data-driven outcomes predominantly be a political decision based more of the concessions that can be obtained than any demonstrated need for any extension of the IP waiver from these data. Should increasing the waiver come to pass the consequences are unlikely to be immediately felt, unless another virulent COVID-19 variant arises that cause higher morbidity and mortality than the current crop of variant COVID-19 species has developed. The greater risk is in the precedent that a widened waiver would set and whether such a waiver would in the next pandemic reduce the capability for the type of rapid development of vaccines, diagnostics, and therapeutics that have (at least for now) stemmed the tide of the COVID-19 pandemic to manageable levels. That is an outcome that will only become apparent through experience, and such an experience may be very costly indeed and completely unpredictably so. Prudence suggests it would be wise to court such an outcome carefully.
For additional information regarding this topic, please see:
• "Top Four Stories of 2022," January 18, 2023
• "WTO TRIPS Council Recommends That General Council Extend Waiver Deadline," December 18, 2022
• "Nine Countries Seek Extension of WTO Waiver to COVID-19 Therapeutics and Diagnostics," December 11, 2022
• "Status of Proposed Extension of TRIPS Waiver in WTO," December 8, 2022
• "C4IP Presents Webinar on COVID Waiver Extension," December 5, 2022
• "Senators Send Letter to Commerce Secretary Regarding WTO Waiver Compromise," March 28, 2022
• "The Proposed WTO IP Waiver: Just What Good Can It Do? — An Analysis," March 24, 2022
• "IP Associations "Concerned" by Reports of TRIPS Waiver Compromise," March 24, 2022
• "More on Leaked WTO COVID-19 Vaccine Patent Waiver Compromise," March 21, 2022
• "Compromise Reportedly Reached on COVID-19 Vaccine Patent Waiver," March 16, 2022
• "Sen. Tillis Writes to U.S. Trade Representative (Again) Regarding TRIPS Waiver," December 12, 2021
• "U.S. Trade Representative Responds to Letters from Senators Regarding TRIPS Waiver," November 14, 2021
• "U.S. Chamber of Commerce Urges Administration to 'Double Down' on Global Vaccine Distribution," November 3, 2021
• "Is This the WTO Waiver End Game?" July 25, 2021
• "BIO Declaration on Global Access to COVID-19 Vaccines and Treatments and Role of IP," June 24, 2021
• "GOP Legislators Write in Opposition to Proposed TRIPS Waiver," May 16, 2021
• "Population of Patents at Risk from Proposed WTO Patent Waiver," May 12, 2021
• "Sen. Daines Urges Biden Administration to Withdraw Support for COVID-19 IP Waiver," May 12, 2021
• "Pfizer CEO Pens Open Letter on COVID-19 Vaccine IP Waiver," May 10, 2021
• "If the Devil of the WTO IP Waiver Is in the Details, What Are the Details?" May 9, 2021
• "The Road to Hell Is Paved with What Everybody Knows," May 6, 2021
• "BIO & IPO Issue Statements on Biden Administration's Support for Proposed WTO Waiver," May 6, 2021
• "Biden Administration Supports Waiver of IP Protection for COVID-19 Vaccines," May 5, 2021
• "Suspending IP Protection: A Bad Idea (That Won't Achieve Its Desired Goals)," April 26, 2021
• "Sen. Tillis Asks Biden Administration to Oppose WTO Waiver Proposal," April 21, 2021
• "IP Organizations Support Continued Opposition to Waiver Proposal," April 5, 2021
• "Industry Coalition Supports Continued Efforts to Oppose Waiver Proposal," March 29, 2021
• "BIO and PhRMA Urge Biden Administration to Oppose Proposed WTO TRIPS Waiver," March 11, 2021
• "IPO Sends Letter on IP Law and Policy to President-Elect and Vice President-Elect," January 4, 2021
 Proper construction of claim limitations reciting the chemical property of pH (which denotes the concentration of hydrogen ions in a solution as an indication of acidity) has arisen several times in district court and Federal Circuit opinions, perhaps the most notable being in Warner-Jenkinson v. Hilton Davis Chemical (1997), which revitalized the doctrine of equivalents. The issue arose recently before the Federal Circuit in Actelion Pharmaceuticals Ltd. v. Mylan Pharmaceuticals Inc., with the Court vacating and remanding the District Court's claim construction for failure to consider extrinsic evidence. The opinion illustrates the difficulties posed and considerations considered by the Court when addressing claim constriction for terms comprising pH.
Proper construction of claim limitations reciting the chemical property of pH (which denotes the concentration of hydrogen ions in a solution as an indication of acidity) has arisen several times in district court and Federal Circuit opinions, perhaps the most notable being in Warner-Jenkinson v. Hilton Davis Chemical (1997), which revitalized the doctrine of equivalents. The issue arose recently before the Federal Circuit in Actelion Pharmaceuticals Ltd. v. Mylan Pharmaceuticals Inc., with the Court vacating and remanding the District Court's claim construction for failure to consider extrinsic evidence. The opinion illustrates the difficulties posed and considerations considered by the Court when addressing claim constriction for terms comprising pH.