By Kevin E. Noonan –
One of the characteristics of patent infringement litigation in the aftermath of the Supreme Court's decision in Markman v. Westview Instruments, Inc. (holding that claim construction was a matter of law to be reviewed de novo by the Federal Circuit; but see Teva Pharma. USA, Inc. v. Sandoz, Inc.) was that an inordinate proportion of claim construction decisions were overturned in whole or part. Those proclivities have been attenuated in the years following the Cybor v. FAS Technologies* opinion, but the Court's recent decision in Malvern Panalytical Inc. v. TA Instruments-Waters LLC is reminiscent of those bygone days of Federal Circuit preeminence.
The case arose in patent infringement litigation when Malvern sued Waters for infringement of U.S. Patent Nos. 8,827,549 and 8,827,549. The claims of each patent asserted by Malvern were directed to isothermal titration microcalorimeters, used to measure energy absorbed or released during chemical reactions between two compounds. The claimed microcalorimeters comprise an automatic pipette assembly 220 illustrated in the opinion by this diagram:
 The limitation at issue was another component, the pipette guiding mechanism that guides the pipette assembly "between and into at least two positions of operation." Disclosed are two embodiments, one being "a pipette arm 520 that supports the pipette assembly 220, and an essentially vertical guide rod 530," illustrated by this Figure 5a:
The limitation at issue was another component, the pipette guiding mechanism that guides the pipette assembly "between and into at least two positions of operation." Disclosed are two embodiments, one being "a pipette arm 520 that supports the pipette assembly 220, and an essentially vertical guide rod 530," illustrated by this Figure 5a:
The operation of this structure is described in the specification to be:
"The pipette arm 520 is moveably attached by a sleeve 540 to the guide rod 530, but its motion about the guide rod is restricted by a guide grove 550 in the guide rod 530 and a guide pin 560 that protrudes from the inner surface of the sleeve 540 and which fits into the guide groove 550" wherein the "movement of the pipette assembly 220 in the vertical direction is restricted to the angular positions of the positions of operation," and "rotational movement of the pipette assembly 220 between the angular positions only is permitted when the titration needle 260 is fully retracted from respective positions of operation."
The second embodiment of this claimed element is illustrated by this Figure 6:
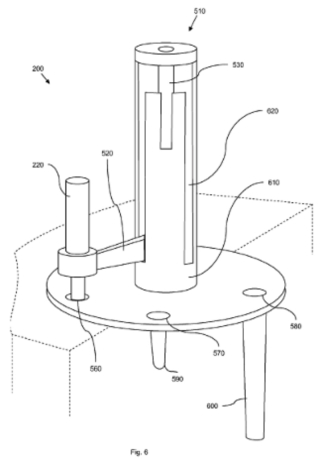
In this embodiment, the guide groove in the guide rod 530 is replaced by a coaxial external guide sleeve 610 with corresponding guide paths 620 for the guide arm 520."
Claim 1 of the '549 patent and claim 9 of the '175 patent are set forth as representative:
1. A micro titration calorimetry system comprising:
an automatic pipette assembly comprising a titration needle arranged to be inserted into a sample cell for supplying titrant, a syringe for supplying titrant to the titration needle, a stirring paddle for stirring fluid in the sample cell, and a stirring motor for rotating the stirring paddle, and
a pipette guiding mechanism arranged to restrict the movement of the pipette assembly along safe paths to ensure that the titration needle cannot be damaged during movement thereof between different positions of operation.
9. A micro titration calorimetry system comprising:
an automatic pipette assembly comprising a titration needle arranged to be inserted into a sample cell for supplying titrant, a syringe for supplying titrant to the titration needle, a stirring paddle for stirring fluid in the sample cell, and a stirring motor
for rotating the stirring paddle, and
a pipette guiding mechanism arranged to guide the pipette assembly between and into at least two positions of operation, wherein a first position of operation is a pipette washing position wherein the titration needle is inserted in a washing apparatus, and a second position of operation is a titration position wherein the titration needle is inserted into the sample cell for calorimetric measurements.
The opinion states as being "relevant to this appeal" prosecution of an unrelated patent, U.S. Patent No. 9,103,782 owned by Malvern. In that case, applicants were able to overcome an anticipation rejection over U.S. Patent Application Publication No. 2010/0238968 based on common ownership. Also relevant was a supplemental examination request under 35 U.S.C. § 257 that Malvern filed upon acquisition of the '175 patent. As part of that effort, Malvern filed an Information Disclosure Statement including references cited against the '782 patent, including the '968 application (although as noted in the opinion Malvern did not characterize the references). During supplemental examination, the Examiner cited an owner's manual for a prior art microcalorimeter disclosing a manually operated pipette guiding mechanism, which Malvern overcame by affidavit that this manual was the inventors' own work.
Turning to claim construction, the opinion recites the parties' proposed constructions of the term "pipette guiding mechanism," wherein Malvern argued the term meant a "mechanism that guides the pipette assembly," while Waters argued that it should mean a "mechanism that manually guides the pipette assembly"; the District Court adopted Waters' construction. In ruling on this decision, the District Court characterized the phrase "pipette guiding mechanism" to be a "coined term" that required an examination of the intrinsic evidence to determine the "objective boundaries to the scope of the term," citing Iridescent Networks, Inc. v. AT&T Mobility, LLC, 933 F.3d 1345, 1353 (Fed. Cir. 2019). In doing so, the District Court relied on statements on the meaning of the term made during prosecution of the '782 patent and that applicants had limited the meaning of the term to manual embodiments. These arguments were attributed to Malvern as the common assignee of the '782, '549, and '175 patents and because statements made during prosecution of the '782 patent were incorporated into the intrinsic record during supplemental examination. The parties consented to a stipulated judgment of infringement based on the disputed claim construction and this appeal followed.
The Federal Circuit vacated the stipulated judgment in an opinion by Judge Prost, joined by Judges Hughes and Cunningham. The panel agreed with Malvern that the term "pipette guiding mechanism" encompassed both manual and automated embodiments based on the intrinsic evidence, specifically the plain meaning of the claims, the specification, and the prosecution history. Regarding the claim language, the panel considered the plain meaning of the words individually, citing Littelfuse, Inc. v. Mersen USA EP Corp., 29 F.4th 1376, 1381 (Fed. Cir. 2022), and 3M Innovative Props. Co. v. Tredegar Corp., 725 F.3d 1315, 1329 (Fed. Cir. 2013), and found no basis for limiting the pipette guide mechanism to only manual embodiments ("Instead, the broad claim language supports the conclusion that the 'pipette guiding mechanism' encompasses both manual and automatic embodiments.") Interpreting the claim broadly is consistent, in the panel's view, with how the Court construed the claims in Hill-Rom Services, Inc. v. Stryker Corp., 755 F.3d 1367 (Fed. Cir. 2014), wherein broad claim language supported broad claim interpretation. And the Court found that the remaining language of the claims similarly supported this interpretation. According to the opinion, while "[t]he claims clarify and restrict what the guiding mechanism does, . . . they provide no language suggesting the restriction to manual embodiments Waters advocates."
The specification supports this interpretation according to the Court, based on disclosure of two embodiments neither of which is limited to manual pipette guiding mechanisms. "In fact," the Court says, "the specification states nothing about whether the guiding mechanism is manual or automatic," and cites Hill-Rom Services, Inc. v. Stryker Corp., 755 F.3d 1367, 1373 (Fed. Cir. 2014), for the principle that "[t]here are no magic words that must be used, but to deviate from the plain and ordinary meaning of a claim term to one of skill in the art, the patentee must, with some language, indicate a clear intent to do so in the patent" and, as in Hill-Rom, "there is no such language here." The Court dismissed Waters' arguments to the contrary because they were based on portions of the disclosure that "say nothing about whether the guiding mechanism is manually or automatically operated." Nor was the Court persuaded by the fact that other components were described as automatic (and thus not describing the pipette guiding mechanism as being automatic implied the term should be limited to manual embodiments).
Finally, regarding the prosecution history, the Court was not persuaded to Waters' construction by the supplemental examination of the '175 patent, wherein the inventors testified that a manual for a prior art microcalorimeter comprising a manual pipette guide mechanism was their own work and thus not prior art. Waters argued that this testimony should be interpreted to mean the claimed microcalorimeters of the '175 patent were themselves limited to manual embodiments of the pipette guide mechanisms. (In a footnote the panel explains that they found the statements to be "too vague or ambiguous to qualify as a disavowal of claim scope.")
Turning to the "coined term" issue which provided one basis for the district court's decision, the opinion notes the District Court's reliance on Indacon, Inc. v. Facebook, Inc., 824 F.3d 1352, 1357–58 (Fed. Cir. 2016), in construing the claims. While recognizing that their Indacon decision involved the question of construing a claim that did not have an established meaning in the art, the panel maintained that what the District Court had needed to do was "answer the question of what plain and ordinary meaning a term has in the context of a patent," which the District Court here did not do, in the Federal Circuit's opinion.
Finally, the panel considered what weight (if any) to give the prosecution history of the '782 patent. The Court concluded that the '782 prosecution was not relevant to claim construction of the '175 and '549 patents-in-suit merely because that history had been submitted in an IDS during the supplemental examination of the '175 patent. The Court relied on its precedent in Goldenberg v. Cytogen, Inc., 373 F.3d 1158, 1167 (Fed. Cir. 2004), that "[i]n the absence of an incorporation into the intrinsic evidence, this court's precedent takes a narrow view on when a related patent or its prosecution history is available to construe the claims of a patent at issue and draws a distinct line between patents that have a familial relationship and those that do not." And the opinion further asserts that citing the prosecution history in an IDS is at most an admission that "'references in the disclosure may be material to prosecution of the pending claims,' but [] "does not admit materiality," citing Abbott Labs. v. Baxter Pharm. Prods., Inc., 334 F.3d 1274, 1279 (Fed. Cir. 2003) (emphasis added). The Court concluded here that there was no such admission by Malvern's "bare listing" of the IDS. And even if considered to be so, the opinion further states that this disclosure was insufficient to amount to disclaimer of automatic pipette guiding mechanisms, because the applicant of the '782 patent had abandoned arguments having to do with scope of claims reciting pipette guiding mechanisms. Reaching this conclusion was supported by their decision in Ecolab, Inc. v. FMC Corp., 569 F.3d 1335, 1343 (Fed. Cir. 2009), in the Court' view.
For these reasons the panel vacated the District Court's claim construction and remanded, where questions of infringement and claim invalidity will be assessed under the Federal Circuit's construction of the "pipette guiding mechanism" term. The Court also awarded costs to Malvern.
* In his dissent from this en banc decision upon which de novo review of claim construction by the Federal Circuit was established until partially overturned by the Supreme Court in Teva Pharma v. Sandoz, Judge Rader cited statistics that in 1997, the Federal Circuit reversed 53% of the cases decided (27% reversed completely, 26% reversed in part) and for 246 cases decided by the Federal Circuit after Markman, 141 explicitly reviewed the District Court's claim construction, and reversed, in whole or part, in 54 of these cases (about 38%). He identified eight problem areas caused by Markman: multiple trials, bias towards summary judgment, new evidence, the "learning curve problem," and a propensity for every decision to be appealed to the CAFC for de novo review.
Malvern Panalytical Inc. v. TA Instruments-Waters LLC (Fed. Cir. 2023)
Panel: Circuit Judges Prosy, Hughes, and Cunningham
Opinion by Circuit Judge Prost










