By Kevin E. Noonan —
 There are (at least) two ways of looking at the course of the Federal Circuit's evolving interpretation of the Supreme Court's subject matter eligibility jurisprudence under Mayo Collaborative Services v. Prometheus Labs., Inc. and Alice Corp. v. CLS Bank Int'l. One way is to consider the Court to be adrift, unable to come to a consensus on how the law should be interpreted and thus calling into question the wisdom of Congress's decision to establish a court with purportedly specialized expertise in patent law (see, e.g., "In Defense of the Federal Circuit"). Alternatively, the exercise can be analogized to sausage-making (more usually an analogy used for how legislation is crafted), in recognition that the Supreme Court's Mayo/Alice jurisprudence, while enunciated as a two-part test has been, in the practice, much less straightforward in application. The understanding and interpretation of either analysis is influenced anew with every Federal Circuit decision, and the Court's recent decision in Cardionet, LLC v. Infobionic, Inc. illustrates anew the internal divisions, where one judge (Judge Dyk) agreed with the outcome but felt compelled to write in partial dissent on one aspect of the majority's reasoning.
There are (at least) two ways of looking at the course of the Federal Circuit's evolving interpretation of the Supreme Court's subject matter eligibility jurisprudence under Mayo Collaborative Services v. Prometheus Labs., Inc. and Alice Corp. v. CLS Bank Int'l. One way is to consider the Court to be adrift, unable to come to a consensus on how the law should be interpreted and thus calling into question the wisdom of Congress's decision to establish a court with purportedly specialized expertise in patent law (see, e.g., "In Defense of the Federal Circuit"). Alternatively, the exercise can be analogized to sausage-making (more usually an analogy used for how legislation is crafted), in recognition that the Supreme Court's Mayo/Alice jurisprudence, while enunciated as a two-part test has been, in the practice, much less straightforward in application. The understanding and interpretation of either analysis is influenced anew with every Federal Circuit decision, and the Court's recent decision in Cardionet, LLC v. Infobionic, Inc. illustrates anew the internal divisions, where one judge (Judge Dyk) agreed with the outcome but felt compelled to write in partial dissent on one aspect of the majority's reasoning.
The case arose in litigation between the parties over cardiac monitoring technology protected under U.S. Patent No. 7,941,207. The Court considered claims 1, 2, 3, 7, 10-12, and 22 to be relevant:
1. A device, comprising:
a beat detector to identify a beat-to-beat timing of cardiac activity;
a ventricular beat detector to identify ventricular beats in the cardiac activity;
variability determination logic to determine a variability in the beat-to-beat timing of a collection of beats;
relevance determination logic to identify a relevance of the variability in the beat-to-beat timing to at least one of atrial fibrillation and atrial flutter; and
an event generator to generate an event when the variability in the beat-to-beat timing is identified as relevant to the at least one of atrial fibrillation
and atrial flutter in light of the variability in the beat-to-beat timing caused by ventricular beats identified by the ventricular beat detector.
2. The device of claim 1, wherein the relevance determination logic is to accommodate variability in the beat-to-beat timing caused by ventricular beats by weighting ventricular beats as being negatively indicative of the one of atrial fibrillation and atrial flutter.
3. The device of claim 1, wherein the variability determination logic is to compare times between R-waves in three successive QRS complexes to determine the variability in the beat-to-beat timing.
7. The device of claim 1, wherein the event generator is to generate an event by performing operations comprising: collecting data associated with the collection of beats; and transmitting the data associated with the collection of beats to a remote receiver.
10. The device of claim 1, wherein the relevance determination logic comprises logic to identify the relevance of the variability using a non-linear function of a beat-to-beat interval.
11. The device of claim 1, wherein the beat detector comprises a QRS detector.
12. The device of claim 1, further comprising a sensor that includes two or more body surface electrodes subject to one or more potential differences related to cardiac activity.
22. An article comprising one or more machine-readable media storing instructions operable to cause one or more machines to perform operations, the operations comprising: determining a beat-to-beat variability in cardiac electrical activity; determining a relevance of the variability over a collection of beats to one of atrial fibrillation and atrial flutter using a non-linear function of a beat-to-beat interval; and identifying one of an atrial fibrillation event and an atrial flutter event based on the determined relevance, the event being a period in time when the information content of the cardiac electrical activity is of increased relevance to the one of atrial fibrillation and atrial flutter [wherein] determining the relevance comprises: identifying a beat of the collection as a ventricular beat, and weighting the beat as being negatively indicative of the one of atrial fibrillation and atrial flutter.
(where the claim language in italics is recited in independent claim 20 from which claim 22 depends). The opinion also illustrated the invention with a reproduction of Figure 10*:
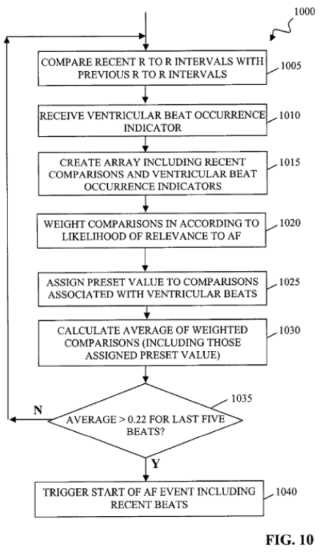 As the opinion sets forth, the invention permits a clinician to distinguish atrial fibrillation and atrial flutter from other forms of cardiac arrhythmias with regard to "beat-to-beat variability in heart rate over a series of successive heartbeats." Importantly, the invention achieves its objectives "by analyzing the beat-to-beat timing for atrial fibrillation or atrial flutter while also taking into account the variability in the beat-to-beat timing caused by premature ventricular beats." As a consequence, "the device can more accurately distinguish atrial fibrillation and atrial flutter from other types of arrythmias and has 'improved positive predictability of atrial fibrillation and atrial flutter," resulting (as set forth in the specification) in "a sensitivity to [these two arrhythmias] in excess of 90% and a positive predictivity in excess of 96%."
As the opinion sets forth, the invention permits a clinician to distinguish atrial fibrillation and atrial flutter from other forms of cardiac arrhythmias with regard to "beat-to-beat variability in heart rate over a series of successive heartbeats." Importantly, the invention achieves its objectives "by analyzing the beat-to-beat timing for atrial fibrillation or atrial flutter while also taking into account the variability in the beat-to-beat timing caused by premature ventricular beats." As a consequence, "the device can more accurately distinguish atrial fibrillation and atrial flutter from other types of arrythmias and has 'improved positive predictability of atrial fibrillation and atrial flutter," resulting (as set forth in the specification) in "a sensitivity to [these two arrhythmias] in excess of 90% and a positive predictivity in excess of 96%."
CardioNet asserted claims 1, 2, 3, 7, 10-12, and 22 against Infobionic. The District Court dismissed CardioNet's complaint under Fed. R. Civ. Proc. 12(b)(6), on the ground that the claims were ineligible for patenting under 35 U.S.C. § 101 as interpreted under the Supreme Court's Mayo/Alice test. The District Court determined that, under step 1 of the test the claims were directed to an abstract idea, that these arrhythmias "can be distinguished by focusing on the variability of the irregular heartbeat." And under step 2 of the test, the District Court held that "CardioNet 'd[id] not identify improvements to any particular computerized technology'" related to detecting electrical signals relevant to cardiac function. This appeal followed.
The Federal Circuit reversed and remanded, in an opinion by Judge Stoll joined in full by Judge Plager and in part by Judge Dyk, who dissented over the terminal portion of the opinion as set forth in further detail below. The opinion does not start promisingly, citing Athena Diagnostics, Inc. v. Mayo Collaborative Servs., LLC, 915 F.3d 743, 749 (Fed. Cir. 2019), for the standard of review (de novo, with factual issues construed in favor of the non-moving party). After setting forth the details of the Mayo/Alice test, as well as citing Federal Circuit precedent on how that jurisprudence has been applied, the opinion turned to its assessment of how this jurisprudence should be applied to the claims before it.
The panel renders its decision on step 1, holding that the claims are not merely directed to an abstract idea. According to the opinion (which is unanimous on this point), the claims are directed to an improvement in cardiac monitoring technology. The opinion states:
[T]he language of claim 1 indicates that it is directed to a device that detects beat-to-beat timing of cardiac activity, detects premature ventricular beats, and determines the relevance of the beat-to-beat timing to atrial fibrillation or atrial flutter, taking into account the variability in the beat-to-beat timing caused by premature ventricular beats identified by the device's ventricular beat detector.
In the Court's view, the claims "focus" on specific methods for improving this technology, and are not directed to an abstract idea itself or ones that "merely invoke generic processes and machinery." These conclusions, the opinion maintains, are supported by the written description, the opinion citing portions of the specification directed to "multiple technological improvements" (citations to the specification omitted):
First and foremost, the device more accurately detects the occurrence of atrial fibrillation and atrial flutter—as distinct from [ventricular tachycardia] V-TACH and other arrhythmias—and allows for more reliable and immediate treatment of these two medical conditions. . . . Indeed, the written description reports that when analyzing real-world arrhythmia data, the device demonstrated both high "positive predictivity" of, and high "sensitivity" to, atrial fibrillation and atrial flutter, meaning that it effectively avoids false positives and false negatives, respectively, in detecting these two conditions. In addition, the device is able to identify sustained episodes of atrial fibrillation and atrial flutter that have "increased clinical significance."
(The opinion recites additional aspects of the invention in support of its conclusion recited in dependent claims.)
The opinion cites Federal Circuit precedent consistent with its opinion here, including Visual Memory LLC v. NVIDIA Corp. for the proposition that improvements to computer processing (in Visual Memory, computer memory) could be patent-eligible. The opinion notes that it was "important to our determination" in favor of patent eligibility in Visual Memory that the specification set forth "advantages offered by" the claimed invention (supplying a seemingly bright-line practice tip to patent prosecutors, at least for now). The panel found the same type of "advantages" recited in the '207 specification, with the same result regarding patent-eligibility. The Court found similar support, based on similar analogies, in McRO, Inc. v. Bandai Namco Games Am. Inc. and support for the principle that "patent's written description[ can] inform[] our understanding of the claims" in Chamberlain Grp., Inc. v. Techtronic Indus. Co.
The panel found the District Court's error in "the incorrect assumption that the claims are directed to automating known techniques" (as InfoBionic argued below and maintained in its arguments to the Federal Circuit). But "nothing in the record" supports this view nor the fact-finding by the District Court, according to the opinion. The lower court also erred, in the Federal Circuit's view, in its consideration of the disclosure in the '207 patent specification, where it disregarded CardioNet's attempts to point out the relevant differences between the invention and the prior art. Part of this error was also procedural, because in considering a motion to dismiss, the District Court was obliged to "construe all facts and draw all reasonable inferences in favor of CardioNet, the non-moving party," which the District Court did not do here. Finally, the Federal Circuit disagreed that the facts and issues before the court were comparable to the facts and legal determinations in Berkheimer v. HP Inc. and FairWarning IP, LLC v. Iatric Systems, Inc., relied upon by the District Court.
The opinion then turns to a discussion of the question of whether the Court can decide patent eligibility or must remand to the District Court for factual assessments as to the state of the art and whether the claims are directed merely to automating practices long used by physicians (it is here that Judge Dyk parts company with his brethren). The panel majority held that step 1 of the Mayo/Alice test is directed to the question of "whether the claims as a whole are 'directed to' an abstract idea, regardless of whether the prior art demonstrates that the idea or other aspects of the claim are known, unknown, conventional, unconventional, routine, or not routine," citing Diamond v. Diehr, 450 U.S. 175, 188–89 (1981) (a holding that puts Diehr and Mayo in direct opposition should the Supreme Court deign to decide the distinctions between the panel majority and Judge Dyk). The majority opinion then distinguishes the purpose of Section 101 with the other provisions of the patent statute, sub silentio restoring the analysis to the state of the law pre-Mayo. While acknowledging Judge Dyk's appreciation of statements in both Mayo and Bilski v. Kappos suggesting that the state of the prior art may be relevant, the majority notes that "in neither Bilski nor Alice did the Court rely on an examination of the prior art as part of its step one inquiry." Instead, the majority cites the intrinsic evidence as the proper source of information for a court to arrive at the answer to step 1 of the Mayo/Alice test for determining patent eligibility. For the majority, "[t]his court's decision in Enfish, LLC v. Microsoft Corp. . . . confirms this point": "the Alice step one inquiry in Enfish and our other decisions began, and ended, with the patent itself," according to the majority. To the extent that there is evidence that the claims recite "longstanding practice where there is no evidence of such practice in the intrinsic record," the majority believes a district court can take judicial notice thereof in appropriate circumstances. For the majority the question, and its answer, is clear:
Thus, we simply clarify that step one of the Alice frame-work does not require an evaluation of the prior art or facts outside of the intrinsic record regarding the state of the art at the time of the invention. Neither Bilski, Alice, nor this court's precedent endorses such an analysis. [O]ur analysis at Alice step one involves examining the patent claims in view of the plain claim language, statements in the written description, and the prosecution history, if relevant. . . . The analysis does not require a review of the prior art or facts outside of the intrinsic record regarding the state of the art at the time of the invention.
Judge Dyk disagreed. The Judge rejects the majority's contention that its decision is consistent with Supreme Court and Federal Circuit precedent, and disparages it as mere dicta, not being a basis for the Court's holding here (the dissent cites to the defendant's admission that there were no factual disputes to be resolved on remand). For this Circuit Judge, the majority's error is "limiting the use of extrinsic evidence to establish that a practice is longstanding," a position that the dissent notes was not taken by either party. Judge Dyk cites Bilski and Alice specifically for the principle that the Court considered extrinsic evidence concerning the conventionality of claimed methods in determining that claims in these cases were ineligible for being directed to an abstract idea. The Judge also cites Federal Circuit precedent, including Intellectual Ventures I LLC v. Erie Indem. Co., Affinity Labs of Texas, LLC v. DIRECTV, LLC, 838 F.3d 1253 (Fed. Cir. 2016), Content Extraction & Transmission LLC v. Wells Fargo Bank, Berkheimer, and cases cited by the majority (BASCOM, FairWarning) in support of this position. In Judge Dyk's view, the Federal Circuit "[has] persistently looked outside the intrinsic evidence" in deciding issues under step 1 of the Mayo/Alice test. And the Judge finds no authority to support its conclusion that a district court is limited to intrinsic evidence in deciding the question of whether a claim is directed to an abstract ides under step 1 of the Supreme Court's Mayo/Alice test.
Cardionet, LLC v. Infobionic, Inc. (Fed. Cir. 2020)
Panel: Circuit Judges Dyk, Plager, and Stoll
Opinion by Circuit Judge Stoll; opinion dissenting in part and concurring in the result by Circuit Judge Dyk
* The opinion also contains an explication of some of the technical terms used in the claims; readers are directed to those explanations, which are outside the scope of this post.