By Kevin E. Noonan —
The Federal Circuit reversed a finding of obviousness in Eurand Inc. v. Mylan Pharmaceuticals Inc. (under the caption In re Cyclobenzaprine Hydrochloride Extended-Release Capsule Patent Litigation), taking the occasion to expound on both the proper consideration of the evidence proffered to support an obviousness determination as well as the evidentiary burdens and their proper application.
 The case involved ANDA litigation under 35 U.S.C. § 271(e)(2) relating to generic versions of "modified-release dosage forms" of cyclobenzaprine hydrochloride, a muscle relaxing drug marketed as Amrix by Cephalon (owner of exclusive patent licensee Anesta AG). The dosage form released API cyclobenzaprine hydrochloride over a 24-hour period, in contrast to immediate-release forms, which require multiple doses over the same time period. The Orange Book listed U.S. Patent Nos. 7,387,793 and 7,544,372; the following are representative claims for these patents (which share an identical specification):
The case involved ANDA litigation under 35 U.S.C. § 271(e)(2) relating to generic versions of "modified-release dosage forms" of cyclobenzaprine hydrochloride, a muscle relaxing drug marketed as Amrix by Cephalon (owner of exclusive patent licensee Anesta AG). The dosage form released API cyclobenzaprine hydrochloride over a 24-hour period, in contrast to immediate-release forms, which require multiple doses over the same time period. The Orange Book listed U.S. Patent Nos. 7,387,793 and 7,544,372; the following are representative claims for these patents (which share an identical specification):
U.S. Patent No. 7,387,793:
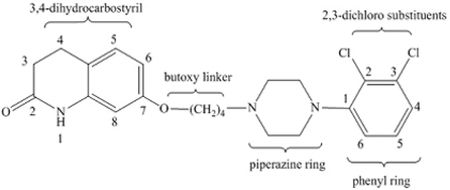
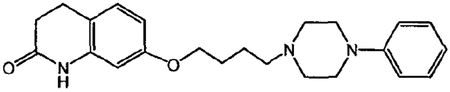
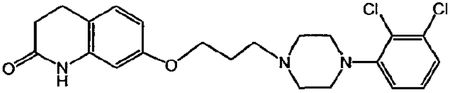
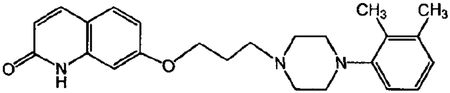
1. A multi-particulate pharmaceutical dosage form of a skeletal muscle relaxant providing a modified release profile comprising a population of extended release beads, wherein said extended release beads comprise an active-containing core particle comprising a skeletal muscle relaxant selected from the group consisting of cyclobenzaprine, pharmaceutically acceptable salts or derivatives thereof and mixtures thereof; and an extended release coating comprising a water insoluble polymer membrane surrounding said core, wherein said dosage form when dissolution tested using United States Pharmacopoeia Apparatus 2 (paddles @ 50 rpm) in 900 mL of 0.1N HCl at 37.degree. C. exhibits a drug release profile substantially corresponding to the following pattern: after 2 hours, no more than about 40% of the total active is released; after 4 hours, from about 40-65% of the total active is released after 8 hours, from about 60-85% of the total active is released; wherein said dosage form provides therapeutically effective plasma concentration over a period of 24 hours to treat muscle spasm associated with painful musculoskeletal conditions when administered to a patient in need thereof; and wherein said water insoluble polymer membrane comprises a water insoluble polymer selected from the group consisting of ethers of cellulose, esters of cellulose, cellulose acetate, ethyl cellulose, polyvinyl acetate, neutral copolymers based on ethylacrylate and methylmethacrylate, copolymers of acrylic and methacrylic acid esters with quaternary ammonium groups, pH-insensitive ammonio methacrylic acid copolymers, and mixtures thereof; and a plasticizer selected from the group consisting of triacetin, tributyl citrate, tri-ethyl citrate, acetyl tri-n-butyl citrate, diethyl phthalate, dibutyl sebacate, polyethylene glycol, polypropylene glycol, castor oil, acetylated mono- and di-glycerides and mixtures thereof.
2. The pharmaceutical dosage form of claim 1, wherein said skeletal muscle relaxant comprises cyclobenzaprine hydrochloride.
3. The pharmaceutical dosage form of claim 2 wherein said pharmaceutical dosage form provides a maximum blood plasma concentration (Cmax) within the range of about 80% to 125% of about 20 ng/mL of cyclobenzaprine HCl and an AUC0-168 within the range of about 80% to 125% of about 740 ng hr/mL and a Tmax within the range of 80% to 125% of about 7 hours following oral administration of a single 30 mg cyclobenzapnine HCl MR Capsule.
U.S. Patent No. 7,544,372:
1. A method of relieving muscle spasms in a patient in need thereof comprising administering a pharmaceutical dosage form of a skeletal muscle relaxant comprising a population of extended release beads, wherein said extended release beads comprise: an active-containing core particle comprising a skeletal muscle relaxant selected from the group consisting of cyclobenzaprine, pharmaceutically acceptable salts or derivatives thereof and mixtures thereof; and an extended release coating comprising a water insoluble polymer membrane surrounding said core, wherein said dosage form when dissolution tested using United States Pharmacopoeia Apparatus 2 (paddles @ 50 rpm) in 900 mL of 0.1N HCl at 37.degree. C. exhibits a drug release profile substantially corresponding to the following pattern: after 2 hours, no more than about 40% of the total active is released; after 4 hours, from about 40-65% of the total active is released; after 8 hours, from about 60-85% of the total active is released; wherein said dosage form provides a therapeutically effective plasma concentration over a period of 24 hours to treat muscle spasm associated with painful musculoskeletal conditions; and wherein said water insoluble polymer membrane comprises a water insoluble polymer selected from the group consisting of ethers of cellulose, esters of cellulose, cellulose acetate, ethyl cellulose, polyvinyl acetate, neutral copolymers based on ethylacrylate and methylmethacrylate, copolymers of acrylic and methacrylic acid esters with quaternary ammonium groups, pH-insensitive ammonio methacrylic acid copolymers, and mixtures thereof; and a plasticizer selected from the group consisting of triacetin, tributyl citrate, tri-ethyl citrate, acetyl tri-n-butyl citrate, diethyl phthalate, dibutyl sebacate, polyethylene glycol, polypropylene glycol, castor oil, acetylated mono- and di-glycerides and mixtures thereof.
2. The method of claim 1, wherein said skeletal muscle relaxant comprises cyclobenzaprine hydrochloride.
3. The method of claim 2 wherein said pharmaceutical dosage form provides a maximum blood plasma concentration (Cmax) within the range of about 80% to 125% of about 20 ng/mL of cyclobenzaprine HCl and an AUC 0-168 within the range of about 80% to 125% of about 740 ng hr/mL and a Tmax within the range of 80% to 125% of about 7 hours following a single oral administration a pharmaceutical dosage form comprising 30 mg of cyclobenzaprine HCl.
In a bench trial, the District Court found the claims infringed but invalid for obviousness (the Court rejected the defendants best mode defense). In addition, the Court imposed an injunction on defendants after the expiration of the 30-month stay and FDA approval of generic Amrix (despite finding the patent claims invalid).
The Federal Circuit reversed the obviousness determination, in an opinion by Judge O'Malley, joined by Judges Newman and Reyna. A succinct statement for the Federal Circuit's decision is that the District Court did not consider that the prior art did not know "the pharmacokinetic/ pharmacodynamics relationship for the claimed drug formulation," and consequently did not properly assess the prior art in performing its obviousness analysis. In making this assessment, the Federal Circuit considered the highlighted (in bold type above) limitations in the claims to be relevant to its decision; the opinion characterized the limitations of claim 3 in each patent as being related to "pharmacokinetic values."
 The opinion defines "pharmacokinetic" as "the study of what a person's body does to a drug after administration, and consider this limitation to limit "various characteristics about the drug's behavior in the patient's blood plasma." The "pharmacokinetic/pharmacodynamic ("PK/PD") profile, according to the Court's opinion, is the relationship between "what a person's body does to the drug" and "the effect that the drug renders on a person's body" ("pharmacodynamics"), here, "the relief of muscle spasms." These considerations are further complicated because while the determination of a drug formulation's pharmacokinetics is a "quantitative exercise," according to the Court, pharmacodynamics is "a qualitative exercise." And the "therapeutically effective plasma concentration" is, according to the Court and Cephalon's expert, "a concentration that the formulation provides when the formulation works." According to expert testimony, these relationships were worked out by the inventors using a combination of computer modeling and in vitro and in vivo assays, verified by clinical testing to show that the claimed formulation was clinically effective.
The opinion defines "pharmacokinetic" as "the study of what a person's body does to a drug after administration, and consider this limitation to limit "various characteristics about the drug's behavior in the patient's blood plasma." The "pharmacokinetic/pharmacodynamic ("PK/PD") profile, according to the Court's opinion, is the relationship between "what a person's body does to the drug" and "the effect that the drug renders on a person's body" ("pharmacodynamics"), here, "the relief of muscle spasms." These considerations are further complicated because while the determination of a drug formulation's pharmacokinetics is a "quantitative exercise," according to the Court, pharmacodynamics is "a qualitative exercise." And the "therapeutically effective plasma concentration" is, according to the Court and Cephalon's expert, "a concentration that the formulation provides when the formulation works." According to expert testimony, these relationships were worked out by the inventors using a combination of computer modeling and in vitro and in vivo assays, verified by clinical testing to show that the claimed formulation was clinically effective.
The Federal Circuit held that the District Court erred by concluding that "bioequivalence" alone was enough to support its obviousness determination. "Bioequivalence" in this case means that "the body is exposed to the same amount of active pharmaceutical ingredient at the same rate after administration of either an immediate-release or extended-release formulation." This is the standard imposed by the FDA to grant approval of an ANDA (as here). This result depended on a showing that the pharmacokinetic profile (including the AUC and cmax) were the same. However, the Federal Circuit believed that this is not enough because the claim requires the same "pharmacokinetic/pharmcodynamic" ratio, something not established by the evidence adduced before the District Court. The opinion states that the resulting error is that the District Court was also "required to consider the asserted claims' limitation requiring therapeutic effectiveness, and whether it would have been obvious to one of ordinary skill in the art at the time of the invention that a bioequivalent PK value would satisfy that limitation." Key to this failure was that cyclobenzaprine "lacked a known PK/PD relationship at the time of invention," which the Federal Circuit believed was critically important because "[w]ithout such a known relationship [], skilled artisans could not predict whether any particular PK profile, including a bioequivalent one, would produce a therapeutically effective formulation." (In addition, there was testimony that the "mode of action" of the drug was "not really well known," something conceded by defendants' expert).
The Federal Circuit criticized what it characterized as the District Court affirmatively disregarding this deficiency in the evidence. Citing the District Court's opinion that the skilled artisan would have "expect[ed] the extended release formulation to have the same PD effect on the body if it has the immediate release formulation's PK profile," the Federal Circuit found a contradiction to the defendants' acknowledgement that the PK/PD relationship was not known and thus could not support the expectation attributed to the skilled artisan by the District Court. Because of the absence of the PK/PD relationship in the prior art, "there was no way to match the dosage for the extended-release formulation to achieve a known therapeutic effect" according to the Federal Circuit opinion.
In view of this lack of knowledge, the Federal Circuit distinguished KSR Int'l Co. v. Teleflex Inc., quoting that opinion for the proposition that an invention is obvious when the skilled worker pursues "known options" from "a finite number of identified, predictable solutions." The panel analogized the District Court's opinion with the Federal Circuit's language in In re Kubin, 561 F.3d 1351, 1359 (Fed. Cir. 2009), that when the asserted obviousness defense is akin to a defendant "'merely throw[ing] metaphorical darts at a board' in hopes of arriving at a successful result," where "the prior art gave either no indication of which parameters were critical or no direction as to which of many possible choices is likely to be successful"; this is not obviousness but hindsight reconstruction (further citing In re O'Farrell, 853 F.2d 894, 903 (Fed. Cir. 1988)).
The District Court's error, according to the opinion, was in failing to appreciate the lack of a known PK/PD relationship not just for the claimed formulation but for "any [cyclobenzaprine] formulation." The opinion notes that the District Court considered only routine experimentation to be needed for the skilled artisan to calculate the recited pharmacokinetic parameters (AUC and Tmax), specifically using computer modeling based on prior art data. The Federal Circuit opined that what was missing from this analysis was evidence that the formulation would be therapeutically effective. The opinion stated that the expert testimony was inconsistent with the District Court's conclusion, because all of the evidence related to the immediate release formulation. Thus, according to the Federal Circuit, the absence of a known PK/PD relationship would have prevented the skilled artisan from having any expectation of producing a therapeutically effective extended release formulation based on extrapolations from immediate release pharmacokinetic data. This deficiency also extended to comparisons of cmax values between immediate-release and extended-release formulations that fell outside the claimed range, because there was no evidence that these values were consistent with therapeutic effectiveness. And expert testimony regarding computer modeling was unconvincing to the Federal Circuit at least because the expert's own modeling could not produce PK values within the claimed ranges.
In a footnote, the Court explains why what may be common-sensical to a layperson (or judge) is not always sufficient in determining patent law questions such as obviousness:
While it might appear to a layperson that 129.5% is "about" 125%, expert testimony is necessary to establish how a person having ordinary skill in the art would perceive those figures. We have no way of knowing the importance of even small differences in these percentages in the absence of some evidence in the record addressing that point. See Ortho-McNeil Pharm., Inc. v. Caraco Pharm. Labs., Inc., 476 F.3d 1321, 1326 (Fed. Cir. 2007) ("The use of the word "about," avoids a strict numerical boundary to the specified parameter. Its range must be interpreted in its technological and stylistic context." (quoting Pall Corp. v. Micron Separations, Inc., 66 F.3d 1211, 1217 (Fed. Cir. 1995)))
The Court also noted the importance of such evidence in rendering an opinion that a claimed invention is obvious (the antithesis of "we know it when we see it" jurisprudence):
Evidence of obviousness, especially when that evidence is proffered in support of an "obvious-to-try" theory, is insufficient unless it indicates that the possible options skilled artisans would have encountered were "finite," "small," or "easily traversed," and that skilled artisans would have had a reason to select the route that produced the claimed invention. Ortho-McNeil Pharm., Inc. v. Mylan Labs., Inc., 520 F.3d 1358, 1364 (Fed. Cir. 2008) (citing KSR, 550 U.S. at 421). While it is true that Section 103 bars patentability unless "the improvement is more than the predictable use of prior art elements according to their established functions," KSR, 550 U.S. at 417, where the prior art, at best, "[gives] only general guidance as to the particular form of the claimed invention or how to achieve it," relying on an "obvious-to-try" theory to support an obviousness finding is "impermissible." In re Kubin, 561 F.3d at 1359 (quoting In re O'Farrell, 853 F.2d at 903)
The Court further found that the District Court engaged in hindsight reconstruction in some of the evidence relied upon for the obviousness determination, including expert testimony that merely "retraced the inventor's steps." Even this testimony did not contain any evidence that the skilled worker would have a reasonable expectation of success, according to the Federal Circuit, because the expert testified that whether achieving in the extended-release formulation equivalent blood concentrations to those produced using the immediate-release formulations would have a therapeutic effect "[d]epend[ing] on the relationship between blood levels and therapeutic effect," and the opinion found no evidence of this relationship.
The Federal Circuit then turned to the methodology used by the District Court to apply the objective indicia (also known as secondary considerations) of non-obviousness and found the methodology to be lacking. Specifically, the District Court first determined that the defendants had raised a prima facie case of obviousness based on the evidence from the prior art, and then assessed whether Cephalon rebutted that prima facie case with evidence of commercial success, unexpected results, long-felt but unfulfilled need and failure of others. This was error, according to the Federal Circuit, because obviousness must be considered in view of all the evidence, both from the application of the prior art and any objective evidence of non-obviousness, and the two-part procedure employed by the District Court was contrary to the proper application of Section 103 pursuant to Graham v. John Deere. In addition the Court found that the District Court had improperly shifted the burden of rebuttal to Cephalon instead of properly requiring defendants to bear the burden of providing clear and convincing evidence of obviousness. The opinion evinced some sympathy for the District Court's error, however, noting that "this court has inconsistently articulated the burden of proof applicable to an obviousness defense in district court litigation." The proper protocol was recited in Stratoflex, Inc. v. Aeroquip Corp., 713 F.2d 1530, 1538–39 (Fed. Cir. 1983):
It is jurisprudentially inappropriate to disregard any relevant evidence on any issue in any case, patent cases included. Thus evidence rising out of the so-called "secondary considerations" must always when present be considered en route to a determination of obviousness. . . . Indeed, evidence of secondary considerations may often be the most probative and cogent evidence in the record. It may often establish that an invention appearing to have been obvious in light of the prior art was not. It is to be considered as part of all the evidence, not just when the decisionmaker remains in doubt after reviewing the art.
Cases following this proper procedure cited in the opinion include Ruiz v. A.B. Chance Co., 234 F.3d 654, 663 (Fed. Cir. 2000); Cable Elec. Prods. v. Genmark, Inc., 770 F.2d 1015, 1026 (Fed. Cir. 1985); Simmons Fastener Corp. v. Illinois Tool Works, Inc., 739 F.2d 1573, 1575 (Fed. Cir. 1984); Richardson-Vicks Inc. v. Upjohn Co., 122 F.3d 1476, 1483 (Fed. Cir. 1997); Rockwell Int'l Corp. v. United States, 147 F.3d 1358, 1366 (Fed. Cir. 1998); and Kan. Jack, Inc. v. Kuhn, 719 F.2d 1144, 1150–51 (Fed. Cir. 1983). But the Court notes that other cases were not so consistently decided, including Innovention Toys, L.L.C. v. MGA Entm't, Inc., 637 F.3d 1314, 1323 (Fed. Cir. 2011); Transocean Offshore Deepwater Drilling, Inc. v. Maersk Contractors USA, Inc., 617 F.3d 1296, 1305 (Fed. Cir. 2010); Muniauction, Inc. v. Thomson Corp., 532 F.3d 1318, 1327 (Fed. Cir. 2008); Eli Lilly & Co. v. Zenith Goldline Pharms., Inc., 471 F.3d 1369, 1380 (Fed. Cir. 2006); Iron Grip Barbell Co. v. USA Sports, Inc., 392 F.3d 1317, 1325 (Fed. Cir. 2004); and WMS Gaming Inc. v. Int'l Game Tech., 184 F.3d 1339, 1359 (Fed. Cir. 1999). (It should not escape our notice that deviation from well-established principles has occurred with greater frequency over the past decade, after many of the original members of the Court have left the bench.) But even where panels have used language non-rigorously, this panel opines that those panels have consistently recognized that "a fact finder must consider all evidence of obviousness and nonobviousness before reaching a determination" and in error the District Court did not apply the proper analysis in this case. The opinion also asserts that the prescribed methodology that prevents the burden-shifting entertained by the District Court has support in Supreme Court precedent (a useful inclusion in an era where disgruntled litigants are prone to petition for certiorari with enhanced expectations for Supreme Court review of any Federal Circuit opinion), citing a wide range of predominantly non-patent cases for this proposition. From Graham:
Such secondary considerations as commercial success, long felt but unsolved needs, failure of others, etc., might be utilized to give light to the circumstances surrounding the origin of the subject matter sought to be patented," along with the scope and content of the prior art, the differences between the prior art and the claims at issue, and the level of ordinary skill in the pertinent art.
383 U.S. at 17–18, and, "[n]otably, the Court did not characterize the objective factors as after-the-fact considerations or relegate them to 'secondary status.'"
Finally in this regard, the opinion notes the "soundness" of a court considering all the evidence including the objective indicia, to prevent "judicial hunches" and hindsight bias created by the existence of the claimed invention. This was at least part of the District Court's error appreciated by the Federal Circuit, which further opined that the evidence of long-felt but unfulfilled need and failure of others "strongly support a conclusion of nonobviousness" and were "particularly telling" (while being unpersuaded that Cephalon had provided sufficient proof of unexpected results or commercial to support its non-obviousness position).
The Court's affirmance of the District Court's finding of no best mode violation was unremarkable, and it dismissed the appeal of the injunction as being premature. Finally, although the Court noted that the parties had filed a Rule 54(b) motion only after the fact (a nunc pro tunc filing), the panel accepted jurisdiction while noting it did not encourage parties to rely on the Court's largess rather than following the proper procedure from the outset.
In re Cyclobenzaprine Hydrochloride Extended-Release Capsule Patent Litigation (Fed. Cir. 2012)
Panel: Circuit Judges Newman, O'Malley, and Reyna
Opinion by Circuit Judge O'Malley
 Law Seminars International (LSI) will be offering a one-hour telebriefing on the "FDA's New Biosimilar Guidances" on May 15, 2012 from 3:00 – 4:00 pm (Eastern). Timothy J. Shea, Jr. of Sterne Kessler will moderate a panel including Jennifer L. Fox of Brinks Hofer Gilson & Lione and Dr. Denise M. Kettelberger of Faegre Baker Daniels. The panel will discuss the practical and legal implications of the proposed guidance documents issued by the FDA in February, and will consider strategies for companies seeking to market biosimilar products and for brand companies seeking to maintain market share in the face of biosimilar competition. Among the topics to be covered are:
Law Seminars International (LSI) will be offering a one-hour telebriefing on the "FDA's New Biosimilar Guidances" on May 15, 2012 from 3:00 – 4:00 pm (Eastern). Timothy J. Shea, Jr. of Sterne Kessler will moderate a panel including Jennifer L. Fox of Brinks Hofer Gilson & Lione and Dr. Denise M. Kettelberger of Faegre Baker Daniels. The panel will discuss the practical and legal implications of the proposed guidance documents issued by the FDA in February, and will consider strategies for companies seeking to market biosimilar products and for brand companies seeking to maintain market share in the face of biosimilar competition. Among the topics to be covered are:









 May 8, 2012 –
May 8, 2012 – 
