By Kevin E. Noonan —
 Senior Party The Broad Institute, Harvard University, and the Massachusetts Institute of Technology (collectively, "Broad") filed its motion in opposition to Junior Party The University of California/Berkeley, the University of Vienna, and Emmanuelle Charpentier (collectively, "CVC") motion for priority in Interference No. 106,115. Although Broad argued in its own priority motion that the invention as defined by the Count was one that could only be conceived once it was reduced to practice (a standard originally applied to since-invalidated claims to isolated DNA) and, not coincidentally that Broad's earliest actual reduction to practice (ARTP) antedated CVC's ARTP, in its opposition to CVC's priority motion, Broad takes a more conservative albeit more strongly supported tack.
Senior Party The Broad Institute, Harvard University, and the Massachusetts Institute of Technology (collectively, "Broad") filed its motion in opposition to Junior Party The University of California/Berkeley, the University of Vienna, and Emmanuelle Charpentier (collectively, "CVC") motion for priority in Interference No. 106,115. Although Broad argued in its own priority motion that the invention as defined by the Count was one that could only be conceived once it was reduced to practice (a standard originally applied to since-invalidated claims to isolated DNA) and, not coincidentally that Broad's earliest actual reduction to practice (ARTP) antedated CVC's ARTP, in its opposition to CVC's priority motion, Broad takes a more conservative albeit more strongly supported tack.
In its most recent motion, Broad argues that CVC did not have conception of the invention defined by the '115 Interference Count because the person of ordinary skill in the art could not have any reasonable expectation of success in ARTP from the evidence CVC uses in support of its motion, citing Hitzeman v. Rutter, 243 F.3d 1345, 1357-58 (Fed. Cir. 2001). As a reminder, conception is "the formation in the mind of the inventor, of a definite and permanent idea of the complete and operative invention, as it is hereafter to be applied in practice." Hybritech Inc. v. Monoclonal Antibodies, Inc., 802 F.2d 1367, 1376, 231 U.S.P.Q. 81, 87 (Fed. Cir. 1986), citing Coleman v. Dines, 754 F.2d 353, 359, 224 USPQ 857, 862 (Fed. Cir. 1985). Since conception occurs in the mind of the inventor, there must be "corroborating evidence of a contemporaneous disclosure that would enable one of ordinary skill to make the invention." Burroughs Wellcome, Id. at 1919, citing Coleman v. Dines, 754 F.2d 353, 359, 224 USPQ 857, 862 (Fed. Cir. 1985). However, conception of a method does not require knowledge that the invention will work for its intended purpose. Burroughs Wellcome Co. v. Barr Labs, Inc., 40 F.3d 1223, 32 USPQ 2d 1915 (Fed. Cir. 1994). Relevant to Broad's arguments in their motion, in Burroughs Wellcome, the claims of the patents-in-suit were directed to methods for using AZT for treating AIDS, and the issue was whether the AZT inventors had conceived of the claimed methods before obtaining evidence that AZT could indeed provide an effective treatment for HIV infection. Id. at 1225. The Burroughs Wellcome defendants argued that for an invention in an "uncertain or experimental discipline, where the inventor cannot reasonably believe an idea will be operable until some result supports that conclusion," conception occurs only when there is experimental confirmation that the invention works for its intended purpose. Id. at 1228. The Federal Circuit was clear, stating: "[b]ut this is not the law. An inventor's belief that his invention will work or his reasons for choosing a particular approach are irrelevant to conception." Id., citing MacMillan v. Moffett, 432 F.2d 1237, 1239, 167 U.S.P.Q. 550, 552 (CCPA 1970). This is sufficient for conception, unless there is evidence of subsequent experimental failure (the argument Broad relies upon in their argument against CVC): "[a] conception is not complete if the subsequent course of experimentation, especially experimental failures, reveals uncertainty that so undermines the specificity of the inventor's idea that it is not yet a definite and permanent reflection of the complete invention as it will be used in practice." Id. at 1229, citing Rey-Bellet v. Engelhardt, 493 F.2d 1380, 1387, 181 U.S.P.Q. 453, 457-58 (CCPA 1974).
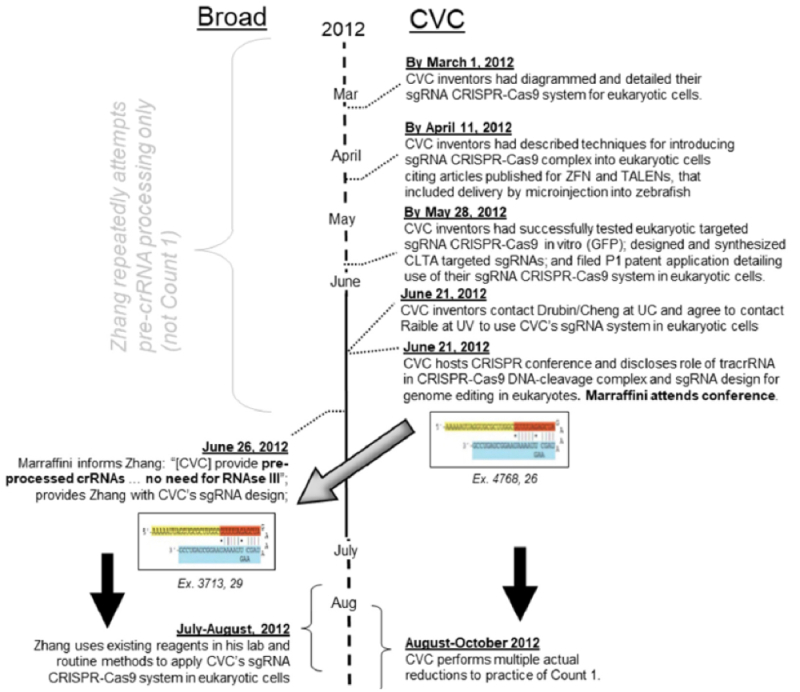
Although stated differently (and in a way that is supported by the Board's decision in prior Interference No. 106,048 as affirmed by the Federal Circuit), Broad's argument is that CVC's conception was flawed as evidenced by repeated failures to reduce the invention to practice. Broad also supports this assertion by contemporaneous statements by CVC's named inventors as well as statements by experts CVC recruited in its efforts to achieve ARTP from the time of its asserted conception (March 1, 2012) to the priority date accorded by the Board in this interference, the filing date of U.S. Provisional Application No. 61/757,640, January 28, 2013.
Broad references its own priority motion for the comparator of having achieved ARTP "no later than July 31, 2012, followed by further ARTP in its October 5, 2012." This is Broad's line in the sand, establishing that CVC be required to show conception prior to the July 31, 2012 date followed by diligence until actual or constructive reduction to practice. This, of course, Broad argues CVC cannot do.
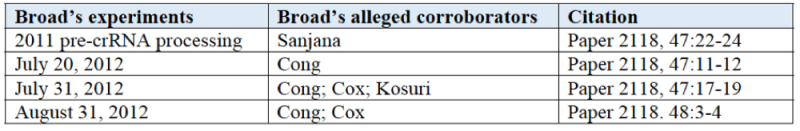
Broad's brief argues three grounds for its assertion that CVC's conception failed. First, Broad argues that CVC lacked a reasonable expectation of successfully targeting and cleaving DNA in a eukaryotic cell. Second, Broad argues CVC lacked a definite and permanent idea of the operative invention as of their alleged conception date. Third, Broad argues that CVC did not have possession of a system that could target and cleave eukaryotic DNA as required by the Count. Broad further argues that CVC's asserted ARTP (when finally achieved) failed for proof that DNA was cleaved in zebrafish cells on August 9, 2012, and further failed to show DNA cleavage in human cells on October 31, November 1, 5 and 18, 2012.
The brief characterizes CVC's priority evidence as "no more than an ill-defined research plan" constituting "a laundry list of possible techniques enlisting at least six different highly-skilled research labs" that failed. Indeed, the brief alleges that only one of the six research lab collaborations were revived and that one only after Zhang published Broad's results in the Cong et al. Science article, nicely turning back on CVC its allegations that Broad derived their invention from information disclosed publicly by the Doudna/Charpentier group. These efforts amounted to "failure after failure" according to the brief, due to the obstacles of "RNA degradation, misfolding, complexation, localization, and chromatin access," just the obstacles Broad has maintained prevented the person of ordinary skill in the art from having a reasonable expectation of successfully adapting CRISPR to a eukaryotic cell context, here and in the prior '048 Interference. The brief relies on the earlier PTAB determinations as affirmed by the Federal Circuit but here prudently also asserts evidence that neither CVC's conception nor ARTP had succeeded in time to be entitled to priority over Broad's invention. The Broad contends that CVC's arguments here are the same as in the previous interference, and the outcome here is the same based on CVC's priority evidence which was not assessed in the earlier interference. As it has in other contexts, Broad puts forth statements by the inventors (including by Jennifer Doudna in her book, A Crack In Creation: Gene Editing and the Unthinkable Power to Control Evolution and Walter Issacson's book, The Code Breaker: Jennifer Doudna, Gene Editing, and the Future of the Human Race) which the brief characterizes as admissions, including:
• "this will be fabulous if it works" (Ex. 4406)
• "test whether the strategy can be used to induce DSBs in mammalian cells" (Ex. 4381 at 65)
• "there is a hint it [CRISPR-Cas9] might work but we shouldn't be overexcited now" (Ex. 4911)
• "aspects of the RNA expression/stability/Cas9/assembly/ localization are problematic" (Ex. 5041)
• "I wonder if having a too-efficient NLS on Cas9 is actually counterproductive" (Ex. 4988)
In addition, the brief contains reference to statements by several other scientists supporting Broad's assertions that complete conception of eukaryotic CRISPR required actual reduction to practice (including Dr. Luciano Marraffini, perhaps anticipating his deposition or at least providing Broad with a basis for cross-examining any testimony he provides that support CVC's allegations set forth in their granted motion for leave from the Board to depose him).
With regard to the Jinek laboratory notebook evidence CVC proffered to show its earliest conception date, March 1, 2012, the brief calls it a "cartoon" without any accompanying disclosure regarding how adaptation of CRISPR to the eukaryotic cell context would be achieved. The brief then goes through the evidence provided by CVC regarding these efforts (including the Board's prior determination that CVC was not entitled to priority to its earliest provisional applications (USSN 61/652,086, filed May 25, 2012 (P1) and USSN 61/716,256, filed October 19, 2012 (P2)) for failing to provide sufficient disclosure of eukaryotic embodiments of CRISPR. The brief sets forth with specificity the identities and efforts (failed according to Broad) by third parties to achieve ARTP of eukaryotic embodiments of CRISPR technology encompassed by the Count, specifically:
• Worms – Dr. Meyer, Howard Hughes Medical Institute Investigator and Professor of Cell and Developmental Biology at University of California and her student Te-Wen Lo
• Yeast – Dr. Jamie Cate, Professor of Biochemistry, Biophysics and Structural 1 Biology in Microbiology Professor of Cell and Developmental Biology at University of California
• Mice – Dr. Dirk Hockemeyer, Assistant Professor Department of Molecular & Cell Biology at University of California
• Plants – Drs. Chris and Shauna Somerville, Professors, Plant & Microbial Biology, at University of California
• Medaka fish – Dr. Kristin Teßmar-Raible, Professor and Group Leader Max Perutz Labs, at University of Vienna
• Zebrafish – Dr. Florian Raible, Professor and Group Leader Max Perutz Labs, at University of Vienna, and his post-doctoral researcher Dr. Stephanie Bannister
• Human – Dr. David Drubin, Department Co-Chair and Ernette Comby Chair in Microbiology Professor of Cell and Developmental Biology at University of California
The status of these efforts as failures is important to Broad's argument, because complete conception sufficient to support a claim to priority of invention in an interference requires that "the idea is so clearly defined in the inventor's mind that only ordinary skill would be necessary to reduce the invention to practice, without extensive research or experimentation," citing Dawson v. Dawson, 710 F.3d 1347, 1352 (Fed. 1 Cir. 2013), and "[t]he failures of these experts provide real-world evidence that the inventors' ideas were not 'so clearly defined' but rather required extensive research and experimentation." In addition to the inventors' own comments showing at best skepticism regarding ARTP of CRISPR in eukaryotic cells, the brief sets forth a litany of "confirmation of failure of CRISPR-Cas9 system" by contemporary statements of others:

In addition to these arguments, Broad contends that CVC cannot show possession of the invention defined by the Count because CVC's evidence does not show CRISPR-mediated DNA cleavage (i.e., functional CRISPR activity) in eukaryotic cells, as recited as an affirmative limitation in Count 1 of the Interference, citing Coleman v. Dines, 754 F.2d 353, 359 (Fed. Cir. 1985), for the requirement that "in establishing conception a party must show possession of every 1 feature recited in the count."
Turning to actual reduction to practice (ARTP), the brief then specifically sets out a timeframe for CVC's purported continued failure to achieve ARTP of CRISPR in eukaryotic cells, in support of Broad's argument that CVC's conception was flawed and incomplete (and thus that CVC does not deserve its March 1, 2012 conception date):

Of course, it inures to Broad's benefit that many of these statements concern and highlight precisely the impediments and obstacles Broad has argued here and in the '048 Interference would be relevant to adapting CRISPR to the eukaryotic context. In particular, Broad's brief asserts that CVC has not established CRISPR-mediated DNA cleavage in the zebrafish experiments performed August 9, 2012 nor in the human cell experiments performed on October 31 and November 1, 5, and 18, 2012. (Indeed, the brief characterizes some of CVC's evidence as "a litigation-inspired resurrection of a failed experiment that never saw the light of day in 2012 or any other time before this Interference," which insofar as it is true is due at least in part to Broad's success in obtaining a judgment of no interference-in-fact in the '048 Interference). The brief goes into great detail regarding these purported failures, again using contemporary statements in CVC's evidence to support its argument that the CVC inventors themselves did not recognize these experiments as having successfully demonstrated successful eukaryotic cell CRISPR. And to the extent there is evidence of DNA cleavage in the human cell experiments, Broad contends that by changing the cell lysis conditions CRISPR cleavage occurred in the lysate rather than in the cells themselves. The Broad also spends a good portion of its brief regarding the uncertainties in achieving successful practice of ZFN or TALEN systems or RNA-based genetic methods (including Group II introns, ribozymes and riboswitches) in eukaryotic cells as evidence of the expectations of the skilled worker regarding adapting CRISPR to the eukaryotic cell milieu. Interspersed with these arguments are references to Board decisions denying CVC's motions to be awarded priority benefit and decisions in the earlier '048 Interference.
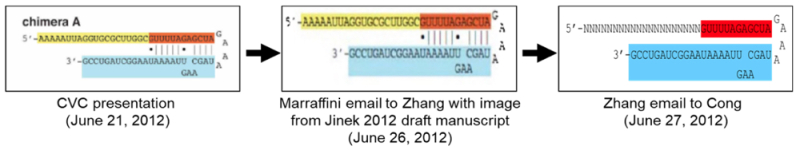
The brief also asserts an experimental distinction between the methods CVC has asserted in its priority papers and the methods used by Broad inventor Zhang that, according to Broad, resulted in successful CRISPR-mediated DNA cleavage in eukaryotic cells no later than July 31, 2012:
The chimera A design showcased by CVC and disclosed in Jinek 2012—a design with a 26 nucleotide tracr sequence—is not what results when expressed from a U6 promoter driven plasmid in eukaryotic cells. MF133; see Ex. 3424 ¶¶ 128, 181; Breaker 3rd Dec. ¶¶ 238-241. This is a key distinction. As Zhang explains in his declaration, when he saw the 26 nucleotide design, he immediately appreciated that this minimal tracrRNA "eliminated the two larger natural stem-loop structures from the tracrRNA segment" which he understood were important in a complex environment, like a eukaryotic cell, to "achieve sufficient loading onto Cas9 in a cell, particularly because the secondary structures (in the form of stem-loops) could be important for loading the RNA duplex onto Cas9, and important for forming an RNA-protein complex with Cas9." Ex. 3424, Zhang Dec. ¶¶17-18. Thus, Zhang deliberately chose a system "using a vector [with a U6 promoter] that would express chimeric RNA with a truncated tracrRNA segment of 30 nucleotides—four nucleotides longer than the chimeric RNA" in Jinek 2012. Id. ¶19. That is because Zhang understood that "[t]he addition of those four nucleotides could assist in protecting the nucleotides on the 3' end of the tracrRNA [which are important for loading/complexing] from RNA degradation by endogenous RNases present in the cell." Id. A point seemingly lost on the CVC inventors. Ex. 6207 at 192:9-201:6, 199:0-201:6.
Almost as an afterthought, the brief contends that CVC has not shown diligence from its earliest claimed conception date to ARTP, citing "gaps and a hodgepodge of efforts" in these efforts; this argument is blunted somewhat by the brief's efforts to show CVC's continued but failed efforts to reduced eukaryotic CRISPR to practice.
Science, particularly cutting-edge science, can be messy as it is being performed, and Broad takes advantage of this messiness to make its case that CVC had not satisfied the requirement for conception that CVC's inventors had a "definite and permanent idea of the complete and operative invention" at the claimed conception date. CVC will have its chance to reply to this motion. However, Broad has amassed an impressive array of documentary and contemporaneous witness statements in support of its arguments in this brief for CVC to rebut. CVC's reply brief is due on April 6, 2021.
 April 14, 2021 – "Protecting Software Related to a Medical Device: A Case Law Review & Strategy" (Intellectual Property Owners Association) – 2:00 pm to 3:00 pm (ET).
April 14, 2021 – "Protecting Software Related to a Medical Device: A Case Law Review & Strategy" (Intellectual Property Owners Association) – 2:00 pm to 3:00 pm (ET).