By Kevin E. Noonan —
 In view of the Supreme Court's "long conference" on September 30th, it seems timely to review the arguments, pro, con, and amicus briefs submitted to the Court asking for certiorari over the Federal Circuit's In re Cellect decision. While that Court's recent Allergan USA Inc. v. MSN Laboratories Private Ltd. opinion may have made the issues (and whether the Supreme Court grants cert) less urgent, the pending U.S. Patent and Trademark Office (PTO) proposed rules limiting terminal disclaimer practice for overcoming obviousness-type double patenting and the permissible scope of those rules by an administrative agency after Loper-Bright Enterprises v. Raimondo continue to garner interest regarding whether the Court will take up review of this decision. Last week, we reviewed the decision and arguments of the parties. Today, we review the seven amicus briefs that were filed on behalf of Petitioner and the brief filed on behalf for Respondent.
In view of the Supreme Court's "long conference" on September 30th, it seems timely to review the arguments, pro, con, and amicus briefs submitted to the Court asking for certiorari over the Federal Circuit's In re Cellect decision. While that Court's recent Allergan USA Inc. v. MSN Laboratories Private Ltd. opinion may have made the issues (and whether the Supreme Court grants cert) less urgent, the pending U.S. Patent and Trademark Office (PTO) proposed rules limiting terminal disclaimer practice for overcoming obviousness-type double patenting and the permissible scope of those rules by an administrative agency after Loper-Bright Enterprises v. Raimondo continue to garner interest regarding whether the Court will take up review of this decision. Last week, we reviewed the decision and arguments of the parties. Today, we review the seven amicus briefs that were filed on behalf of Petitioner and the brief filed on behalf for Respondent.
Amici for Petitioner:
NYIPLA: provided three Questions Presented that ask the Court to consider "whether [ODP] can [properly] vitiate Congressional guarantees of timely patent examination"; whether the Federal Circuit erred in creating a "rigid rule [that] eliminated all equitable considerations [regarding ODP] and ignoring binding precedent"; and whether ODP was abrogated by Congress enacting legislation conforming U.S. patent law with the URAAA/TRIPS provisions regarding patent term?
The brief asserts that the Federal Circuit in its Cellect decision "usurped Congress's authority [by] abrogating [the statutory patent term] guarantees" and "rejected ODP's equities as well as "significantly adversely impact[ing] innovation." Significantly, the brief provides this table showing the effects of PTA on U.S. patents from 2015 to 2023:

While providing an erudite and thorough discussion on ODP, its origins, and equitable character, the brief makes its most significant arguments with regard to the importance of the Court granting cert., including that "It has been almost a century since the Court substantively addressed double patenting, and in the interim Congress has significantly amended the Patent Code at least five times." Additionally, amicus argues that the Cellect decision will have widespread impact (based inter alia on the data set forth above) and in the NYIPLA's view that the Cellect decision "destroys" the guarantees regarding patent term embodied in the statute. Indeed, amicus argues that "it is time to retire ODP" in view of the changes in U.S. patent law that have made irrelevant the equitable motivation for the doctrine stemming from the potential for serial filings that would award serial 17 year patent terms on patentably indistinct versions of an invention.
AIPLA: argued that the Cellect decision was an improper expansion of ODP to override statutory PTA, based on statutory construction (plain meaning) and amounting to a violation of the clear intent of Congress. PTA is not an unjust extension of the patent term but a statutory restoration of term lost due to PTO delay. The brief notes the structure of the patent term provisions of the statute, wherein § 154(a)(2) defines 20 year term; § 154(b) restores loss of term cause by PTO delay, with § 154(b) being entitled "Adjustment of Patent Term" and § 154(b)(1) being entitled "Patent Term Guarantees." The brief performs some dictionary-based semantics on meanings of "adjust" and "extend" and asserts that "[p]ut simply, there is no unjust extension of term by PTA, but rather recovery of unjustly lost term due exclusively to PTO delay," stating that the Federal Circuit "conjured" Congressional intent contrary to these provisions of the statute. With regard to the crux of the Federal Circuit's construction of the statute, the AIPLA maintains that § 154 "does not address ODP" and § 154(b)(2)(B) does not relate to ODP but rather "provides a commonsense limitation on PTA: if an applicant has already specified a patent's expiration date by disclaiming some amount of term (a 'terminal disclaimer'), the applicant may not go beyond that date [by obtaining PTA]." The Federal Circuit erred by implicating ODP in the application of § 154(b)(2)(B) because terminal disclaimers are purportedly "two sides of the same coin" (even as here under circumstances where TD not filed, although to be fair the basis was that a TD should have been filed). The Court's analysis was error because it violates the "strong presumption" of congressional intent not to be rebutted except under "rare and exceptional circumstances," citing United States v. Clintwood Elkhorn Mining Co., 553 U.S. 1, 11 (2008) (quoting Ardestani v. INS, 502 U.S. 129, 135 (1991)). Here, the brief argues that Congressional intent can be found in the language that the Office shall provide PTA to restore term lost due to PTO delay, citing Smith v. Spizzirri, 144 S. Ct. 1173, 1177 (2024) (quoting Lexecon Inc. v. Milberg Weiss Bershad Hynes & Lerach, 523 U.S. 26, 35 (1998), and Petrella v. MGM, 572 U.S. 663, 685 (2014). The brief also asserts that the Federal Circuit raised a "negative inference" which is improper under circumstances of having a "limited exception" which is itself improper: "voluntarily surrender of statutorily granted PTA via a terminal disclaimer does not mean that Congress intended to nullify PTA entirely by exposing PTA-adjusted patents, where no terminal disclaimer is filed, to ODP." The brief contends that this is not a case of the Federal Circuit "unearth[ing] Congress's hidden intent" but rather that the Court "just rewrote the statute."
The brief also characterizes the decision as being a departure from the equitable underpinnings of ODP, because that doctrine was meant to limit patent term mischief and thus requires a showing of such mischief. The basis is not just an extension of the patent term but an unjust extension, whereas PTA was intended to do exactly the opposite: restore deserved patent term lost due to USPTO delay. The brief accuses the opinion of "gutt[ing] [the ODP] doctrine" of its equitable purpose (citing inter alia Immunex Corp. v. Sandoz Inc., 964 F.3d 1049, 1059 (Fed. Cir. 2020), for declaring ODP to be an equitable doctrine) and that prior ODP jurisprudence was directed towards preventing gamesmanship under Abiomed, Inc. v. Maquet Cardiovascular LLC, Civil Action No. 16-10914-FDS, 2023 U.S. Dist. LEXIS 104095, at *101 (D. Mass. June 15, 2023); Mitsubishi Tanabe Pharma Corp. v. Sandoz, Inc., 533 F. Supp. 3d 170, 214 (D.N.J. 2021); and Abbott Lab'ys v. Lupin Ltd., No. 09-152-LPS, 2011 U.S. Dist. LEXIS 53846, at *26-27 (D. Del. May 19, 2011), on the basis of unjust extension (there being no evidence of gamesmanship here).
The brief also notes that the opinion discards the equitable requirements in favor of a "bright line rule" (which might resonate with the Court as the strongest argument for granting cert if history be our guide) and that some cases in the district court have already opined that "ODP depends solely on patent expiration dates and should not [be] influenced by equitable concerns," citing Allergan USA, Inc. v. MSN Lab'ys Priv. Ltd., No. 19-1727-RGA, 2023 U.S. Dist. LEXIS 172641 (D. Del. Sep. 27, 2023), later reversed by the Federal Circuit (by the same panel thar decided Cellect) as well as Ex parte Clantech, Inc. (PTAB 2024) and ACADIA Pharm. Inc. v. Aurobindo Pharma Ltd. (D. Del. Dec. 13, 2023).
As to the consequences of the decision and the need for the Supreme Court to apply correction, the brief states that the decision has created uncertainty, upset settled expectations, harmed and innovation. The magnitude of the possible implications is raised in the brief by citation of a law review article by Mark A. Lemley & Jason Reinecke, Our More-than-Twenty-Year Patent Term 1, 14-15 (Stanford L. & Econ. Olin Working Paper No. 586, 2023), https://ssrn.com/ab-stract=4529670 regarding the high frequency of patents obtaining PTA having an average of 411 days (some even longer) as well as the burden of monitoring patent estates "claim by claim" to avoid invalidation and the current increase in terminal disclaimer filings.
Intellectual Property Owners (IPO): In a brisk nine pages the IPO's brief emphasizes the Congressional mandate that the term of a patent shall be extended if PTO delay causes term to be lost under § 154(b)(1)(A). "Congress intended these patent term adjustments to be mandatory, not permissive," according to the brief, relying, inter alia on the "black letter law" meaning of the word "shall." It was legal error for the Federal Circuit to "in effect[] overrule[] the statute enacted by Congress based on a judge-made doctrine." This assessment of the Court's error is supported by the Supreme Court's decision that "applied a traditional principle of statutory construction to the Federal Arbitration Act ("FAA") and determined that a judicially-created exception to an FAA provision could not stand," citing Henry Schein, Inc. v. Archer & White Sales, Inc., 139 S.Ct. 524, 530 (2019). The brief also notes that the Federal Circuit came to the opposite conclusion (i.e., that ODP could not overrule a statute, § 156), in Novartis AG v. Ezra Ventures LLC, 909 F.3d 1367, 1375 (Fed. Cir. 2018).
Terminal disclaimers in the PTA regime are "very clear[ly] defined" to have but "a limited role" in the statute: firstly, it is the sole exception to PTA in the statute and accordingly, the scope of the exception should be limited, the brief citing United States v. Johnson, 529 U.S. 53, 58 (2000), for the principle that "[t]he proper inference . . . is that Congress considered the issue of exceptions and, in the end, limited the statute to the ones set forth." The Federal Circuit's error in IPO's opinion was to "assume[] that ODP always applies and only briefly referred to the language of Section 154 after concluding that ODP invalidated Appellant's claims" (emphasis in brief). The proper analysis, according to IPO, is to have "started (and ended) its analysis with the statute" and the proper result, arising from the proper analysis, would be that Cellect was entitled to the PTA because it had never filed a terminal disclaimer (i.e., it would be patentee's actions in doing so that provoked the loss of PTA). The Federal Circuit's opinion to the contrary amounts to "a Catch-22 'gotcha,' [wherein a] patent owner may be entitled to a patent term adjustment under the statute, but according to the Federal Circuit's misinterpretation of the law, the patent term adjustment is a poison pill that can invalidate the patent in its entirety."
The brief asserts that the consequences of the Federal Circuit's decision is that "valuable patent rights will be unfairly invalidated, which in turn will reduce the incentive for future investment in critical research," the evidence for which is that over the past 8 years more than one million patents have been granted PTA. Other consequences include "unnecessary confusion" in the lower courts, citing contrary decisions on similar fact patters, in Acadia Pharms. Inc. v. Aurobindo Pharma Ltd., 2023 U.S. Dist. LEXIS 221663, at *21–24 (D. Del. 2023) (that ODP did not apply), and Allergan USA Inc. v. MSN Labs. Priv. Ltd., 2023 U.S. Dist. LEXIS 172641, at *59–60 (D. Del. 2023) (the latter case having been reversed in the interim by the Federal Circuit). And this uncertainty will apply to continuation practice under § 120 and evoke "retroactive invalidation of many otherwise valid patents" IPO warns in its brief.
Sonos, Ring Central, Nagra Kudelski Group, and Capstan Therapeutics: This collection of smaller innovation-based companies argue in their brief that it is important for patent holders to know how long the patent term will be received in exchange for disclosure of their inventions to the public. This consideration involves both practical (need for investment decisions) and ethical (comprising the quid pro quo of the patent system) realities in their view.
The Federal Circuit's decision in the Cellect case undermines basic certainty for a wide range of inventions, this brief asserts. Reviewing the bases for ODP development these amici argue that, absent gamesmanship it makes "very little sense" to apply ODP to today's circumstances, where all patents expire 20 years from their earliest priority date. Here, the Federal Circuit expanded ODP to extend to additional patent term "(i) expressly provided by statute and (ii) created solely and exclusively by administrative delays within the Patent Office."
This decision and application of ODP to PTA determinations are important, these amici assert, because such administrative delays occur frequently (over 50% of patents filed after enactment of Section 154(b) have been awarded PTA, on average in excess of 6 months, relying on Mark A. Lemley & Jason Reinecke, Our More-than-Twenty-Year Patent Term 1, 14-15 (Stanford L. & Econ. Olin Working Paper No. 586, 2023), https://ssrn.com/ab-stract=4529670 and Dennis Crouch, Prosecution Delays and Patent Term Adjustment on the Rise Again (Nov. 13, 2022), https://pa-tentlyo.com/). In the face of such statistics, the Federal Circuit's decision mandates that "even when a challenged patent expires later than the reference patent due to the Patent Office's delay and not as the result of any tactics by the patent owner, a court must invalidate that patent if it believes an earlier-to-expire patent in the same family renders it obvious" according to amici. This is error because it "allows a judge-made doctrine to cut short a statutory patent term mandated by Congress; contradicts Federal Circuit precedent; treats two forms of statutory term adjustments differently even though the language of the statutes are similar; and upsets equitable considerations underlying the purpose of ODP."
Amici assert that this brief is filed not to rehash these arguments but to "highlight the practical concerns the Federal Circuit's ruling creates for technology-driven companies, and the fundamental ways the ruling contradicts both basic fairness and predictability," stating that this is "not a situation where the [Supreme] Court should wait and see how the doctrine develops" (emphasis in brief) because it "impacts the investments (many of which will be years in the making) that companies like the amici are making now."
Their argument further asserts that "[p]redictability is crucial to innovation," because "[c]ompanies rely on knowing the duration of patent protection in a variety of circumstances including: (i) when making decisions about research and development budgets, (ii) when deciding how to protect their innovations (e.g., whether to apply for patents or rely on trade secret protection), and (iii) when evaluating acquisitions of other companies." The Cellect decision has upended this predictability for at least these reasons:
First, the structure of patents and patent claiming, and how they are used to protect technologies for innovative companies, result in each individual claim protecting only "a small fraction of what has been invented." As a result while the patent specification is a detailed description of several related inventions or aspects thereof each claim set encompasses (and protects) only a small subset of what is disclosed in the specification. These are typically pursued in continuation applications filed according to the priority (i.e., value) of these different aspects. These practices are sanctioned by the patent statute and do not raise concerns regarding gamesmanship (the brief saying it would be counterproductive and expensive to file all these claims in one application). The result is filing many applications in patent portfolios for which, prior to the Cellect decision, patent holders understood their expiration dates (based on their earliest priority dates and any PTA earned during prosecution as a result of PTO delay). This is no longer the case under Cellect, because now patent owners must determine expiration dates for each family member and the claims in each and then assess "the likelihood that a court will later find each individual claim of the later-expiring patents to be obvious relative to one or more claims of the reference [i.e., earliest-expiring] patent" (emphasis in brief) (this assessment constituting "only an educated guess" according to amici). In addition to the legal uncertainties, this exercise poses "a gargantuan problem" in terms of number of patents and "millions in patent costs" required to be performed. "If the Federal Circuit intended to undermine the settled expectations of patent holders, it would be hard to imagine a more effective way to do so," the brief asserts.
Second, there is a great (and negative) impact on investment decisions by innovative companies, amici argue, affecting research and development, whether to pursue patent protection or rely on trade secrets or abandon intellectual property protection entirely. This outcome is contrary to the well-recognized importance of patent protection for supporting innovation, as evinced by the Court's decisions in Pfaff v. Wells Elecs., Inc., 525 U.S. 55, 63, (1998); Apple Inc. v. Samsung Elecs. Co., 809 F.3d 633, 647 (Fed. Cir. 2015); Sanofi-Synthelabo v. Apotex, Inc., 470 F.3d 1368, 1383 (Fed. Cir. 2006); and Patlex Corp. v. Mossinghoff, 758 F.2d 594, 599 (Fed. Cir.), on reh'g, 771 F.2d 480 (Fed. Cir. 1985), and indeed even the Founders' inclusion of provisions for IP protection in the Constitution. These legal principles, amici contend, are supported by academic and other studies showing that "legal decisions that change the expected value of patents have impacts on technology investments," citing as examples Alice Corp. v. CLS Bank Int'l, 573 U.S. 208 (2014) (where "a survey of 475 venture capital and private equity firms reported that 62% of investors said that their firm was less likely to invest in companies developing technologies that may not be patent eligible"; David O. Taylor, Patent Eligibility and Investment, 41 Cardozo L. Rev. 2019, 2027-28 (2020), https://pa-pers.ssrn.com/sol3/papers.cfm?abstract_id=3340937) and another survey after the Supreme Court's Bilski v. Kappos, 561 U.S. 593 (2010), and Mayo Collaborative Services v. Prometheus Laboratories, Inc., 566 U.S. 66 (2012), decisions that showed "investment in diagnostic technologies was nearly $9.3 billion dollars lower than it otherwise would have been"; A. Sasha Hoyt, The Impact of Uncertainty Regarding Pa-tent Eligible Subject Matter for Investment in U.S. Medical Di-agnostics Technologies, 79 Wash. & Lee L. Rev. 397, 445-46 (2022), https://scholarlycommons.law.wlu.edu/wlulr/vol79/iss1/8/).
The Cellect decision not only reduces patent protection value but also makes protection "less predictable" (emphasis in brief), under circumstances where the amount of PTA to which a patentee is entitled depends on "unknown and stochastic processes within the Patent Office." As a consequence, amici argue, the traditional exchange of patent protection for disclosure to the public becomes "something of a lottery" (emphasis in brief). And these are the circumstances that arise if innovative companies continue to pursue patenting; amici contend that the reduction of return on (the quite large) investment in patenting "materially undermines the incentive to apply for patents in the first place" which "represents a serious loss to the public in the long term."
Sanofi, Beigene, Merck Sharp & Dohme, EMD Serono, and Pfizer: This brief provides an extensive history of the development of ODP (also termed herein "non-statutory double patenting" or "NSDP") and also that certainty and predictability has been disrupted by the Federal Circuit's Cellect decision. The brief emphasizes the legal history of ODP and how it has become outdated inter alia by adoption of the 20-year patent term for U.S. patents, as well as the equitable aspects arising before adoption of the URAA/TRIPS regime, as a judicially created doctrine preventing serial filing and issuance of obvious variants of an invention.
The brief asserts that despite elimination of the opportunity for "gamesmanship" in patent term under U.S. law after adoption of the URAA/TRIPS regime the Federal Circuit in Cellect "expands [the scope of] NSDP anyway," according to amici, citing In re Hubbell, 709 F.3d 1140, 1145 (Fed. Cir. 2013), and Gilead Scis., Inc. v. Natco Pharma Ltd., 753 F.3d 1208, 1217 (Fed. Cir. 2014). Cellect in another example, here on the novel question of "whether NSDP could invalidate claims of patents within a family solely because of a statutory grant of PTA to some members of that family" (the answer being yes, of course). In addition, amici argue that the decision has "sow[n] confusion in district courts, citing the poster-children for this confusion, Allergan USA, Inc. v. MSN Lab'ys Priv. Ltd., 2023 WL 6295496 (D. Del. Sept. 27, 2023), and Acadia Pharms. Inc. v. Aurobindo Pharma Ltd., 2023 WL 8803448 (D. Del. Dec. 13, 2023).
Amici argue that this history establishes that the Cellect decision is "destabilizing" to the patent regime, particularly for the pharmaceutical industry, in part due to the "enormous" cost of investment in the face of patent term that is no longer "definite and predictable" being subject to "administrative inefficiencies and judicial whim." The decision not only undermines the equitable principles upon which the ODP doctrine was established but is also legally incorrect based on proper construction of § 154(b)(2)(B) of the statute, these amici argue. "The Federal Circuit applied a judge-made equitable doctrine in an inequitable way, contravening Congress's statutory guarantees in the process," the brief summarizes. The brief also supports the value of patents (and the need for predictability thereof) in the pharmaceutical and other industries with numerous academic legal and other studies attesting to the public benefit of this investment. The brief further addresses the "balance" between lowering drug prices and encouraging (through profit) investment to develop new drugs ("a policy question for Congress—not courts—and Congress has addressed that policy question many times," citing examples such as the Hatch-Waxman Act).
In addition to these policy and practical errors, the brief expounds on the legal errors committed by the Federal Circuit, including misconstruing § 154(b)(2)(B). These number three: first, contrary to the panel's rationale, "terminal disclaimers are not tied to NSDP the way the Federal Circuit presumed" (i.e., that filing a terminal disclaimer was always associated with NSDP). Second, while NSDP was crafted (and § 154(b)(2)(B) enacted) for circumstances involving "serially filed applications with different default patent terms" (emphasis in brief) the Federal Circuit applied the doctrine to patents within the same patent family sharing a common default patent term (i.e., 20 years from a common earliest priority date). Properly understood, amici argue that § 154(b)(2)(B) was intended to apply to situations where "original patent applications containing patentably indistinct claims are filed separately on different dates, rather than as part of the same patent family, resulting in patents with different default terms," [wherein] "NSDP might apply to the later-filed, later-expiring patent because the patents would have different default patent terms—different start and end dates—which is controlled by the patentee." (And to the extent Congress wanted the Federal Circuit's interpretation to apply, the brief argues it would have said so expressly.)
Amici also contend that applying NSDP in this situation is a misapplication of a doctrine intended to police unjustified extension of patent term to patent term extensions justified by a grant expressly enacted by Congress. "Statutory guarantees like PTA are not unjustified, nor are they even in the patentee's control," the brief asserts, providing two reasons why applying NSDP as the Federal Circuit did in this case was error. Finally in this regard amici argue that applying NSDP to situations not involving gamesmanship or other culpable behavior is a misapplication of the equitable principles underpinning NSDP.
The brief concludes with an exhortation of the importance for the Court to grant certiorari, inter alia, to address the Federal Circuit's replacement of "Congress's carefully calibrated patent regime with one of judicial design" specifically with regard to pharmaceutical innovation including the costs thereof and the accompanying need for predictability for this technology.
Pharmaceutical Research and Manufacturers of America (PhRMA) and Biotechnology Innovation Organization (BIO): The principle argument in this brief is that the Federal Circuit used judicially created doctrine to overrule statutory PTA, while secondarily the brief argues that the decision upsets settled expectations. Amici's stated interests are "[t]o protect [their] investments, members of PhRMA and BIO depend on a patent system that is robust, fair, and predictable. In particular, amici rely on the patent system to protect the diverse array of innovations they make in connection with developing a new medicine or treatment, including the unique compositions required to safely and effectively use new medicines in patients and novel manufacturing technologies needed to safely produce amounts of those medicines sufficient to meet an often immense patient demand."
Amici argue that the nature of these technologies requires "filing a series of related patent applications derived from an original filing based on the inventive work." As a consequence, they obtain patents with various expiration dates that are "dictated by the statutory scheme at issue here," wherein those terms vary according to the PTO "failing to meet its statutory deadlines in conducting the examination of the associated application."
The brief contains these graphics to illustrate the effects on patent term created by the U.S. adopting the URAA/TRIPS regime that changed the term of U.S. patents from 17 years from issue to 20 years from earliest priority date:

The prejudice illustrated in this graphic motivated Congress to enact 35 U.S.C. § 154(b) to restore term lost due to PTO delay:

With specific instances of how the regime responds illustrated in this figure:

The brief asserts that this "judge-made doctrine of non-statutory double patenting cannot displace these statutory requirements for patent-term adjustments" which is "clear from the statutory text of 35 U.S.C. § 154(b)." (The brief also notes that the equitable basis for ODP was largely eliminated by the changes in term occasioned by adopting the URAA/TRIP regime.)
The patent term adjustment regime according to the brief is "an essential part of the PTO's application-by-application examination process," consisting of a "back-and-forth dialogue between the PTO and the inventor." In order to adequately protect innovation in these disciplines, "[l]ife sciences innovators commonly file robust applications reflecting the broad array of innovations that come with developing a new medicine," which include "not only the most promising active ingredient discovered but other promising candidates as well." The result is that innovators can obtain "patent rights that provide commercially viable protection" that, in addition to preventing competitors from avoiding their patents by making simply narrow changes also benefits the public due to publication that occurs whether or not a patent is ever obtained. But such patent applications take time to secure and often involve several rounds of continuation applications to pursue some claims not included in a patent arising from the first application filed but nevertheless ultimately deemed patentable (the brief explicating the history of such applications and the benefits that arise therefrom).
The brief further sets out their argument that judge-made law cannot negate the statutory mandate, citing the differences between Novartis Pharmaceuticals Corp. v. Breckenridge Pharmaceutical Inc. (regarding PTA under § 154(b)) and Novartis AG v. Ezra Ventures LLC, (regarding PTE under § 156(a)). Also argued are amici's contentions that the Federal Circuit misinterpreted the statutory language, which was related to instances where a terminal disclaimer had been filed, not ones where a court determines it should have been (and in a footnote echoes other amici that the Federal Circuit erred in considering ODP as the exclusive reason for filing a terminal disclaimer).
With regard to settled expectations, the brief emphasizes the risk to innovative industries due to the time and expense of bringing pharmaceutical and biotechnology products to market and the related need for reliable patent protection with predictable term to ensure a chance at sufficient return to justify investment. The brief says that the Federal Circuit's error relates to all technologies, citing Festo Corp. v. Shoketsu Kinzoku Kogyo Kabushiki Co., 535 U.S. 722, 739 (2002), but particularly affects innovation for pharmaceutical and biotechnology inventions (supported by several academic and industry studies) due to the excessive costs and time it takes to bring products to market in these industries and the propensity for failure.
Finally, the brief reminds the Court that they have "often granted certiorari to realign the Federal Circuit's doctrines with governing statutes," naming SAS Inst. Inc. v. Iancu, 138 S. Ct. 1348 (2018); Halo Elecs., Inc. v. Pulse Elecs., Inc., 136 S. Ct. 1923 (2016); and Nautilus, Inc. v. Biosig Instruments, Inc., 572 U.S. 898 (2014) (frankly the brief could have named several more but the point was undoubtedly made).
Teige P. Sheehan: Teige P. Sheehan, a patent attorney, filed an amicus brief on his own behalf and argued specifically that the Federal Circuit's reasoning was circular and that the statute requires ODP to be resolved before PTA is assessed. Regarding the circularity argument, the brief contends that a terminal disclaimer is a "cure" for an ODP defect by truncating the patent term to an earlier expiration date and that § 154(b)(2)(B) states that a PTA award cannot extend the expiration date later than the date set by the terminal disclaimer. The Federal Circuit held that "because § 154 states that a terminal disclaimer—the cure for ODP—limits a PTA award, a PTA award itself can confer unpatentability for ODP in the absence of a terminal disclaimer" (emphasis in brief). Mr. Sheehan asserts in his brief that "[t]his is a classic instance of circular reasoning, an illogical basis for analysis repeatedly dismissed by this Court (e.g., in Nationwide Mut. Ins. Co. v. Darden, 503 U.S. 318, 327 (1992); King v. St. Vincent's Hosp., 502 U.S. 215, 222 (1991); Morse v. Republican Party of Virginia, 517 U.S. 186, 233 n.43 (1991); and Chevron, U.S.A., Inc. v. Nat. Res. Def. Council, Inc., 467 U.S. 837 (1984)) and which "provides no insight at all into whether Congress intended for a PTA award to confer ODP invalidity." "[T]he statute explains the consequence on PTA of a terminal disclaimer having been entered to overcome ODP," the brief argues, [but] "[i]t does not mean, as the Federal Circuit would have it, [that] PTA creates the need for the terminal disclaimer by causing ODP invalidity." And as with other amici (and Cellect itself) the brief also argues that this application of ODP doctrine by the Federal Circuit thwarts Congress's scheme for restoring patent term due to PTO delays under § 154(b).
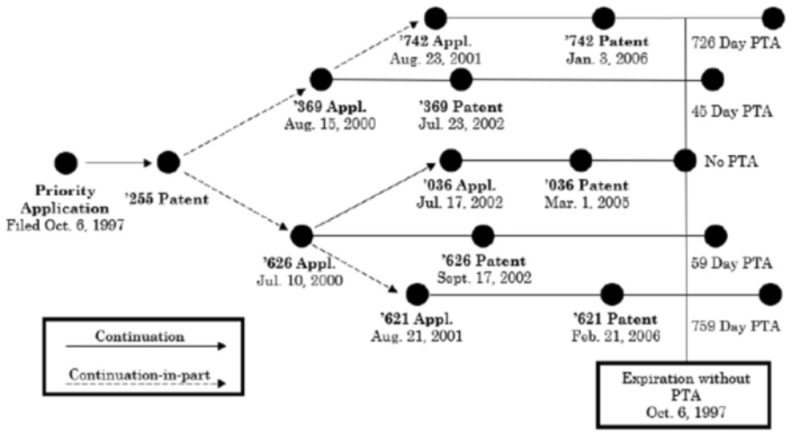
The statute asserts that the proper application of the statute, consistent with Congressional intent, is for ODP issues to be resolved before application of any PTA is awarded. Such an application of the law would avoid instances that cause their own, unique anomalies, such as where "a patent can validly issue with a PTA award and its corresponding expiration date publicized, only to be rendered invalid for ODP later, when another patent subsequently issues." This is exactly what occurred for two patents at issue in Cellect: "U.S. Patent No. 6,424,369 ("the '369 Patent") issued with an award of 45 days of PTA. Over two-and-a-half years later, another patent was issued to Petitioner, U.S. Patent No. U.S. 6,862,036 ("the '036 Patent"), without receiving a PTA award. . . . The '369 Patent's 45 days of PTA meant it was set to expire after the '036 Patent did, resulting in a supposedly unjust extension of patent term, according to Respondent [and the Federal Circuit]."
The brief concludes that "[t]his inflexible application of ODP is not only contrary to Congress's intent that ODP should be determined based on a patent's expiration date absent PTA, as expressed in § 154, but also serves none of the purported purposes of ODP doctrine while delivering an inequitable result to a patentee."
Amicus for Respondent:
Inari Agriculture, Inc.: The only amicus brief submitted in support for Respondent was filed by Inari Agriculture, Inc. and this brief takes a decidedly aggressive and aggrieved tone in favor of the Federal Circuit's decision and the rationales behind it. The company describes itself as being formed to "develop pioneering technology to selectively edit plant genes to enhance agronomic traits to increase crop yields and decrease inputs such as water and fertilizer," that "partners with independent seed companies to develop improved seeds using Inari's technology" but for whom ODP is needed to "protect[] innovators like Inari-from earlier patentees seeking to leverage legacy technology after their patents expire." It castigates "an oligopoly of entrenched incumbents" that "dominate seed distribution and suppress competition" by abusing the patent system, spurred on by "commentaries [that] highlighted brazen strategies for exploiting loopholes in the PTA system [citing blog posts directed to maximizing PTA-increased term] and delaying patent issuance while wrongly attributing such delays to the PTO." Examples cited in the brief include "[t]wo companies-Corteva and Bayer/Monsanto[that] control over 70% of the U.S. corn seed market and 85% of corn-related intellectual property" and that "[t]ogether with BASF and ChemChina's Syngenta Group, these oligopolists own 95% of corn-related IP," by exerting patent rights over genetically modified seed. The brief also cites extensively from briefs and other assertions by the Federal Trade Commission regarding such putative abuses, which the brief contends will (and in some instances at least prospectively already have begun to) address these issues (albeit a large portion of the brief also seems to set forth Inari's own arguments against these companies rather than address the question of certiorari grant, except to the extent that by doing so the Court might take away a tool that Inari believes will aid it in competing with the "oligarchs.")
One final consideration remains (not addressed in these briefs because it arose after they were filed), which is the Federal Circuit's recent decision in Allergan USA Inc. v. MSN Laboratories Private Ltd. In that case, a panel made up of the same Federal Circuit judges that decided In re Cellect reversed a district court decision that putatively applied the Cellect calculus for assessing when PTA can invalidate claims under the ODP doctrine. The relationship between the patents at issue in Allergan was as follows:
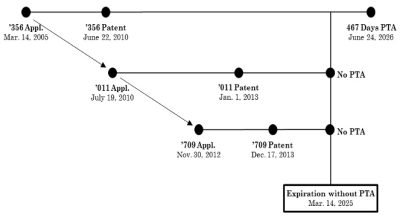
In this case, the district court held that the '356 patent was invalid on obviousness-type double patenting grounds (ODP) for having patentably non-distinct claims over the '011 and '709 patents. The district court expressly relied on the Federal Circuit's Gilead Scis., Inc. v. Natco Pharma Ltd. and In re Cellect decisions in reaching this judgment. Nevertheless, the Federal Circuit reversed. The rationale behind this (seemingly contrary) decision was that "Cellect established a rule that, when it comes to evaluating ODP on a patent that has received PTA, the relevant expiration date is the expiration date including PTA—not the original expiration date measured twenty years from the priority date." That was not the factual posture in Allergan, according to the Court, which asserted the following rule for determining the effects of OPD on PTA: "a first-filed, first-issued, later-expiring claim cannot be invalidated by a later-filed, later-issued, earlier-expiring reference claim having a common priority date"; as applied here the district court erred "[b]ecause the '356 patent was the first patent in its family to be filed and to issue, it [thus] does not extend any period of exclusivity on the claimed subject matter." A comparison of the timelines in Allergan and Cellect reveals that these decisions are not completely consistent. The existence of the decision, however, might be enough to convince the Supreme Court that it would be more prudent not to grant Cellect's certiorari petition and to permit the Federal Circuit to resolve a consistent application of ODP and PTA law before the Justices wade into these murky waters themselves. We should know how the Court decides by tomorrow.