By Kevin E. Noonan —
 On November 19th, Senior Party Sigma-Aldrich filed its Substantive Preliminary Motion No. 1 in CRISPR Interference No. 106,133 (where the University of California, the University of Vienna, and Emmanuelle Charpentier, collectively "CVC" is Junior Party) asking the Board to substitute the Count pursuant to 37 C.F.R. §§ 41.121(a)(1)(iii) and 41.208(a)(1).
On November 19th, Senior Party Sigma-Aldrich filed its Substantive Preliminary Motion No. 1 in CRISPR Interference No. 106,133 (where the University of California, the University of Vienna, and Emmanuelle Charpentier, collectively "CVC" is Junior Party) asking the Board to substitute the Count pursuant to 37 C.F.R. §§ 41.121(a)(1)(iii) and 41.208(a)(1).
In its Statement of Precise Relief Requested, Sigma-Aldrich sets forth its proposed Count 2 as, in the alternative, CVC Application No. 15/947,680, claim 164 or Sigma-Aldrich Application No. 15/456,204, claim 31 (the latter alternative Count based on Sigma-Aldrich's claim remains the same as in the Count as the interference was declared). Claim 164 of CVC's '680 patent recites (dependent on claims 156 and 157):
156. A method of cleaving or editing a target DNA molecule or modulating transcription of at least one gene encoded thereon, the method comprising:
contacting a target DNA molecule having a target sequence with an engineered and/or non-naturally-occurring Type II Clustered Regularly Interspaced Short Palindromic Repeats (CRISPR)—CRISPR associated (Cas) (CRISPR-Cas) system comprising:
a) a single molecule DNA-targeting RNA comprising
i) a targeter-RNA that hybridizes with the target sequence, and
ii) an activator-RNA that hybridizes with the targeter-RNA to form a double-stranded RNA duplex of a protein-binding segment,
wherein the targeter-RNA and the activator-RNA are covalently linked to one another with intervening nucleotides; and
b) a Cas9 protein,
wherein the single molecule DNA-targeting RNA forms a complex with the Cas9 protein, thereby targeting the Cas9 protein to the target DNA molecule,
whereby said target DNA molecule is cleaved or edited or transcription of at least on gene encoded by the target DNA molecule is modulated, and
wherein said contacting occurs in a eukaryotic cell.
157. The method of Claim 156, wherein, prior to the contacting step, the method comprises:
introducing into the eukaryotic cell containing the target DNA molecule:
1) the single molecule DNA-targeting RNA, or a DNA molecule comprising a nucleotide sequence that
(i) encodes the single molecule DNA-targeting RNA and
(ii) is operably linked to a regulatory element operable in said eukaryotic cell; and
2) the Cas9 protein, an RNA molecule comprising a nucleotide sequence encoding the Cas9 protein, or a DNA molecule comprising a nucleotide sequence that
(i) encodes the Cas9 protein and
(ii) is operably linked to a regulatory element operable in said eukaryotic cell.
164. The method of Claim 157, wherein the method comprises creation of a double strand break in the target DNA molecule which is repaired by a homology-directed repair mechanism which incorporates a sequence of a donor polynucleotide into the target DNA molecule, thereby editing the target DNA molecule (emphasis in brief).
Sigma-Aldrich's argument in favor of substituting the Count is that the so-called McKelvey count as declared encompasses two patentably distinct inventions: "(1) CRISPR-Cas9 in a eukaryotic cell to cleave a target DNA; and (2) CRISPR-Cas9 in a eukaryotic cell to cleave a target DNA and subsequently to integrate a donor DNA sequence into the target DNA via homology-directed repair ("HDR)" (emphasis in brief). According to Sigma-Aldrich, this second, subsequent step is not obvious over merely cleaving a DNA target and thus claims to these embodiments are patentably distinct.
Citing 37 C.F.R. § 41.201, Sigma-Aldrich maintains that an interference count should not encompass two patentably distinct inventions. This principle is supported by Judge McKelvey's and subsequent Board decisions, citing Ashurst v. Brugger, Int'f No. 105,482 (McK), 2008 Pat. App. LEXIS 5953, *51-52 (BPAI July 16, 2008), and Edelman v. Stomp, Int'f No. 105,261 (SGL), 2006 Pat. App. LEXIS 17, *18-19 (PTAB Apr. 14, 2006). CVC's portion of the Count as declared encompasses "cleaving or editing the target DNA molecule or modulating transcription of at least one gene encoded by the target DNA molecule" whereas Sigma-Aldrich's portion of the Count as declared is expressly limited to "modifying a chromosomal sequence in a eukaryotic cell by integrating a donor sequence" (emphasis in brief). (And, facing inventors who were awarded the Nobel Prize for their achievements it is perhaps understandable that Sigma-Aldrich characterizes "its" portion of the Count as "this final integration step represents a milestone accomplishment in CRISPR-Cas9 genome engineering—not simply cleaving a DNA strand, but thereafter actually modifying the chromosomal sequence by integrating into that cleaved strand a donor polynucleotide by HDR.") And importantly for arguments set forth later in the brief (and the non-obviousness argument supporting patentable distinctiveness), Sigma-Aldrich states that at the time of its first filing date, December 6, 2012, "while . . . a POSITA would have hoped that this advance in genome engineering could be accomplished, whether such an achievement would be accomplished remained speculative."
The brief then explicates what it recited in its portion of the Count as declared and makes its argument that "current Count 1 permits CVC to submit proofs of invention for cleavage alone [based on its claims encompassing "cleaving or editing the target DNA molecule or modulating transcription of at least one gene encoded by the target DNA molecule"], which do not constitute proofs of invention for the separately patentable cleavage plus integration invention." Permitting CVC to prevail on this basis would be contrary to the "fundamental purposes of a first-to-invent patent interference" as well as being "manifestly unfair to Sigma," the brief asserts because it would deprive Sigma-Aldrich of patent protection for "its distinct and more technically challenging invention based on CVC's proofs for a different and considerably simpler invention." In addition, Sigma-Aldrich asserts that none of CVC's applications disclose embodiments for cleavage + integration methods (even if their claims encompass such CRISPR methods in eukaryotic cells). "CVC's evidence in CVC P3 that CRISPR-Cas9 could cleave a DNA strand in a eukaryotic cell — full stop — should not entitle CVC to an invention that CVC did not demonstrate that it actually possessed, namely, preparing a donor polynucleotide, introducing that donor polynucleotide into the cell, and actually modifying the chromosomal sequence by integrating that donor polynucleotide into the cleaved DNA strand by HDR," Sigma-Aldrich argues. CVC disclosure of potential uses of CRISPR for integrating DNA into a targeted site should not entitle CVC to claims for such embodiments that they did not possess (particularly under circumstances where Sigma-Aldrich achieved such inventive embodiments first, Sigma-Aldrich argues). And the brief notes that there are three other interferences before the Board, none of which involve a priority dispute as to which party showed the "further technological advance of integrating a donor polynucleotide into the cleaved target DNA via HDR."
In the remainder of the brief Sigma-Aldrich sets forth the distinctions between "cleavage" embodiments of CRISPR in eukaryotic cells and "cleavage + integration" methods. These include that "DNA integration via HDR in eukaryotes was known to be a complex and minimally understood biological process that required significant experimental effort" precluding the skilled worker from having a reasonable expectation of success in achieving it. Sigma-Aldrich supports these assertions with quotations from contemporary statements by CVC inventors in publications such as Jinek et al. (2012, "A Programmable Dual-RNA–Guided DNA Endonuclease in Adaptive Bacterial Immunity," Science 337(6096): 816-21) emphasizing characterizations such as "possibility" and "potential" which Sigma-Aldrich maintains illustrate what was holed might be possible. Sigma-Aldrich contends that in light of these statements a "POSITA, understanding the complexity and uncertainty of HDR repair in eukaryotic cells, would have been concerned whether integrating donor DNA into a target chromosome using a CRISPR-Cas9 system would be successful, and would not have had a reasonable expectation of success in doing so." In contrast, in the same timeframe Sigma-Aldrich argues that its P1 provisional application, filed December 6, 2012, actually demonstrated use of CRISPR in eukaryotic cells to achieve "cleavage + integration" results. In an argument reminiscent of Broad's success in Interference No. 106,048 in convincing the Board (and Federal Circuit) that eukaryotic cells are sufficiently complex that the skilled worker would not have had a reasonable expectation of success in achieving targeted DNA cleavage in eukaryotic cells, Sigma-Aldrich asserts considerations including:
(1) in eukaryotic cells, the available and competing DNA repair pathways for [double-stranded breaks] DSBs are highly complex and not well understood;
(2) DNA repair in higher eukaryotic cells especially favors the competing nonhomologous end joining ("NHEJ") repair pathway over HDR;
(3) the window for successful HDR is limited to only certain stages of the cell cycle (late S and G2), while the competing NHEJ repair pathway is always available;
(4) "error-prone" repair of a DNA break by the preferred NHEJ pathway creates indels that destroy the DNA target sequence and thereby halt successive rounds of DNA cleavage and further attempts at HDR integration;
(5) prior experience using nuclease-generated DNA breaks for genome editing purposes (e.g., ZFNs and TALENs) had consistently shown significantly greater frequencies of NHEJ-mediated repair outcomes than HDR-mediated outcomes in higher eukaryotes[;]
(6) HDR in eukaryotes is an extremely complicated and highly regulated DNA repair process that involves a cascade of events initiating at the ends of the DNA break, and that process could be disrupted by the introduction of the barely understood prokaryotic CRISPR-Cas9 system into eukaryotic cells;
(7) in particular, inherent characteristics of the chromatin environment of a cleaved DNA site could limit its ability to support HDR integration, and CRISPR-Cas9 could adversely influence the chromatin environment and/or the regulatory proteins that were known to play essential roles in regulating DNA repair pathway choice;
(8) cleavage by CRISPR-Cas9 was understood to create blunt ends (i.e., without overhangs) on the cleaved DNA strands, which are distinct from the staggered overhangs present at the DSBs generated by other nuclease cleavage systems that had been shown to be compatible with HDR-mediated integration (e.g., ZFNs, TALENs, and SceI meganuclease), creating concerns that such different DNA ends might not be compatible with HDR-mediated donor integration for genome editing purposes;
(9) the Cas9 protein was known to remain bound to the cleaved DNA ends, which could physically interfere with DNA repair in general, and more specifically with the selection of the HDR pathway which requires the initial steps of forming a specific protein complex at the DNA ends and thereafter resecting the DNA ends;
(10) it was unknown whether CRISPR-Cas9 could create cleavage at the target site in a eukaryotic genome with enough specificity that would allow a site-specific HDR-mediated integration application;
(11) unpredictable off-target effects (e.g., multiple DNA breaks at untargeted loci) would be expected to occur with CRISPR-Cas9 as they did in other nuclease cleavage systems (e.g., ZFNs and TALENs), and perhaps at higher levels, and those off-target effects could arrest the cell and prevent its entry into the specific stages of the cell cycle where HDR is possible (late S/G2); and
(12) it would be expected that Cas9, a large prokaryotic enzyme, would not be able to access the eukaryotic nucleus outside of the stage in the cell cycle where the nuclear membrane is dissolved, thereby imposing a potential temporal restriction on Cas9's access to the nucleus and preventing the creation and persistence of an unrepaired (by NHEJ) DNA break until the stages of the cell cycle that could support HDR-mediated integration.
Followed by a detailed consideration of these potential effects and the state of the art as of December 2012, supported by citations to myriad exhibits illustrating the art (an important assertion being that in eukaryotic cells the NHEJ repair mechanism was "far more common[ly]" used than the HDR mechanism Sigma-Aldrich's CRISPR-mediated] "cleavage + integration" mechanism.
Sigma-Aldrich also addresses an expected counterargument that HDR-mediated integration of heterologous DNA occurs in ZFN and TALEN systems, arguing that these characteristics of very different DNA editing methods would not have engendered a reasonable expectation of success in the skilled worker regarding CRISPR-mediated cleavage. These arguments are supported by evidence from the Board's determination of no interference-in-fact in the '048 Interference related to expectations arising from ZFN and TALEN prior art with regard to eukaryotic CRISPR. And Sigma-Aldrich asserts, "[i]ntegrating a donor polynucleotide into a target site of a cleaved DNA in a eukaryotic cell by HDR-mediated integration was an additional challenge to overcome even when mechanisms to create site-specific DNA breaks had been developed." The brief sets forth the extended history of using ZFN and TALEN for integrating DNA at target sites, and notes that Doudna herself did not publish "using CRISPR-Cas9 for successful HDR integration in a eukaryotic cell until more than two years after Jinek."
Having made its case for the patentable distinctions between cleavage by CRISPR and CRISPR-mediated cleavage and HDR-mediated integration at a target site, Sigma-Aldrich then sets out the basis for their assertion that all their own claims-in-interference correspond to Proposed Substitute Count 2, based in part on the Sigma-Aldrich portion of the Count as declared. For CVC's claims, the brief argues that "many, but not all, of CVC's involved claims (a total of 412 according to the brief) correspond to Proposed Substitute Count 2. Which claims should remain in this interference is set forth in the following tables:

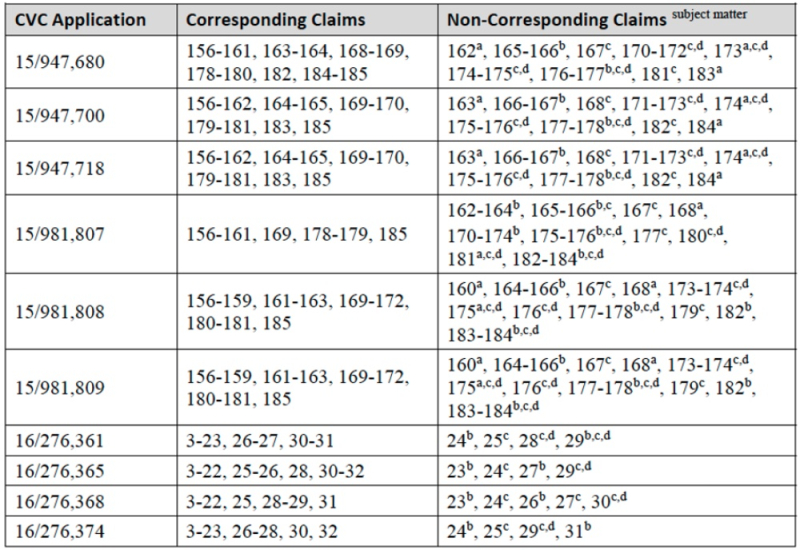
Where a-e designations correspond to the following characteristics for claims not corresponding to Proposed Substitute Count 2:
(a) > 1 targeting RNA (aka "multiplexing");
(b) a Cas9 protein that includes a Protein Transduction Domain (PTD);
(c) one or more mutation(s) in the Cas9 RuvC/HNH domain(s);
(d) a nickase for a creating a "nick" or a single stranded break in the target DNA; and
(e) a chimeric Cas9 protein.
Finally, with regard to four pending CVC applications directed to "chimeric and mutated Cas9 protein," Sigma-Aldrich proposes these claims and applications be designated as not corresponding to Proposed Substitute Count 2:

The brief concludes with Sigma-Aldrich's arguments regarding entitlement to their earliest P1 provisional application and all intervening applications, as set forth in this diagram:

Specifically relied upon for priority entitlement are Examples 4 and 5 of the '256 provisional application as illustrated (albeit somewhat faintly) in Figure 5:

And perhaps cautioned by arguments raised in earlier interferences, Sigma-Aldrich sets forth the priority relationships between its applications with regard to the effects on entitlement to interference practice by enactment of the Leahy-Smith America Invents Act:
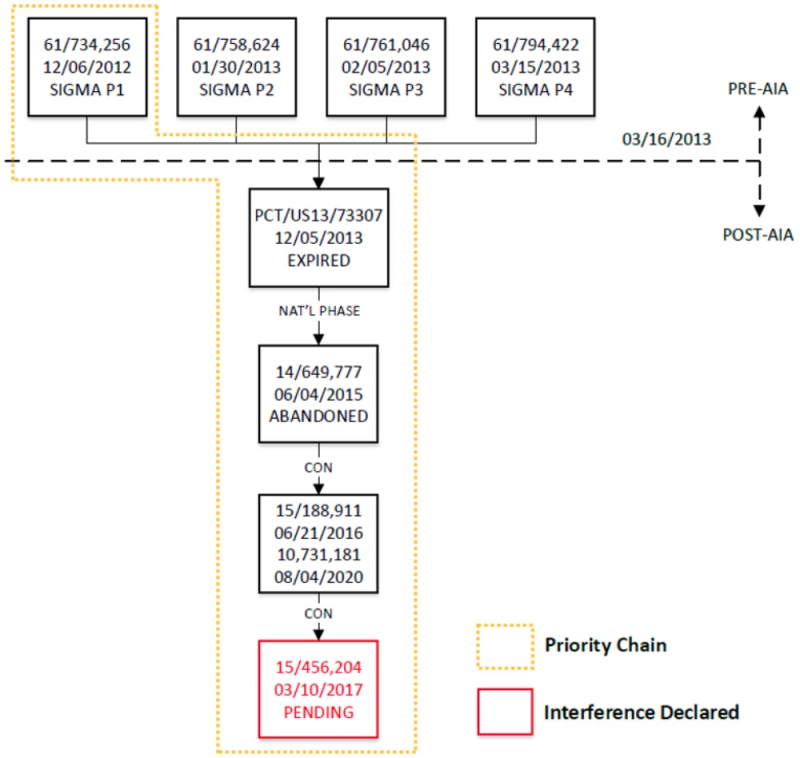
The brief then sets forth Sigma-Aldrich's arguments regarding why CVC is not entitled to priority benefit of its January 28, 2013 provisional application filing ("P3") or Provisional Application No. 61/765,576 (filed Feb. 15, 2013) ("P4") which, while containing dependent claims that encompass "cleavage + integration" embodiments of eukaryotic CRISPR, do not contain any disclosure showing that CVC's inventors possessed them.
Finally, Sigma-Aldrich closes the brief with its bases for asserting that Proposed Substitute Count 2 is patentable over the prior art, based on arguments similar to the reasoning regarding the patentable distinctions between CVC's invention and the Sigma-Aldrich claims-in-interference discussed above including the Board's determination of the limits of the reasonable expectations for success set forth in the '048 Interference.
![]() The Federal Circuit Bar Association (FCBA) Rules Committee will be offering a remote program entitled "A Conversation with the Federal Circuit Clerk's Office" on February 16, 2022 from 2:00 pm to 3:00 pm (ET). Erik R. Puknys of Finnegan, Henderson, Farabow, Garrett & Dunner, LLP will moderate a panel consisting of Peter Marksteiner, Circuit Executive and Clerk of Court, U.S. Court of Appeals for the Federal Circuit; and Jarrett Perlow, Chief Deputy Clerk, U.S. Court of Appeals for the Federal Circuit. The webinar will provide an introduction to the role of the Clerk's Office, an overview of the Federal Circuit Rules of Practice, and practical advice for complying with the Rules and interacting with the Clerk's Office.
The Federal Circuit Bar Association (FCBA) Rules Committee will be offering a remote program entitled "A Conversation with the Federal Circuit Clerk's Office" on February 16, 2022 from 2:00 pm to 3:00 pm (ET). Erik R. Puknys of Finnegan, Henderson, Farabow, Garrett & Dunner, LLP will moderate a panel consisting of Peter Marksteiner, Circuit Executive and Clerk of Court, U.S. Court of Appeals for the Federal Circuit; and Jarrett Perlow, Chief Deputy Clerk, U.S. Court of Appeals for the Federal Circuit. The webinar will provide an introduction to the role of the Clerk's Office, an overview of the Federal Circuit Rules of Practice, and practical advice for complying with the Rules and interacting with the Clerk's Office.


















