By Kevin E. Noonan —
 It is apparent that the facts are often
It is apparent that the facts are often
at risk or ignored by opponents of gene patenting. Indeed, the various forms of fact-twisting or outright
falsehood ("corporations own your genes") suggests that it isn't
facts that are at issue but opinions, and how the public's opinion on gene
patenting is to be best manipulated to oppose the practice.
One example (of the very many) of this
phenomenon came to us in a reader comment to our post, "Huffington Post Provides Latest Ant-gene Patenting Soapbox" last week. Our commenter
writes:
While the errors
throughout her "editorial" are too numerous to recount, her
discussion of African sleeping sickness deserves special recognition. She writes "In 1990 Sanofi-Aventis
developed the drug Ornidyl (eflornithine) which is safe and effective against
trypanosomiasis but it was withdrawn from the market in 1995 because few poor
subSaharan sufferers could afford it. Ornidyl can no longer be bought for love or money but Aventis, and
later, Bristol-Myers Squibb, began to market eflornithine to the West as
Vaniqa, a prescription-only facial depilatory."
First, Orindyl for
sleeping sickness she mentions above is eflornithine via intravenous injection,
while the eflornithine currently marketed as Vaniqua is formulated as a topical
creme. This is a major difference
she conveniently overlooks. And
much more importantly, her claim that the drug is unavailable is completely
false. "In 2001, Aventis (now Sanofi-Aventis) and the WHO formed a five-year
partnership, during which more than 320,000 vials of pentamidine, over 420,000
vials of melarsoprol, and over 200,000 bottles of eflornithine were produced by
Sanofi-Aventis, to be given to the WHO and distributed by the association
Médecins sans Frontières (also known as Doctors Without Borders) in countries
where the sleeping sickness is endemic … From 2001 (when production was
restarted) through 2006, 14 million diagnoses were made. This greatly
contributed to stemming the spread of sleeping sickness, and to saving nearly
110,000 lives. This changed the epidemiological profile of the disease, meaning
that eliminating it altogether can now be envisaged.[9] . . . The positive results
of the 2001-2006 partnership between Sanofi-Aventis and the WHO in the fight
against sleeping sickness motivated and justified the decision taken by the
Sanofi-Aventis Group's senior management to continue supporting the WHO at the
same level for another five years, 2006-2011," citing "IFPMA Health Initiatives:
Sleeping Sickness."
I guess I must concede
that Ms. Washington's point may actually have been accurate, as eflornithine
really is not avialable for "love or money" since Sanofi, WHO and MSF
have collaborated to make it available for free. All this information is available at Wikipedia or the Sanofi
website http://en.sanofi-aventis.com/binaries/brochure_aam_en_tcm28-18133.pdf.
(Quoted citations from Wikipedia, see, http://en.wikipedia.org/wiki/Eflornithine).
In addition, our commenter states
Ms. Washington "is wrong that there is only [one] drug for HCV, with a 20%
cure rate (she is off on both points)." Again quoting from Wikipedia (with references to supporting
scientific journal articles; http://en.wikipedia.org/wiki/Hepatitis_C#Treatment):
Current
treatment is a combination of Pegylated interferon-alpha-2a
or Pegylated interferon-alpha-2b
(brand names Pegasys or PEG-Intron) and the antiviral drug ribavirin for a period of 24 or 48 weeks, depending on
hepatitis C virus genotype.
Pegylated interferon-alpha-2a
plus ribavirin may increase
sustained virological response among patients with chronic hepatitis C as
compared to pegylated interferon-alpha-2b
plus ribavirin according to
a systematic review of randomized controlled trials. (Awad et al., (2009). "Peginterferon alpha-2a is associated with
higher sustained virological response than peginterferon alfa-2b in chronic
hepatitis C: Systematic review of randomized trials.". Hepatology
(Baltimore, Md.) 51 (4): 1176–84. doi:20187106. PMID 20187106). The relative benefit increase was 14.6%. For patients at similar risk to those in this study (41.0% had sustained
virological response when not treated with pegylated interferon alpha 2a plus
ribavirin), this leads to an absolute benefit increase of 6%. 16.7 patients
must be treated for one to benefit ( number needed to treat = 16.7. click here to adjust
these results for patients at higher or lower risk of sustained virological
response). However, this study's results may be biased due to uncertain
temporality of association, selective dose response.
Treatment is
generally recommended for patients with proven hepatitis C virus infection and
persistently abnormal liver function tests.
Treatment during the
acute infection phase has much higher success rates (greater than 90%) with a
shorter duration of treatment; however, this must be balanced against the
15-40% chance of spontaneous clearance without treatment (see Acute Hepatitis C
section above).
Those with low
initial viral loads respond much better to treatment than those with higher
viral loads (greater than 400,000 IU/mL). Current combination therapy is
usually supervised by physicians in the fields of gastroenterology, hepatology or infectious
disease.
The treatment may be
physically demanding, particularly for those with a prior history of drug or
alcohol abuse. It can qualify for temporary disability in some cases. A substantial proportion of
patients will experience a panoply of side effects ranging from a 'flu-like'
syndrome (the most common, experienced for a few days after the weekly
injection of interferon) to severe adverse events including anemia, cardiovascular events and
psychiatric problems such as suicide or suicidal ideation. The latter are
exacerbated by the general physiological stress experienced by the patient.
Responses can vary
by genotype. (Approximately 80% of hepatitis C patients in the United States
have genotype 1. Genotype 4 is more common in the Middle East and Africa.)
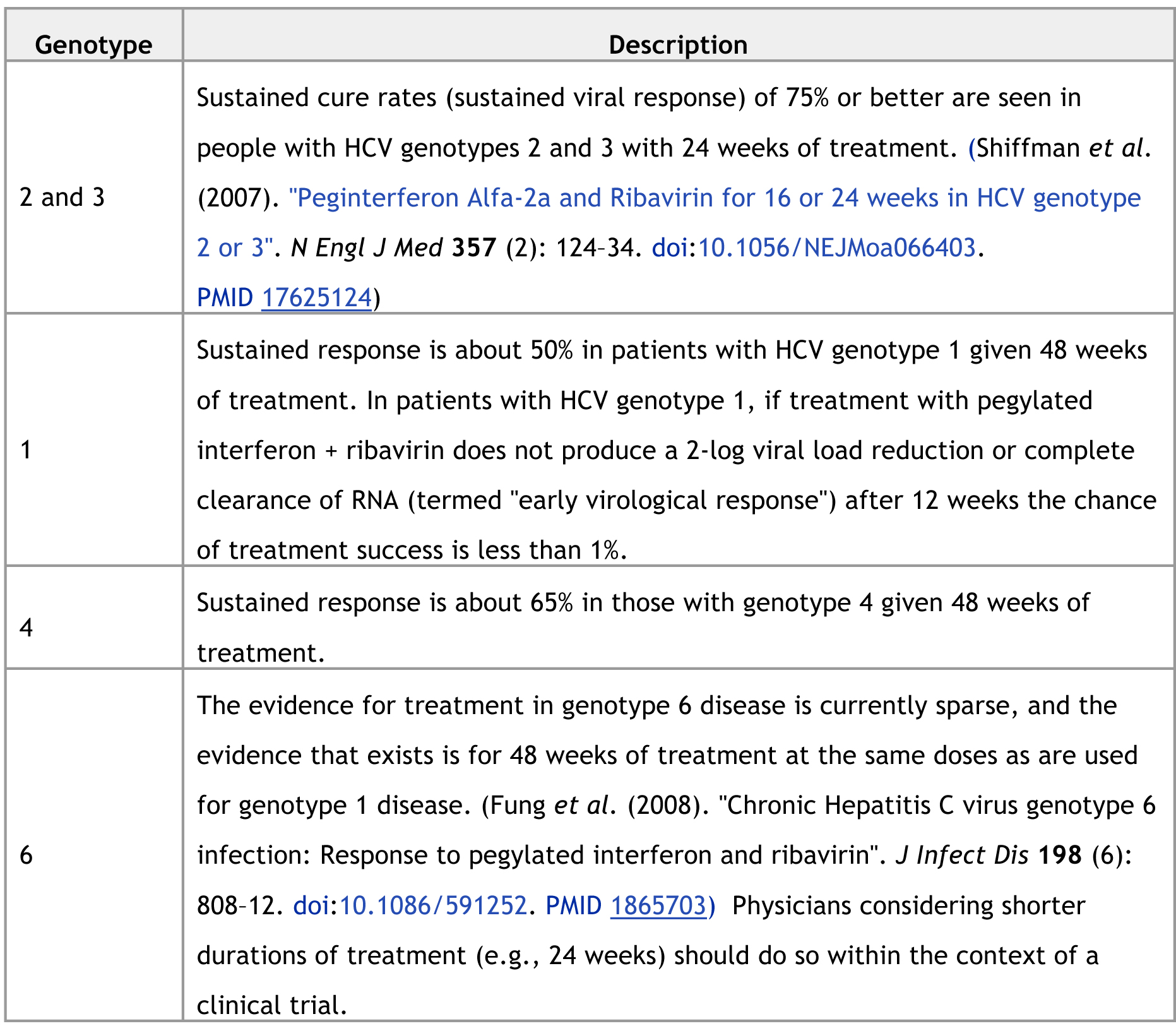
Early virological
response is typically not tested in non-genotype 1 patients, as the chances of
attaining it are greater than 90%. The mechanism of cure is not entirely clear,
because even patients who appear to have a sustained virological response still
have actively replicating virus in their liver and peripheral blood mononuclear
cells. (Castillo et al., (2006). "Hepatitis C virus
replicates in the liver of patients who have a sustained response to antiviral
treatment". Clin Infect Dis 43 (10): 1277–83. doi:10.1086/508198. PMID 17051492)
In the face of such consistent disinformation, it
can be difficult to decide the most effective course for combating it. However, some suggestions come to mine:
 First, don't
First, don't
challenge the morality issue component, since it is generally hard if not
impossible to change deeply-held moral beliefs (no matter how out of touch with
reality).
Second,
however, do challenge the flawed logic. For example, using claims made in Ms. Washington's article, just because
the National Health Service in Britain or a private insurance company in the
U.S. won't pay for a test or a treatment doesn't mean the test is too
expensive. It means the payor has
decided it can wait until the cost of the test goes down when the patent
expires, and the patients denied the test be damned — the bottom line is more
important.
Third, while
not necessarily convincing those whose minds are closed to information (aka,
the facts), it is also good to use actual data, such as the limited numbers of
patents granted to U.S. universities before enactment of the Bayh-Dole Act and
the veritable flood of university patents thereafter. The point is that the amount of basic research isn't what's
different (after all, President Nixon started the "War on Cancer" in
1971 with increased funding for the NIH and NCI), but that universities now
have ways to prevent predation from private businesses that would otherwise
turn publicly-funded research into private investment without many of the
advantages of the Bayh-Dole Act, including preference for U.S.-based licensees
(i.e., jobs); royalty revenue to the universities to be used for teaching and
further research; "march-in" rights in a case of a private licensee
enacting a public harm by failing to commercialize an important invention (such
as a cure or treatment of a disease, something that has never happened in the
30 years since the Act was passed); and the many milestones and other positive
incentives on the patent right incorporated into licenses from universities
because of the university's unique public service mission. These facts turn the anti-corporate
argument on its head, since by being able to protect its intellectual
property, universities have real leverage for deciding how and by whom their
technology is commercialize. Coupled with the fact that, prior to Bayh-Dole,
many of the predating companies were overseas or large international
conglomerates, and that such technology piracy cost American jobs, there is a
good argument that university patenting is good for society.
Finally, it is
good to paint the picture of what would happen if these inventions could not be
protected. As has been argued here in the past, the patent system promotes
disclosure and the advancement of the useful arts, i.e., technology. Patents require disclosure to a degree
not required by scientific journal articles and other sources, with "a written description of
the invention, and of the manner and process of making and using it, in such
full, clear, concise, and exact terms as to enable any person skilled in the
art to which it pertains, or with which it is most nearly connected, to make
and use the same, and shall set forth the best mode contemplated by the
inventor of carrying out his invention." 35 U.S.C. §112, first paragraph. Since patents expire, this information can be used by anyone
thereafter, and importantly, can be used as
information even during the lifetime of the patent. Thus, arguments that basic (as opposed
to clinical for profit) genetic research is impeded by patenting is wrong,
since the information (whether of a genetic sequence or the existence of a
mutation) is not patented. The public availability of this
information, and the requirements as to its completeness, fulfill the
Constitutional mandate that patenting "promote the progress" of the
useful arts.
The
alternative would be the antithesis of this reality, where there would be
significant incentive not to disclose
genetic and other information. In
situations where an invention is easy to "reverse engineer" (as for many
mechanical inventions), this would not be a fruitful path to protect intellectual
property. For something as
complicated as human genetics, the possibilities for being able to "hide"
basic discoveries are greater and the consequences much more dire. Ironically, universities would be the
victims of this alternative, since the university's mission would not permit it
to use trade secrets and other forms of undisclosed intellectual property
protection. The predictable result
of this alternative would be to inhibit if not destroy the university-company
partnership that has promoted the biotechnology industry for a generation, to
society's benefit. Perhaps new
diagnostics and treatments would arise "eventually" under such an
alternative reality, but not as rapidly as in the past. Thus, the counter-argument for the "anti" groups is that they are
(in philosophy or as a consequence of their vision of morality) anti-science,
anti-progress, anti-better diagnostics, and anti-treatment. If this argument is effectively made, it's hard to see how the public would
prefer the "eventually" world their position would produce.
 Last
Last
 In
In

 In
In


















