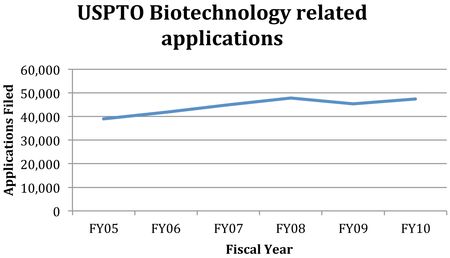
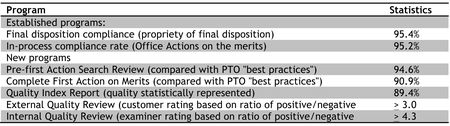
By Donald Zuhn —
 In an appeal decided last month, the Federal Circuit reversed a decision by the District Court for the Southern District of New York awarding prejudgment interest to Plaintiffs-Appellees Sanofi-Aventis, Sanofi-Synthelabo, Inc., and Bristol-Myers Squibb Sanofi Pharmaceuticals Holding Partnership ("Sanofi"); affirmed the District Court's denial of a motion for leave to file a supplemental answer, affirmative defenses, and counterclaims by Defendants-Appellants Apotex Inc. and Apotex Corp. ("Apotex"); and affirmed the District Court's holding that Apotex Inc. is jointly and severally liable for all damages. The appeal was the third heard by the Court regarding clopidogrel bisulfate tablets, which are sold by Sanofi under the brand name Plavix®. In the first appeal, the Federal Circuit affirmed the District Court's grant of a preliminary injunction (see "Sanofi-Synthelabo v. Apotex, Inc. (Fed. Cir. 2006)"). In the second appeal, the Federal Circuit affirmed the District Court's judgment that Sanofi's U.S. Patent No. 4,847,265 was not invalid, infringed, and not unenforceable (see "Sanofi-Synthelabo v. Apotex, Inc. (Fed. Cir. 2008)").
In an appeal decided last month, the Federal Circuit reversed a decision by the District Court for the Southern District of New York awarding prejudgment interest to Plaintiffs-Appellees Sanofi-Aventis, Sanofi-Synthelabo, Inc., and Bristol-Myers Squibb Sanofi Pharmaceuticals Holding Partnership ("Sanofi"); affirmed the District Court's denial of a motion for leave to file a supplemental answer, affirmative defenses, and counterclaims by Defendants-Appellants Apotex Inc. and Apotex Corp. ("Apotex"); and affirmed the District Court's holding that Apotex Inc. is jointly and severally liable for all damages. The appeal was the third heard by the Court regarding clopidogrel bisulfate tablets, which are sold by Sanofi under the brand name Plavix®. In the first appeal, the Federal Circuit affirmed the District Court's grant of a preliminary injunction (see "Sanofi-Synthelabo v. Apotex, Inc. (Fed. Cir. 2006)"). In the second appeal, the Federal Circuit affirmed the District Court's judgment that Sanofi's U.S. Patent No. 4,847,265 was not invalid, infringed, and not unenforceable (see "Sanofi-Synthelabo v. Apotex, Inc. (Fed. Cir. 2008)").
 Seeking approval to market a generic clopidogrel bisulfate tablets, Apotex filed an Abbreviated New Drug Application (ANDA) with the FDA. In response to Apotex's ANDA filing, Sanofi brought suit against Apotex for infringement of the '265 patent, which triggered a thirty-month stay of FDA approval for Apotex's ANDA, pursuant to 21 U.S.C. § 355(j)(5)(B)(iii). Eight months after the stay expired, the FDA approved Apotex's ANDA.
Seeking approval to market a generic clopidogrel bisulfate tablets, Apotex filed an Abbreviated New Drug Application (ANDA) with the FDA. In response to Apotex's ANDA filing, Sanofi brought suit against Apotex for infringement of the '265 patent, which triggered a thirty-month stay of FDA approval for Apotex's ANDA, pursuant to 21 U.S.C. § 355(j)(5)(B)(iii). Eight months after the stay expired, the FDA approved Apotex's ANDA.
Prior to FDA approval, the parties began settlement negotiations that culminated in a tentative agreement. Under that agreement, Sanofi granted a license to Apotex, allowing it to begin selling generic clopidogrel bisulfate tablets before the '265 patent expired, and agreed not to launch an authorized generic during the pendency of the license. The Federal Trade Commission objected to the agreement, in part because of the provision not to launch an authorized generic (FTC approval was required as a result of a previous litigation involving Bristol-Myers Squibb Company, which is an owner of Bristol-Myers Squibb Sanofi Pharmaceuticals Holding Partnership). As a result of the FTC's objections, Sanofi withdrew the agreement and the parties reinstituted settlement negotiations.
 Two months later, the parties entered into a second settlement agreement that did not include a provision prohibiting Sanofi from launching an authorized generic. Instead, the BMS executive negotiating the agreement orally promised Apotex that Sanofi would not launch an authorized generic during the pendency of the license. However, when BMS presented the second settlement agreement to the FTC for approval, it did not disclose the oral agreement. After Apotex subsequently disclosed the oral agreement to the FTC, the FTC requested written certification from BMS that there was no oral promise not to launch an authorized generic. Although BMS provided such certification, the second settlement agreement was not approved pending an investigation into the existence of the oral agreement.
Two months later, the parties entered into a second settlement agreement that did not include a provision prohibiting Sanofi from launching an authorized generic. Instead, the BMS executive negotiating the agreement orally promised Apotex that Sanofi would not launch an authorized generic during the pendency of the license. However, when BMS presented the second settlement agreement to the FTC for approval, it did not disclose the oral agreement. After Apotex subsequently disclosed the oral agreement to the FTC, the FTC requested written certification from BMS that there was no oral promise not to launch an authorized generic. Although BMS provided such certification, the second settlement agreement was not approved pending an investigation into the existence of the oral agreement.
Subject to a regulatory denial provision in the second agreement, Apotex launched its clopidogrel bisulfate product. In response, Sanofi moved for a preliminary injunction, which the District Court granted. The District Court then held a bench trial regarding liability, finding the '265 patent to be valid and enforceable, and bifurcating on the issue of damages.
During the damages stage, Apotex sought leave to file a supplemental answer, in which Apotex alleged that the '265 patent was unenforceable for patent misuse due to BMS's failure to disclose the oral agreement to the FTC and its subsequent false certification to the FTC. The District Court denied Apotex's motion, determining in part that BMS's actions likely did not constitute patent misuse. Sanofi, meanwhile, moved for summary judgment on the issue of damages, noting that the second settlement agreement set Sanofi's "actual damages" as 50% of Apotex's net sales. The District Court granted Sanofi's motion, and ordered Apotex to pay $442,209,362 in damages (50% of Apotex's net sales) and an additional $107,930,857 in prejudgment interest, rejecting Apotex's argument that the second settlement agreement precluded an award or prejudgment interest. The District Court also held that Apotex Inc. and Apotex Corp. were jointly and severally liable for the damages.
On appeal, Apotex argued, in part, that the District Court erred by awarding Sanofi prejudgment interest, holding Apotex Inc. and Apotex Corp. jointly and severally liable, and denying Apotex's motion for leave to file a supplemental answer. With respect to the award of prejudgment interest, Apotex contended that as a result of paragraph 14(ii) of the second settlement agreement, Sanofi contractually limited its full recovery to 50% of Apotex's net sales. Paragraph 14(ii) states:
If the litigation results in a judgment that the '265 patent is not invalid or unenforceable, Sanofi agrees that its actual damages for any past infringement by Apotex, up to the date on which Apotex is enjoined, will be 50% of Apotex's net sales of clopidogrel products . . . . Sanofi further agrees that it will not seek increased damages under 35 U.S.C. § 284.
Apotex also noted that paragraph 10 of the agreement, which defined the amount Sanofi would reimburse Apotex for Apotex's investment in inventory, expressly provided for a separate interest payment, in contrast with paragraph 14(ii). The District Court, however, determined that because the second settlement agreement only explicitly limited damages, the agreement did not preclude an additional award of interest on those damages pursuant to 35 U.S.C. § 284.
 In reversing the District Court's award of prejudgment interest, the Federal Circuit concluded that the parties intended that the phrase "actual damages" in the second settlement agreement include all damages necessary to compensate Sanofi for Apotex's infringement, and because prejudgment interest is a form of compensatory damages, the majority concluded that the District Court had erred in awarding additional prejudgment interest pursuant to 35 U.S.C. § 284. The Federal Circuit explained that:
In reversing the District Court's award of prejudgment interest, the Federal Circuit concluded that the parties intended that the phrase "actual damages" in the second settlement agreement include all damages necessary to compensate Sanofi for Apotex's infringement, and because prejudgment interest is a form of compensatory damages, the majority concluded that the District Court had erred in awarding additional prejudgment interest pursuant to 35 U.S.C. § 284. The Federal Circuit explained that:
By agreeing to a simple formula to quantify Sanofi's "actual damages" in paragraph 14(ii), the parties avoided litigating a potentially complex issue. Such certainty is beneficial to both parties. Apotex benefited by knowing the exact amount of its potential liability. Sanofi benefited because it could fix damages without having to resort to further litigation, including complex and potentially lengthy discovery. These benefits disappear, however, if prejudgment interest is not included in "actual damages" since — as demonstrated here — the parties must engage in further litigation over an additional large liability.
On the issue of liability, Apotex argued that Apotex Inc. was not liable for damages because Apotex Inc. never engaged in the "commercial manufacture, use, offer to sell, or sale within the United States or importation into the United States of an approved drug," as required by 35 U.S.C. § 271(e)(4)(C). Apotex noted instead that while Apotex Inc. manufactured the drug in Canada, Apotex Corp. imported the drug and made all commercial sales in the United States. Apotex contended that Apotex Inc. could not be held liable for infringement under § 271(e)(4)(C) because Sanofi failed to present evidence that Apotex Inc.'s actions took place in the United States. In affirming the District Court's determination that Apotex Inc. was jointly and severally liable for Sanofi's damages, the Federal Circuit pointed to the second settlement agreement, which defined Sanofi's compensatory damages from Apotex's infringement, and broadly defined "Apotex" to include "Apotex Inc. and Apotex Corp., collectively and individually, and including any entity now or hereafter owned or controlled by any of them."
Finally, with respect to Apotex's supplemental answer, the Federal Circuit determined that "BMS's failure to disclose the oral side deal with Apotex, and its false certification to the FTC regarding the same, in no way broadened the scope of the ’265 patent grant." As a result, the Federal Circuit determined that "[t]he district court properly rejected Apotex's patent misuse defense as futile," citing Princo Corp. v. International Trade Commission, 616 F.3d 1318 (Fed. Cir. 2010) (en banc), for the proposition that "the key inquiry under the patent misuse doctrine is whether . . . the patentee has impermissibly broadened the physical or temporal scope of the patent grant and has done so in a manner that has anticompetitive effects," and adding that "[p]aragraph 14 of the Settlement Agreement states the way damages shall be measured, and does not mention that such damages shall be free of the routine award of interest."
Judge Newman, dissenting with respect to the reversal of the District Court's award of prejudgment interest, argued that the second settlement agreement "did not alter the general rule that prejudgment interest is awarded on damages for patent infringement." As to the interest provision in paragraph 10 of the second settlement agreement, she stated:
That the parties, in a different section of the Agreement dealing with purchase of inventory, expressly provided for payment of interest on those purchases, does not support the panel majority's theory, for prejudgment interest on sale of inventory is not the established rule, as is prejudgment interest on infringement damages.
Sanofi-Aventis v. Apotex Inc. (Fed. Cir. 2011)
Panel: Circuit Judges Newman, Schall, and Moore
Opinion by Circuit Judge Moore; opinion dissenting-in-part by Circuit Judge Newman
 The Intellectual Property Owners Association (IPO) and U.S. Patent and Trademark Office will be co-sponsoring the 22nd Annual Conference on USPTO Law and Practice (PTO Day) on December 5, 2011 in Washington, D.C. The PTO Day program includes patent general sessions on the following topics:
The Intellectual Property Owners Association (IPO) and U.S. Patent and Trademark Office will be co-sponsoring the 22nd Annual Conference on USPTO Law and Practice (PTO Day) on December 5, 2011 in Washington, D.C. The PTO Day program includes patent general sessions on the following topics: In addition, USPTO Director David Kappos will provide a luncheon address. A copy of the program, including a list of speakers, can be found here.
In addition, USPTO Director David Kappos will provide a luncheon address. A copy of the program, including a list of speakers, can be found here.