By Donald Zuhn —
 Earlier this month, the World Intellectual Property Organization (WIPO) released a report on intellectual property trends in 2008 and 2009 showing that innovative activity and demand for IP rights dipped during the global economic crisis, but began to recover this year. Commenting on the report, WIPO Director General Francis Gurry stated that "[t]he post-crisis innovation landscape will invariably look different from that of a decade ago," adding that "[w]hile the strength of the recovery remains uncertain, there will likely be a continuing geographic shift of innovative activity toward new players, especially in Asia."
Earlier this month, the World Intellectual Property Organization (WIPO) released a report on intellectual property trends in 2008 and 2009 showing that innovative activity and demand for IP rights dipped during the global economic crisis, but began to recover this year. Commenting on the report, WIPO Director General Francis Gurry stated that "[t]he post-crisis innovation landscape will invariably look different from that of a decade ago," adding that "[w]hile the strength of the recovery remains uncertain, there will likely be a continuing geographic shift of innovative activity toward new players, especially in Asia."
According to the report, there were an estimated 1.91 million worldwide patent application filings in 2008, which represented a slowdown in the growth of such filings from 2007. While patent application filings grew by 2.6% in 2008, the report notes that this marked the lowest growth rate since the dot-com crisis in the early 2000's. The slowdown was a result of 0% growth in the U.S. and drops in Japan (-1.3%) and the Republic of Korea (-1.1%). Only an 18.2% increase in filings in China prevented the 2008 worldwide numbers from showing zero growth.
Preliminary data for 2009 showed a drop in patent application filings in many of the largest IP offices (see chart below; click on image to enlarge).
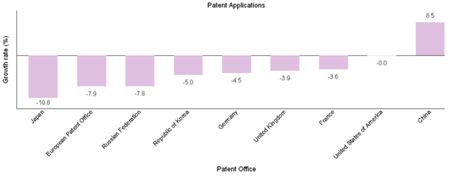
As in 2008, China was the exception in 2009, as that country once again showed substantial growth (+8.5%) in the face of economic adversity (see table below; click on image to enlarge).

With respect to patent grants, the report indicates that grants rose only 0.6% in 2008, with the Republic of Korea experiencing a 32.5% drop. According to the report, approximately 6.7 million worldwide patents were in force in 2008, with U.S. and Japanese residents owning about 48% of these patents.
The report notes that there are grounds for optimism moving forward as patent applications filed under the Patent Cooperation Treaty (PCT) have returned to growth this year. This return to growth followed a 4.5% decline in PCT filings in 2009, which the report indicated was due primarily to a drop in U.S. resident PCT applications (the U.S. accounted for three-fourths of the drop). In contrast, PCT filings originating from China, Japan, and the Republic of Korea increased in 2009.
In addition to analyzing patent application filings, the report also examined R&D expenditures in 2008-09, noting that companies started to reduce their R&D budgets from early 2009 onward. After showing a slowdown in R&D growth between 2007 and 2008, the report found a 1.7% decrease in R&D spending between 2008 and 2009. The report indicates that R&D spending varied from company to company, however, with some pharmaceutical companies showing substantial R&D growth over the two-year period.
Data regarding patent application filings, patents granted, patents in force, and patent intensity (filings per gross domestic product, population, or R&D expenditures) can be downloaded in xls or csv formats here.



 September 27-28, 2010 –
September 27-28, 2010 – 













