By Kevin E. Noonan –
 Last week, the Federal Circuit handed down a pair of non-precedential decisions affirming the Patent Trial and Appeal Board (PTAB) in inter partes review (IPR) proceedings. This post concerns the decision in Cardiovalve Ltd. v. Edwards Lifesciences Corp., which has as its most significant aspect an unnecessary untethering of obviousness with evidence regarding the skill and inventiveness of the person of ordinary skill in the art under KSR International Co. v. Teleflex Inc.
Last week, the Federal Circuit handed down a pair of non-precedential decisions affirming the Patent Trial and Appeal Board (PTAB) in inter partes review (IPR) proceedings. This post concerns the decision in Cardiovalve Ltd. v. Edwards Lifesciences Corp., which has as its most significant aspect an unnecessary untethering of obviousness with evidence regarding the skill and inventiveness of the person of ordinary skill in the art under KSR International Co. v. Teleflex Inc.
The case arose with Edwards's challenge of claims 1-3, 5, 6, and 8-11 of Cardiovalve's U.S. Patent No. 10,226,341 directed to an apparatus for implanting a prosthetic heart valve in a patient in need thereof. Claim 1 was considered representative by the panel:
1. Apparatus for use at a native valve of a subject, the native valve including at least a first native leaflet and a second native leaflet, the apparatus comprising:
an implant, comprising:
an annular portion, being configured to be placed against an upstream side of the native valve, and having an inner perimeter that defines an opening, and
at least one leaflet clip:
(i) coupled to the annular portion,
(ii) comprising:
at least two clip arms, movable with respect to each other to open and close the clip; and a clip-controller interface, the clip-controller interface being coupled to at least one of the clip arms, and
(iii) configured:
to be coupled to a portion of the first native leaflet by the clip arms being brought together to close around the first native leaflet,
to be coupled to a portion of the second native leaflet by the clip arms being brought together to close around the second native leaflet, and
to hold together the portion of the first leaflet and the portion of the second leaflet; and
a delivery apparatus, configured to deliver the implant to the native valve, and comprising at least one clip controller, the at least one clip controller being reversibly couplable to the clip-controller interface, and configured to facilitate opening and closing of the clip, and the delivery apparatus being intracorporeally decouplable from the implant.
wherein the bold, italicized language is relevant to the issues before the Court.
The prior art cited in the IPR petition, U.S. Patent No. 7,635,329, in support of Edwards's obviousness contentions, disclosed a device for stabilizing valve leaflets illustrated in Figure 9B of this patent:
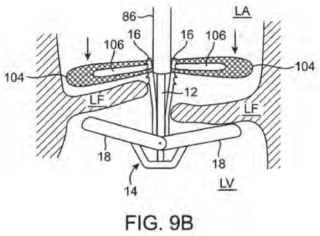
where the "device, 14, [that] includes two proximal elements, 16, and two distal elements, 18, [is] configured such that a proximal and distal element pair, when brought together, form a clip that grasps a heart valve leaflet, LF, from the top and bottom [and wherein the] fixation device also includes flaps, 104, which restrict upward motion of the leaflets to better enable the proximal and distal elements to grasp the leaflets." Also noted in the opinion is that "[o]nce the leaflets have been grasped, the flaps . . . may be removed . . . or may be left behind to assist in holding the leaflets" (emphasis in opinion). Before the Board, Cardiovalve conceded that all the limitations recited in its claims were disclosed in the '329 patent except that the device comprise "at least one leaflet clip" "coupled to the annular portion" of the device. The Board found that the pairs of proximal and distal elements disclosed in the '329 patent together constitute a "leaflet clip" as disclosed and claimed in the '341 patent, that the flaps "constitute or contain" an "annular portion, and further that any direct or indirect attachment is a "coupling." Based on these conclusions, the Board found the challenged claims of the '341 to be obvious, and this appeal followed.
The Federal Circuit affirmed in a decision by Judge Taranto joined by Judges Chen and Stoll. Cardiovalve's sole argument on appeal was that the Board erred in concluding that it would have been obvious to attach, "either directly or indirectly, [the flaps] disclosed in the '329 patent" to its proximal and distal elements." The opinion notes that the Board applied the seminal teaching from KSR that:
When there is a design need or market pressure to solve a problem and there are a finite number of identified, predictable solutions, a [relevant artisan] has good reason to pursue the known options within his or her technical grasp. If this leads to the anticipated success, it is likely the product not of innovation but of ordinary skill and common sense. In that instance the fact that a combination was obvious to try might show that it was obvious under § 103.
The panel held that the Board had substantial evidence for its conclusion that Edwards satisfied these requirements. The panel's reasoning was that the disclosure in the '329 patent that the flaps could be "left behind" necessitated that they be attached to something fixed either to the device disclosed therein or to heart tissue, which provided a "finite number of predictable options." The Board also relied on Edwards's expert witness regarding why the skilled artisan would know to attach the flaps to the device and why such an attachment would be expected to succeed, thus providing the reasonable expectation of success required by KSR (something Cardiovalve did not challenge). And the fact that there might have been a small number of other alternatives, as argued by Cardiovalve, did not rebut the Board's determination that the one consistent with the invention claimed in the '341 patent was obvious.
The one interesting aspect of the decision, and one that has garnered comment, was the panel's rejection of Cardiovalve's argument that recitation of the "known options" in the cited passage of the KSR opinion should be limited to what was actually known in the prior art. (Indeed, it was the genius of Giles Rich and PJ Federico to tether obviousness under Section 103 to the prior art in ways that a century of Supreme Court precedent never did, leaving a determination of invalidity under what philosophically was obvious to the "flash of genius" and other inchoate and unpredictable standards.) The Federal Circuit's basis for negating this requirement on the prior art was that the requirement was "nowhere articulated in KSR, and adopting it would be inconsistent with the Supreme Court's rejection in KSR of a rigid demand for the steps in an obviousness analysis to be proved by prior-art documents, to the exclusion of the knowledge and skill of the relevant artisan." And the Court refused to recognize such a requirement in its own precedent, abjuring such existing precedent cited by Cardiovalve (specifically, Uber Technologies, Inc. v. X One, Inc., 957 F.3d 1334, 1339–40 (Fed. Cir. 2020); and Bayer Schering Pharma AG v. Barr Laboratories, Inc., 575 F.3d 1341, 1350 (Fed. Cir. 2009); indeed, this reasoning smacks not a little of being equivalent to stating that just because the Court had reasoned correctly in the past did not necessitate continuing to do so). Of course there is nothing in KSR's averments regarding the basis for making an obviousness determination based on the knowledge and skill of the prior art artisan that does away with the requirement for evidence that the alternatives relied upon to make that determination would have been understood by the worker of ordinary skill at the time the invention was made and not simply presumed to have existed (being the epitome of hindsight, which KSR also warns against).
The remainder of the panel's reasoning for its decision merely begs the question, i.e., assumes the conclusion, based on Edwards's argument that "a relevant artisan would have found it obvious to attach Goldfarb's flaps to its fixation device (and therefore indirectly to its proximal and distal elements), because the flaps, if left behind, had to be attached somewhere and this was an obvious location." In the Court's application of the cited portion of the KSR decision the opinion states that "when a relevant artisan would have recognized a problem, a consideration of whether only a small number of solutions existed is a natural part of an evaluation of whether a patent-claimed solution would have been obvious. The Board's undertaking such consideration here was therefore a legitimate exercise of its duty to evaluate whether Edwards was right or wrong about the position Edwards consistently maintained—that attachment to the implant would have been obvious" (the relevance of the italicized language being itself questionable). The questionable nature of this basis for affirming the Board's decision is even more unfortunate because the opinion contains an ample basis for affirmance, whereby "the Board actually found reasons for a relevant artisan to choose the Edwards-urged solution over the alternative(s)—at the least, greater simplicity," reliance on which would have avoided disturbing dicta in what was otherwise (and fortunately) just another non-precedential opinion by the Court.
Cardiovalve Ltd. v. Edwards Lifesciences Corp. (Fed. Cir. 2024)
Nonprecedential disposition
Panel: Circuit Judges Taranto, Chen, and Stoll
Opinion by Circuit Judge Taranto