By Kevin E. Noonan —
 Appellant Myriad Genetics (the real "target" in interest of the ACLU/PubPat lawsuit over isolated human DNA molecules) filed its Supplemental Brief pursuant to Federal Circuit order in the remand of AMP v. USPTO. Not surprisingly, the brief urges the Court to reaffirm its judgment that claims to isolated DNA molecules are patent-eligible. Perhaps more surprisingly, the brief argues that Claim 20 is not before the Court on remand, but that if the Court considers it then the Court should recognize and reaffirm its prior determination that Claim 20 is patent-eligible.
Appellant Myriad Genetics (the real "target" in interest of the ACLU/PubPat lawsuit over isolated human DNA molecules) filed its Supplemental Brief pursuant to Federal Circuit order in the remand of AMP v. USPTO. Not surprisingly, the brief urges the Court to reaffirm its judgment that claims to isolated DNA molecules are patent-eligible. Perhaps more surprisingly, the brief argues that Claim 20 is not before the Court on remand, but that if the Court considers it then the Court should recognize and reaffirm its prior determination that Claim 20 is patent-eligible.
Myriad argues that the Supreme Court's decision in Mayo v. Prometheus does not affect Federal Circuit's decision that Myriad's composition of matter claims are patent eligible. The brief uses the Supreme Court's own language and considerations rather than their own, amici, or the Federal Circuit's rationales in making this argument. This language includes the Court's express intent not to "break new ground" and merely to provide "an examination of the particular [method] claims before [it] in light of the Court's precedents." Supreme Court precedents cited in the brief as "supporting" the Court's Mayo decision include Bilski v. Kappos, Diamond v. Diehr, Parker v. Flook, Gottschalk v. Benson, O'Reilly v. Morse, and Neilson v. Hartford (the British case); all cases involved solely method claims, according to the brief. Myriad asserts that the Mayo decision by its own terms is limited to method claims, and consistent with this earlier precedent. When the Court wishes to address composition of matter or manufacture claims patent-eligibility, as it did in Diamond v. Chakrabarty, the Court cites other precedent – Hartranft v. Weigmann and Funk Bros. Seed Co. v. Kalo Inoculant Co. — as precedent. And the brief notes that "no appellate court has ruled a product patent ineligible since the 1952 Act," citing In re Kratz, In re Chakrabarty, In re Bergstrom, and Merck & Co. v. Olin Mathieson Chem. Corp. in addition to In re Bergy.
The brief asserts that the Mayo decision stands for there being a requirement for something "significantly more" than reciting a "law of nature" coupled with "well-understood, routine, conventional activity." By satisfying the Chakrabarty standard, Myriad argues that the composition of matter claims satisfy the Mayo standard. According to the brief, the "overarching" question posed by Mayo is whether there is "invention" and that the changes in the naturally occurring genetic materials as claimed by Myriad satisfy that requirement. The brief expressly cites In re Bergy as the paradigm for the current situation before the Federal Circuit. In that case, there was a decision by the appellate court (the Court of Customs and Patent Appeals, C.C.P.A.) on composition of matter claims, later vacated by the Supreme Court and remanded for reconsideration over an intervening Supreme Court decision on method claims (Parker v. Flook). The CCPA (in an opinion by Judge G.S. Rich) found its earlier decision, that an isolated and purified culture of a naturally occurring bacterium was patent eligible, to be unaffected by the intervening Supreme Court precedent. Myriad contends that "[t]he same result is compelled here" because "Mayo sheds no new light on the patent eligibility of the isolated DNA claims" and thus the Mayo decision "should not alter the Court's prior judgment."
The brief argues that the Supreme Court standards for patent eligibility for method claims and composition of matter claims are different, and Myriad's claims satisfy the Chakrabarty standard that the claimed compositions have "a distinctive name, character and use." The brief characterizes the Mayo decision as requiring "the application of human ingenuity" as a requirement for patent-eligibility, and cites Mayo for the proposition that, for composition of matter claims, "a novel and useful structure is patent-eligible," based on Mackay Radio & Tel. Co. v. RCA. This is because "[e]very product has its origins in something that exists in nature" (echoing Chief Judge Rader's comment that "all we have is nature"), according to the brief. Thus, there is not (and indeed, cannot) be a requirement that all inventions be "wholly artificial"; "[h]uman ingenuity must simply have played a role," the brief argues. The Court's previous, now vacated decision is consistent with these standards according to Myriad.
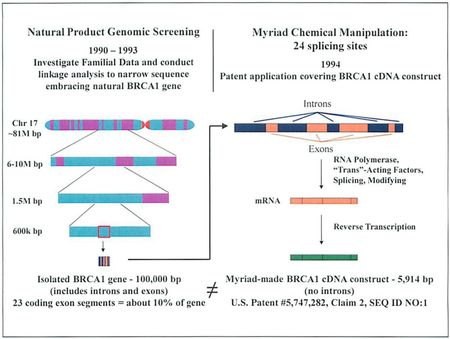
In deference perhaps to the extent to which the Mayo decision blended considerations other than subject matter eligibility into the criteria to be applied by courts (including, specifically, considerations that resemble obviousness), the brief explains that even the isolation of the BRCA genes involved unconventional, non-routine methods that were not well-understood and, indeed, were contrary to conventional wisdom on how to isolate a gene. Reminding the Court that these method steps are not part of the claim, the brief argues that they were nevertheless "groundbreaking" and "a major breakthrough." Myriad's brief takes pains to explain these steps and their unconventionality. The brief also includes what can only be described as "secondary considerations" or "objective indicia" on nonobviousness, including that the methods used and that their successfulness were surprising and unexpected. The brief also ties the inventors' decision to patent the genes, and disclose the information regarding them thereby, as enabling "thousands of researchers to publish thousands of papers" on the genes.
In a footnote, the brief makes creative use of the Court's discussion in Chakrabarty regarding the Plant Protection Act (which the Patent Office used to argue that Congress had not authorized utility patents on "living" inventions). Here, Myriad argues that by creating the PPA, Congress determined that "the work of the plant breeder in aid of nature was a patentable invention." This focuses the inquiry on the difference between what is naturally occurring and what is the product of human ingenuity, a standard reinforced by the Court's decision in J.E.M. Ag Supply Inc. v. Pioneer Hybrid Int'l. Inc. (which extended the scope of plant patent protection, under appropriate circumstances, to utility patenting under Section 101).
And in another footnote, the brief asserts that, under plaintiffs' reading, the Chakrabarty bacterium would have been patent-ineligible:
That bacterium was identical to the naturally occurring one, except for a few small pieces of DNA that had been transferred to it with the aid of human researchers (essentially through a process of selective breeding). It had the same genome and same internal components. It also retained all of its original functions and properties; it just gained a few new ones. In fact, the invention in Chakrabarty merely added known utilities from one bacterium into another (by contrast, the molecules in the present case gave rise to entirely new utilities that were impossible before, such as sequencing a patient's BRCA1 gene).
The policy concerns raised by the Supreme Court in rendering its Mayo decision — for example, that the claims monopolize "the basic tools of scientific and technological work" — are not to the contrary. The brief argues that the Supreme Court has cautioned against "categorical rules" for patent eligibility and requires a "fact-intensive analysis" of the particular claim(s) before it when deciding questions of patent eligibility. And the brief cautions that, if the law should be changed or clarified to exclude patent eligibility for DNA claims nunc pro tunc, the resulting upset of settled expectations is for Congress, not the courts, to decide (characterizing the contrary as "judicial lawmaking"). The role of the PTO in granting patents to isolated DNA molecules, and Congressional disinterest and inaction in modifying the law regarding composition of matter claims are factors to be considered by the Court in reconsidering its earlier determination that the composition of matter claims are patent eligible, Myriad argues.
The brief gets to the heart of the matter with regard to the mischief that the Court could wreak on the patent system by considering the effects of ruling for plaintiffs outside the area of DNA claims:
The impact of this case is not limited to human genetics. In 2012, the era of human gene discovery has been over for more than a decade, and human isolate DNA patents are in their twilight. But the consequence of ruling these claims ineligible would be tragic, and would reach far beyond this field and industry. Striking them down, despite the reliance of the inventive and investing communities on the well-established rule, would send a shiver up the spines of future investors and commercial research and development — and not just in molecular diagnostics. Such a ruling would additionally cast a pall on other innovation industries whose existence depends on patent protection.
In a footnote, the brief enumerated such ancillary consequences to include pharmaceuticals, antibiotics, immunosuppressants, biologics drugs and viruses, vaccines, human therapeutic proteins such as erythropoietin, enzymes used for producing biofuels, nutritionally improved fruits and vegetables, biological pesticides, flavorants, dyes and enzyme used, for example, in detergents.
And the brief reminds the Court that while all claims are "pre-emptive" the standard is that they not be unduly preemptive. Myriad's claims are not, the brief argues, because they are claiming just what the inventors invented: isolated BRCA gene encoding DNA molecules, "not any of the other 20,000 human or other genes." Even claims to the BRCA genes themselves are not unduly preemptive, as evidenced by the cited scientific research and other ways to test for hereditary breast cancer that are not impacted by Myriad's DNA claims.
With regard to Claim 20, the brief's argument that this claim is not properly before the Federal Circuit on remand is based, in part, on the plain language of the certiorari petition which was limited to the composition of matter claims and affirmatively stated that all of the method claims were not appealed — "None of the method claims is the subject of this [certiorari] petition" — so that there is no basis for further review. In any event, Myriad argues that Claim 20 is patent eligible because it encompasses "significantly more than" a "law of nature" and "well-understood, routine, conventional activity." That "more than" includes the transformed cell, which is not naturally occurring, particularly insofar as it has been genetically engineered to include an exogenous human BRCA gene, and these factors are enough to distinguish Claim 20 from the Prometheus claims in Mayo and to be patent eligible.
Oral argument is set for Friday, July 20th and will be available on the Federal Circuit website shortly thereafter.