By Donald Zuhn —
 In Daiichi Sankyo Co. v. Matrix Laboratories, Ltd., decided last Thursday, the Federal Circuit affirmed a determination by the District Court for the District of New Jersey that Matrix Laboratories, Ltd., Mylan Inc., Mylan Laboratories, Inc., and Mylan Pharmaceuticals, Inc. ("Mylan") failed to establish a prima facie case of obviousness with respect to claim 13 of U.S. Patent No. 5,616,599. Daiichi Sankyo Company, Ltd. and Daiichi Sankyo, Inc. own the '599 patent, which relates to 1-biphenylmethylimidazole compounds and their use as angiotensin receptor blockers (ARBs) for the treatment of high blood pressure. Claim 13 of the '599 patent encompasses olmesartan medoxomil, an ARB that is marketed by Daiichi as the active ingredient in Benicar®, Benicar HCT®, and Azor®.
In Daiichi Sankyo Co. v. Matrix Laboratories, Ltd., decided last Thursday, the Federal Circuit affirmed a determination by the District Court for the District of New Jersey that Matrix Laboratories, Ltd., Mylan Inc., Mylan Laboratories, Inc., and Mylan Pharmaceuticals, Inc. ("Mylan") failed to establish a prima facie case of obviousness with respect to claim 13 of U.S. Patent No. 5,616,599. Daiichi Sankyo Company, Ltd. and Daiichi Sankyo, Inc. own the '599 patent, which relates to 1-biphenylmethylimidazole compounds and their use as angiotensin receptor blockers (ARBs) for the treatment of high blood pressure. Claim 13 of the '599 patent encompasses olmesartan medoxomil, an ARB that is marketed by Daiichi as the active ingredient in Benicar®, Benicar HCT®, and Azor®.
The use of ARBs to control blood pressure can be traced back to work done in the 1970s and 1980s by Takeda Pharmaceutical Co. Ltd., which developed a class of compounds comprising an imidazole ring with other chemical moieties bonded to the five positions of the ring. One such compound, Takeda's S-8307, has the chemical structure:
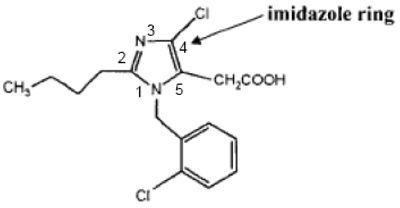
Using Takeda's compounds as leads, E. I. du Pont de Nemours and Company sought to develop compounds having inreased angiotensin receptor binding, and therefore, better ARB activity. DuPont modified S-8307 to develop the compound losartan, which has ten-fold higher binding than Takeda's compounds. DuPont's losartan has the chemical structure:
DuPont disclosed losartan in U.S. Patent No. 5,138,069 along with more than 400 structurally related ARBs, including Example 118, which has the chemical structure:

Based on DuPont's success with losartan, a number of pharmaceutical companies, including Daiichi, initiated efforts to identify even better ARBs. Daiichi's work led to the synthesis of olmesartan, the active metabolite of olmesartan medoxomil, which differs from losartan in that it has a hydrophilic, hydroxy-isopropyl group at the 4-position of the imidazole ring instead of a lipophilic, chlorine atom (like Example 118 above), and a carboxy group masked by a medoxomil prodrug substituent at the 5-position of the ring instead of a hydroxymethyl group. Olmesartan medoxomil and olmesartan have the chemical structures:

The closest prior art structure to Daiichi's olmesartan is DuPont's Example 6, disclosed in U.S. Patent No. 5,137,902, which differs from olmesartan in that it lacks a single oxygen atom at the 4-position of the imidazole ring. DuPont's Example 6 has the chemical structure (wherein the circled hydrogen atom in Example 6 is an -OH in olemsartan):

 Seeking approval to market generic olmesartan medoxomil, Mylan filed multiple ANDAs with the FDA. In response to Mylan's ANDA filings, Daiichi brought suit against Mylan for infringement of claim 13 of the '599 patent. The parties stipulated to infringement, leaving Mylan's counterclaim that claim 13 would have been obvious in light of: (1) the ARBs disclosed in DuPont's '902 patent, (2) Example 118 in DuPont's '069 patent, and (3) the well-known use of medoxomil as a prodrug. In particular, Mylan contended that one of skill in the art would have been motivated to select the ARBs disclosed in DuPont's '902 patent as lead compounds, and then modify the lipophilic alkyl groups at the 4-position of those compounds with olmesartan's hydrophilic hydroxyalkyl group in view of Example 118.
Seeking approval to market generic olmesartan medoxomil, Mylan filed multiple ANDAs with the FDA. In response to Mylan's ANDA filings, Daiichi brought suit against Mylan for infringement of claim 13 of the '599 patent. The parties stipulated to infringement, leaving Mylan's counterclaim that claim 13 would have been obvious in light of: (1) the ARBs disclosed in DuPont's '902 patent, (2) Example 118 in DuPont's '069 patent, and (3) the well-known use of medoxomil as a prodrug. In particular, Mylan contended that one of skill in the art would have been motivated to select the ARBs disclosed in DuPont's '902 patent as lead compounds, and then modify the lipophilic alkyl groups at the 4-position of those compounds with olmesartan's hydrophilic hydroxyalkyl group in view of Example 118.
 Following a bench trial, the District Court held that claim 13 of the '599 patent was not invalid as obvious, finding that Mylan had failed to show by clear and convincing evidence that a skilled artisan would have chosen the ARBs disclosed in DuPont's '902 patent as lead compounds, that the structure of the '902 patent compounds differed significantly from olmesartan medoxomil, and that Mylan had failed to prove that a skilled artisan would have been motivated to modify the 4- and 5-positions of the '902 patent compounds to obtain olmesartan medoxomil. In particular, the District Court determined that the '069 patent and its ARBs taught away from the use of a hydrophilic group at the 4-position and from any expectation that the use of a hydrophilic group would generate an ARB with significantly improved biological properties, and further, that converting olmesartan into a prodrug was a disfavored and unpredictable approach and that medoxomil was a disfavored prodrug. The District Court also found that secondary considerations favored a finding of nonobviousness; specifically that olmesartan medoxomil's enhanced potency constituted evidence of unexpected results and that Daiichi's Benicar® enjoyed commercial success despite being the seventh ARB on the market.
Following a bench trial, the District Court held that claim 13 of the '599 patent was not invalid as obvious, finding that Mylan had failed to show by clear and convincing evidence that a skilled artisan would have chosen the ARBs disclosed in DuPont's '902 patent as lead compounds, that the structure of the '902 patent compounds differed significantly from olmesartan medoxomil, and that Mylan had failed to prove that a skilled artisan would have been motivated to modify the 4- and 5-positions of the '902 patent compounds to obtain olmesartan medoxomil. In particular, the District Court determined that the '069 patent and its ARBs taught away from the use of a hydrophilic group at the 4-position and from any expectation that the use of a hydrophilic group would generate an ARB with significantly improved biological properties, and further, that converting olmesartan into a prodrug was a disfavored and unpredictable approach and that medoxomil was a disfavored prodrug. The District Court also found that secondary considerations favored a finding of nonobviousness; specifically that olmesartan medoxomil's enhanced potency constituted evidence of unexpected results and that Daiichi's Benicar® enjoyed commercial success despite being the seventh ARB on the market.
 In affirming the District Court's finding of nonobviousness, the Federal Circuit agreed with Daiichi in determining that Mylan failed to show (1) that one of ordinary skill in the art would have been motivated to select the ARBs disclosed in DuPont's '902 patent as lead compounds or (2) that the skilled artisan would have been motivated to modify the '902 patent compounds to synthesize olmesartan medoxomil. The Court began its analysis by citing Eisai Co. Ltd. v. Dr. Reddy's Labs., Ltd., 533 F.3d 1353 (Fed. Cir. 2008), and Takeda Chem. Indus., Ltd. v. Alphapharm Pty., Ltd., 492 F.3d 1350 (Fed. Cir. 2007), for the proposition that:
In affirming the District Court's finding of nonobviousness, the Federal Circuit agreed with Daiichi in determining that Mylan failed to show (1) that one of ordinary skill in the art would have been motivated to select the ARBs disclosed in DuPont's '902 patent as lead compounds or (2) that the skilled artisan would have been motivated to modify the '902 patent compounds to synthesize olmesartan medoxomil. The Court began its analysis by citing Eisai Co. Ltd. v. Dr. Reddy's Labs., Ltd., 533 F.3d 1353 (Fed. Cir. 2008), and Takeda Chem. Indus., Ltd. v. Alphapharm Pty., Ltd., 492 F.3d 1350 (Fed. Cir. 2007), for the proposition that:
Proof of obviousness based on structural similarity [between claimed and prior art compounds] requires clear and convincing evidence that a medicinal chemist of ordinary skill would have been motivated to select and then to modify a prior art compound (e.g., a lead compound) to arrive at a claimed compound with a reasonable expectation that the new compound would have similar or improved properties compared with the old.
With respect to the issue of lead compound selection, the panel countered Mylan's argument that because the '902 patent compounds are the closest prior art, this should have been dispositive of the lead compound issue, pointing out that such argument "runs contrary to our case law." The Court noted that "[i]n Takeda, we upheld a district court's finding that one of skill in the art would not have chosen the structurally closest prior art compound, compound b, as the lead compound in light of other compounds with more favor-able characteristics," adding that the Court's cases "illustrate that it is the possession of promising useful properties in a lead compound that motivates a chemist to make structurally similar compounds." Citing Ortho-McNeil Pharm., Inc. v. Mylan Labs., Inc., 520 F.3d 1358 (Fed. Cir. 2008), the Court explained that "attribution of a compound as a lead compound after the fact must avoid hindsight bias; it must look at the state of the art at the time the invention was made to find a motivation to select and then modify a lead compound to arrive at the claimed invention" (emphasis in original). Thus, the selection of a lead compound "depends on more than just structural similarity, but also knowledge in the art of the functional properties and limitations of the prior art compounds," and therefore, "[p]otent and promising activity in the prior art trumps mere structural relationships."
Turning to the issue of motivation to modify, the panel noted that the vast majority of compounds disclosed in the '069 patent contain a lipophilic group at the 4-position of the imidazole ring, and that only four (Examples 342, 329, 118, and 335) have a hydrophilic group like olmesartan medoxomil. According to the Court, "[t]he few compounds with hydrophilic groups at the 4-position are drowned out by the sea of 4-lipophilic compounds, which are the essence of what the ’069 patent teaches." In addition, the Court observed that binding affinity analyses comparing the '069 patent compounds that differed only at the 4-position confirm the preference for lipophilicity at that position. The panel determined that:
Altogether, the '069 patent's [structural-activity relationship] data and the structure of other second-generation ARBs counter any notion that one of skill in the art would have been motivated to modify the '902 compounds' lipophilic alkyl groups to a hydrophilic group. Such a holding would have been based on hindsight.
The panel therefore affirmed both the District Court's finding that Mylan had failed to establish that a skilled artisan would have selected the '902 patent ARBs as lead compounds and the lower court's finding that a skilled artisan would have modified the '902 patent ARBs at the 4-position of the imidazole ring to obtain olmesartan medoxomil. As a result, the Federal Circuit affirmed the District Court's determination that claim 13 of the '599 patent was not shown to be invalid as obvious.
Daiichi Sankyo Co. v. Matrix Laboratories, Ltd. (Fed. Cir. 2010)
Panel: Circuit Judges Lourie, Friedman, and Linn
Opinion by Circuit Judge Lourie























