By Kevin E. Noonan —
 On October 29, the Biotechnology Industry Organization (BIO) and the Association of University Technology Managers (AUTM) filed an amici curiae brief in AMP v. USPTO (the Myriad case). Fittingly (in view of the interest, financial and otherwise, of their members in patented genetic technologies), the brief is focused on the patent-eligibility of isolated DNA (it expressly does not reach the patent-eligibility of the method claim invalidated by the District Court). The brief also makes clear to the Federal Circuit the adverse consequences should it not reverse the District Court's decision striking down Myriad's licensed patents on the BRCA1 and BRCA2 genes.
On October 29, the Biotechnology Industry Organization (BIO) and the Association of University Technology Managers (AUTM) filed an amici curiae brief in AMP v. USPTO (the Myriad case). Fittingly (in view of the interest, financial and otherwise, of their members in patented genetic technologies), the brief is focused on the patent-eligibility of isolated DNA (it expressly does not reach the patent-eligibility of the method claim invalidated by the District Court). The brief also makes clear to the Federal Circuit the adverse consequences should it not reverse the District Court's decision striking down Myriad's licensed patents on the BRCA1 and BRCA2 genes.
 The brief states three grounds for the Court to decide that isolated DNA is patentable subject matter under 35 U.S.C. § 101. First, amici argue that isolated DNA molecules are "man-made compositions of matter" that are not naturally occurring. Second, that isolated DNA molecules are chemical compounds ("A gene is but a chemical compound, albeit a complex one"), which have "new and distinctive properties and uses compared to naturally-occurring DNA." Finally, the brief argues that the District Court erred when it based its decision on the putatively unique properties of DNA (as the "physical embodiment of [genetic] information") that distinguishes it from all other biological molecules, specifically by treating DNA as "mere information" rather than as a chemical compound.
The brief states three grounds for the Court to decide that isolated DNA is patentable subject matter under 35 U.S.C. § 101. First, amici argue that isolated DNA molecules are "man-made compositions of matter" that are not naturally occurring. Second, that isolated DNA molecules are chemical compounds ("A gene is but a chemical compound, albeit a complex one"), which have "new and distinctive properties and uses compared to naturally-occurring DNA." Finally, the brief argues that the District Court erred when it based its decision on the putatively unique properties of DNA (as the "physical embodiment of [genetic] information") that distinguishes it from all other biological molecules, specifically by treating DNA as "mere information" rather than as a chemical compound.
Isolated DNA molecules are "unquestionably" compositions of matter under the statute, the brief argues. The District Court erred in treating these molecules as being "'merely' purified forms of naturally-occurring substances." This is error, the brief argues, because isolated DNA molecules are "new, man-made chemical compositions that do not occur in nature." The District Court also erred because, even if isolated DNA could be considered "purified versions of naturally-occurring DNA, the process of isolation requires such a level of human intervention," i.e., the "hand of man," and "so alters their character and use as to make [isolated DNA molecules] patentable" (or more accurately, patent-eligible), they argue. Finally, the District Court erred in being "led astray [indeed!] by the comparison of DNA to information, 'a common metaphor that is useful in conveying complex science to laypersons [or district court judges], but that does not change the fact that DNA remains a chemical compound, not an alphabet or a language.'" Isolated DNA (again) is a chemical compound, "developed by human ingenuity using complex scientific expertise and equipment" and hence certainly patent-eligible subject matter according to these amici.
Isolated DNA is not merely "isolated" from biological sources, the brief argues, but instead such molecules are "entirely different man-made molecules that do not occur in nature." Moreover, the process of "isolating" DNA transforms it into a "new and different article" having "distinctive form, properties and uses." The brief notes that the District Court "appeared to assume" that isolating DNA was "merely the 'purification' of naturally-occurring substances." This is factually incorrect, in view of the chemical differences between DNA as it occurs in a chromosome and as it exists after its isolation. The brief nicely illustrates the structure of "native DNA" and the structure of genes in the chromosomal context.
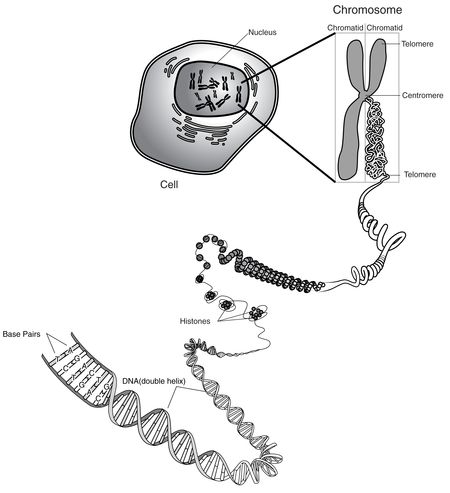
"[C]ritically," according to the brief, "at no point in the process of protein production — or at any other point in an organism's natural life — are genes excised or uncoupled from the rest of the chromosome." That is, "genes do not naturally exist as stand-alone molecules or separate chemical compounds." Thus, DNA as it exists in isolation does not exist, even in "unpurified" form, in nature. The brief illustrates the difference by contrasting the isolated BRCA1 gene, consisting of about 80,000 bp (of which only about 16,000 bp are coding sequence), with chromosome 17 (the chromosomal location of the BRCA1 gene) that comprises about 80 million bp (and about 1200-1500 genes in that chromosome). The process of excising a gene like the BRCA1 gene from a chromosome creates a "new and separate chemical compound that does not exist in nature" according to the brief. These structural differences are even greater when the isolated DNA is cDNA rather than chromosomal DNA, since these molecules are solely the product of human intervention and manipulation; the brief notes and debunks the assertions below that the existence of pseudogenes somehow constitutes a "natural" counterpart of man-made cDNA, based in part on sequence differences between such pseudogenes and cDNA. At least one of the bases for the Court's errors below was it "compared the claimed molecules to naturally-occurring nucleotide sequences, rather than to naturally-occurring DNA molecules" (emphasis in original). Treated as a chemical compound, isolated human chromosomal DNA and cDNA are non-naturally occurring chemical compounds, the brief argues, and this was the proper basis for the Patent Office to determine that isolated human DNA was patent-eligible subject matter.
The brief distinguishes two 19th Century Supreme Court cases used by the District Court to support its decision, by arguing that the District Court misconstrued their holdings. In both American Wood-Paper Co. v. Fiber Disintegrating Co., 90 U.S. (23 Wall.) 566, 595 (1874), and Cochrane v. Badische Anilin & Soda Fabrik, 111 U.S. 293, 311 (1884), the brief argues that the Court rejected patentability of the claimed products because they were not novel, not (as the District Court held) that "products of nature" are not patent-eligible: "[n]either opinion suggested . . . that a man-made molecule that did not previously exist was unpatentable subject matter," they argue. Indeed, this point could have been made even more forcefully, since the Court in the Wood Paper Pulp cases expressly stated that it was not deciding this question:
It is insisted, however, that the paper pulp which had been produced before the invention of Watt & Burgess was not pure cellulose, that it was only approximately pure, and from this it is argued that the pure article obtained from wood by their process is a different and new product, or manufacture. Whether a slight difference in the degree of purity of an article produced by several processes justifies denominating the products different manufactures, so that different patents may be obtained for each, may well be doubted, and it is not necessary to decide.
Id. at 594 (emphasis added).
The brief is carefully to point out that the issue here is patent-eligibility, and that patentability is dependent on satisfaction of the other requirements of patentability, citing cases where the Federal Circuit has held isolated DNA to be unpatentable for want of novelty (In re Gleave, 560 F.3d 1331 (Fed. Cir. 2009)), obviousness (In re Kubin, 561 F.3d 1351 (Fed. Cir. 2009)), lack of utility (In re Fisher, 421 F.3d 1365 (Fed. Cir. 2005)), or failure to satisfy the written description requirement (Regents of Univ. of Cal. v. Eli Lilly & Co., 119 F.3d 1559 (Fed. Cir. 1997)). In the process, the brief sub silentio reinforces the point that in all these cases the Court never questioned that isolated DNA was patent-eligible under 35 U.S.C. § 101.
The brief also sets forth a history of patent-eligibility of "products of nature," including Louis Pasteur's yeast (U.S. Patent No. 141,072), aspirin (obtained from coal tar), adrenaline (Parke-Davis & Co. v. H.K. Mulford Co., 189 F. 95, 103 (C.C.S.D.N.Y. 1911)), vitamin B12 (Merck & Co. v. Olin Mathieson Chem. Corp., 253 F.2d 156, 164-165 (4th Cir. 1958), prostaglandins (In re Merz, 97 F.2d 599, 601 (C.C.P.A. 1938)), and human blood clotting Factor VIII (Scripps Clinic & Research Found. v. Genentech, Inc., 666 F. Supp. 1379, 1389 n.6 (N.D. Cal. 1987)), and distinguishes American Fruit Growers, Inc. v. Brogdex Co., 283 U.S. 1, 12-13 (1931), as being related to the definition of a "manufacture" under the statute rather than defining patent-eligibility for "products of nature." "[I]n addition to creating a whole new chemical composition that does not exist in nature . . . [isolating DNA] imparts new utilities and functions unavailable from native DNA," the brief argues, illustrating these "new utilities" by gene therapy and recombinant production of BRCA1 and BRCA2 proteins. None of these utilities are available for native DNA, and thus "this is not a situation in which natural substances 'serve the ends nature originally provided and act quite independently of any effort of the patentee'" amici argue, citing (and distinguishing) Funk Bros. Seed Co. v. Kalo Inoculant Co., 333 U.S. 127, 131 (1948).
The brief criticizes the basis of the District Court's decision that the utility of isolated DNA molecules is ""primarily a function of the nucleotide sequence identity between native and isolated BRCA1/2 DNA," stating that this similarity "is no more attributable to its similarity to native DNA than the utilities insulin, penicillin, vitamin B12, adrenaline, or other drug preparations are due to their similarity to their naturally-occurring counterparts." The brief gets to the gist of the District Court's error: this similarity between native and isolated DNA "could be made about essentially any biotechnology product." The salient fact is that isolated DNA is useful because it is isolated: the brief argues not that isolated DNA has the same utilities as native DNA but just to a greater degree, but that "native DNA cannot be used for such purposes at all" (emphasis in original). The brief argues that this is the "'character' and 'use" that distinguishes isolated DNA molecules from native DNA for purposes of patentability."
Finally in this portion of the brief, amici argue again that the District Court erred by ignoring the "chemical nature of DNA" and focusing on DNA as a "physical embodiment of information." The District Court misapplied this metaphor for fact, by "likening DNA's nucleotide sequence as 'information,'" citing numerous parts of the District Court's opinion. "DNA as information" is an analogy, not an identity the brief argues, saying (again) that "DNA is a chemical compound." "The description of [the biological functions of DNA, such as transcription and translation] as a relaying of 'information' may be useful as a pedagogical tool," amici argue, "but it does not differentiate DNA from any other chemical compound."
The brief's arguments about the adverse consequences of affirming the District Court's decision to ban isolated DNA from patent eligibility are contrasted with the lack of any evidence that permitting isolated DNA to be patented has had any negative effects on the "progress" of science. The brief forcefully argues that patents on isolated DNA in fact promote innovation: the U.S. biotechnology industry "consists largely of small firms that are engaged in foundational research and dependent on private investment, not product revenues, to fund their work." For such companies, patent protection is "essential" in order for them to obtain the private investment they need. The brief recites a number of "biotechnology success stories," including Amgen's successful production of recombinant erythropoietin, as evidence of such public benefit, as well as advances in the production of EPO and other therapeutic proteins, gene therapy, vaccination, genetic testing, agriculture, food safety, industrial and environmental biotechnology, and future "unforeseeable discoveries" as reasons to uphold patent-eligibility of isolated DNA. To negate patent-eligibility, on the other hand, would have "far-reaching negative consequences," and failure to reverse the District Court would "cast a cloud of uncertainty over thousands of similar patents and compromise the ability of biotechnology firms to pursue groundbreaking discoveries in human healthcare, renewable energy, and sustainable agriculture" and would also harm university technology transfer efforts. The brief cites numerous empirical studies, from industry (Ernst & Young, Beyond Borders: Global Biotechnology Report 34 (2009)) to academe (Grabowski, Follow-On Biologics, 7 Nature Reviews Drug Discovery 479, 482 (2008)) for the importance of patent protection in bringing new biotechnology inventions to market. Patents are particularly important for getting early-stage companies through the "valley of death" — the time after proof of concept but before regulatory approval or commercialization — because patents "are typically the only assets those firms possess that are sufficiently stable and valuable to attract the large amounts of capital they need to exploit promising research toward new drugs and diagnostics," citing Barfield & Calfee, Biotechnology and the Patent System 27 (2007), and Grabowski et al., The Market for Follow-On Biologics, 25 Health Affairs 1291, 1299 (2006). As in other briefs, amici here exemplify the potential for negative consequences of a DNA patenting ban using President Clinton's and Prime Minister Blair's gaffe at suggesting patenting DNA might be curtailed, with a resulting $50 billion loss in value for genomics and other DNA patent-reliant companies. Importantly, the brief also argues that this shake in investor confidence would not be limited to DNA-dependent companies, since the District Court's misguided "product of nature" patent ban extends to "a wide variety of natural substances that have been isolated and purified," which would be "to the detriment of the biotechnology industry and the public.
Patents on isolated DNA in fact benefit the public, amici argue, by promoting commercialization of basic scientific discovery into useful (indeed, in some instances lifesaving) diagnostic and therapeutic advances. Permitting patents on isolated DNA, as the U.S. Patent and Trademark Office and the Federal Circuit have done over the past thirty years, has had no negative effects on innovation or progress in the medical or biological sciences, the brief argues. Indeed, the brief notes that every attempt to detect any such negative effects on innovation (the "tragedy" of the anticommons) has met with failure, as in each and every case there is no evidence that there has been any tragedy or indeed any appreciable negative effects. These efforts to find a "tragedy" resulting from patenting isolated DNA have extended from academia (Walsh et al., Patents, Material Transfers and Access to Research Inputs in Biomedical Research 3 (Sept. 20, 2005) ("patenting does not seem to limit research activity significantly, particularly among those doing basic research"), through the Federal Trade Commission (citing Emerging Health Care Issues 32 (2009)), National Research Council, Reaping the Benefits of Genomic and Proteomic Research 134 (2006), and (unmentioned) to the SAGHHS report. And anecdotal reports of specific instances when companies like Myriad enforced or threatened to enforce its patent rights must not confuse "the ordinary assertion of patent rights against commercial competitors with limits on basic research"; specifically, "Myriad appears to have never asserted its patents based on genetic testing research, but only against substantial direct commercial competitors," citing Holman, The Impact of Human Gene Patents on Innovation and Access, 76 UMKC L. Rev. 295, 347 (2007).
The brief takes on the principle confounding issue in this debate, the conflating of failure in the U.S. healthcare system and inequities in its distribution, with the limited exclusive rights granted by patents:
The stories of the individual Plaintiffs in this case help explain why members have pushed so hard to encourage insurance companies with restrictive reimbursement policies to pay for important therapeutics and diagnostics and to establish company-sponsored plans that provide products and services to patients who cannot afford them. A106-107, A149-150 (discussing patients whose insurance companies would not pay for genetic testing). Plaintiffs' effort to abolish patents on isolated DNA molecules, however, is misdirected and shortsighted. It mistakes problems in the insurance system for problems in the patent system and will harm rather than help patients in the long run.
It is easy to argue after an invention has already been discovered and disclosed that the public would be better off if it were not patented. It is just as easy to single out a particular invention and argue with the benefit of hindsight that patent protection was not necessary for its discovery and development. Such facile arguments ignore the long-term benefits that the public derives from providing patent protection in exchange for the disclosure of new and useful discoveries.
The advances made by the U.S. biotechnology industry under current law were not inevitable, and the industry's future success depends on the ability continue attracting private investors willing to shoulder the substantial risk financing research and development. In the life sciences, early-stage hold roughly two-thirds of the future clinical pipeline. Boston Consulting Group, Rising to the Productivity Challenge 6 & Ex. 4 (2004). Without patent protection for isolated DNA molecules, many companies would be unable to see those projects through to completion. The list of potentially life-enhancing therapeutics and diagnostics that die in the pipeline as a result might never be known. But their absence would be acutely felt by patients.
If there is a need to strike a different balance between the public interest in strong patent rights that promote innovation and making diagnostic tests accessible to patients that cannot afford them, that is a balance that Congress, not the courts, should make "based on sound evidence and with due regard to the reliance interest of existing patentholders, rather than by the courts based an anecdotal evidence about one particular patent-holder."
In its Statement of Interest, BIO asserts that represents "over 1100 companies, academic and biotechnology centers" that "undertake research and development of biotechnological healthcare, agricultural, environmental and industrial products." The "vast majority" of BIO members are "small companies that have yet to bring a product to market or attain profitability, and thus rely heavily on venture capital and other private investment." This has made "critical" patenting on isolated DNA molecules to attract such investment "necessary for development of innovative diagnostic, environmental, renewable energy, and agricultural products." AUTM, according to the Statement, is "the largest association of university technology transfer professionals, with members from over 350 universities, research institutions, teaching hospitals, and government agencies worldwide, as well as hundreds of companies involved with managing and licensing innovations derived from academic and nonprofit research." Its interests in the outcome of this case are self-evident: universities own overwhelmingly numbers of patents on isolated DNA.
Image from National Human Genome Research Institute, Chromosome, <http://www.accessexcellence.org/RC/VL/GG/nhgri_PDFs/chromosome.pdf>
For additional information regarding this and other related topics, please see:
• "AIPLA Submits Amicus Brief in AMP v. USPTO," October 3, 2010
• "IPO Files Amicus Brief in AMP v. USPTO," November 2, 2010
• "AMP v. USPTO — Briefing Update," November 1, 2010
• "DOJ Tries to Be All Things to All Constituencies in Myriad Amicus Brief," October 31, 2010
• "Myriad Files Appeal Brief in AMP v. USPTO," October 28, 2010
 Hoffman-La Roche successfully secured a preliminary injunction against Cobalt Pharmaceuticals in the patent suit over the bone-loss drug Boniva, thus preventing Cobalt from launching a generic version of the drug.
Hoffman-La Roche successfully secured a preliminary injunction against Cobalt Pharmaceuticals in the patent suit over the bone-loss drug Boniva, thus preventing Cobalt from launching a generic version of the drug. Apotex and Cobalt filed Abbreviated New Drug Applications (ANDAs) for generic ibandronate, the active ingredient in Boniva. In September 2007, Roche sued Apotex and Cobalt in the U.S. District Court for the District of New Jersey for infringement of U.S. Patent Nos. 6,143,326, 6,294,196, 7,192,938, 4,927,814, and 7,410,957 (see "Court Report," September 30, 2007). Eventually, a total of six lawsuits were consolidated into two pretrial proceedings, and the claims relating to the '326, '196, and '938 patents were settled. In August, the District Court granted summary judgment to Roche, finding that Apotex's ANDA literally infringes the '814 patent. The defendants maintain that the '814 patent is unenforceable due to inequitable conduct.
Apotex and Cobalt filed Abbreviated New Drug Applications (ANDAs) for generic ibandronate, the active ingredient in Boniva. In September 2007, Roche sued Apotex and Cobalt in the U.S. District Court for the District of New Jersey for infringement of U.S. Patent Nos. 6,143,326, 6,294,196, 7,192,938, 4,927,814, and 7,410,957 (see "Court Report," September 30, 2007). Eventually, a total of six lawsuits were consolidated into two pretrial proceedings, and the claims relating to the '326, '196, and '938 patents were settled. In August, the District Court granted summary judgment to Roche, finding that Apotex's ANDA literally infringes the '814 patent. The defendants maintain that the '814 patent is unenforceable due to inequitable conduct. After a bench trial earlier this year resulted in Allergan's patent covering the pinkeye drop Zymar being found obvious, Allergan moved for a new trial. On November 3, Allergan's motion was denied.
After a bench trial earlier this year resulted in Allergan's patent covering the pinkeye drop Zymar being found obvious, Allergan moved for a new trial. On November 3, Allergan's motion was denied. In 2007, Allergan, the co-owner of U.S. Patent No. 6,333,045, filed suit in the U.S. District Court for the District of Delaware against Apotex after Apotex filed an ANDA covering a generic version of Allergan's 0.3% gatifloxacin opthalmic solution (see "Court Report," December 9, 2007). A bench trial was held in January 2010, and the District Court found that Apotex had shown claim 7 to be obvious in view of a 1989 reference and two patents. Allergan subsequently moved for a new trial.
In 2007, Allergan, the co-owner of U.S. Patent No. 6,333,045, filed suit in the U.S. District Court for the District of Delaware against Apotex after Apotex filed an ANDA covering a generic version of Allergan's 0.3% gatifloxacin opthalmic solution (see "Court Report," December 9, 2007). A bench trial was held in January 2010, and the District Court found that Apotex had shown claim 7 to be obvious in view of a 1989 reference and two patents. Allergan subsequently moved for a new trial. Novo Nordisk and Actavis have moved to dismiss a patent infringement suit over Actavis' plan to bring a generic version of the diabetes drug PrandiMet to market.
Novo Nordisk and Actavis have moved to dismiss a patent infringement suit over Actavis' plan to bring a generic version of the diabetes drug PrandiMet to market. In October 2009, Novo Nordisk brought suit in the U.S. District Court for the Southern District of New York against Actavis, alleging infringement of U.S. Patent No. 6,677,358 following Actavis' ANDA filing (see "Court Report," November 8, 2009). After Novo Nordisk brought the lawsuit, Actavis amended the ANDA and withdrew its Paragraph IV certification.
In October 2009, Novo Nordisk brought suit in the U.S. District Court for the Southern District of New York against Actavis, alleging infringement of U.S. Patent No. 6,677,358 following Actavis' ANDA filing (see "Court Report," November 8, 2009). After Novo Nordisk brought the lawsuit, Actavis amended the ANDA and withdrew its Paragraph IV certification.

















