 The
The
Intellectual Property Law Association of Chicago (IPLAC) will be offering a program
on "Coping With The New First-Inventor-to-File Prior Art Law: What Patent
Prosecutors Need to Know and Do Before and After the Effective Date" on January
25, 2013 from 12:00 to 2:30 pm (Central) at the DePaul University Law School in
Chicago, IL. Edward Manzo of Husch
Blackwell LLP, Joseph Hetz of Brinks Hofer Gilson & Lione, John Paniaguas
of Katten Munchin Rosenman LLP, and George Wheeler of McAndrews, Held &
Malloy, Ltd. will provide patent prosecutors with practical tips and strategies
to file and prosecute patent applications in light of the imminent new
definition of prior art under the Leahy-Smith America Invents Act (AIA),
enacted on September 16, 2011, and soon to be effective. The registration fee for the program is $35
for IPLAC members, $45 for non-members, $25 for students (other than DePaul
students, for whom there is no fee). Those
interested in registering for event can do so here.

Patent Law Weblog
recent posts
- USPTO Announces That It Has Turned the Corner on Unexamined Application Backlog
- Reasons for the PTAB’s Priority Determination in Broad’s Favor (Perhaps)
- Mexico Publishes Amendments to Intellectual Property Law
- PTAB (Again) Awards Priority of Invention to Broad in Interference No. 106,115
- Argentina Repeals Pharmaceutical Patent Examination Guidelines
about
-
-
By Kevin E. Noonan —
 As it has frequently in
As it has frequently in
the past decade, the Federal Trade Commission on Thursday released a Report on
the frequency of reverse payment settlement agreements in ANDA litigation
between generic and branded drug makers, pursuant to its authority to
scrutinize all ANDA agreements under the provisions of the Medicare
Modernization Act of 2003.The FTC Report contains
information from the 2012 fiscal year (from October 1, 2011 through September
30, 2012). There were 40 reverse payment
agreements among 140 "final resolutions" of ANDA litigation. According to the Report, these agreements
involved 31 different branded drugs having total combined sales of $8.3
billion. Nineteen of the 40 agreements
contained provisions wherein the branded drug maker agreed not to bring an "authorized
generic" version of the branded drug to market (thus removing a strong
competitor for the generic drug), at least for some period of time. The Report also notes that seven branded
drugs entered into "multiple" reverse payment settlements, i.e., with different generic
challengers. The Report compares this
trend with historical data from 2004, noting that in one case a branded drug
maker had entered into reverse payment settlement agreements with ten generic
challengers, with 14 branded drugs being involved in payments to three or more
generic drug makers.Of the other 100
settlement agreements, 81 contained provisions without express payments but
with restrictions on time for generic entry, which the final 19 agreements
contained no restrictions of the generic challenger.Forty-three of these
140 settlements were between the branded drug maker and the first ANDA filer. Of these, 23 settlement agreements had provisions both for payments and delay
or other restrictions on generic drug entry with another 16 agreements
providing for generic delay but no express compensation.The Report draws
conclusions from this data, the most significant of which is that there were a "record"
number of reverse payment settlement agreements in fiscal 2012. The 40 agreements containing reverse payment
provisions increased from the 28 such agreements in fiscal 2011, and the 19
agreements providing for no authorized generic entry (but without express
payments) were also a record (up from 11 in fiscal 2012, perhaps reflecting the
effects of the FTC's campaign against agreements containing payment provisions
to generic drugmakers).In the end, however,
the FTC was forced to admit that "[d]espite the record number of potential
pay-for delay settlements in FY 2012, the vast majority of patent settlements
(greater than 70%) continued to be resolved without compensation to the generic
manufacturer," at least suggesting that the Commission itself has created
this particular tempest in its teapot. The Report concludes with a table comparing rates of ANDA settlements
and the frequency of reverse payment settlement agreements since FY 2004.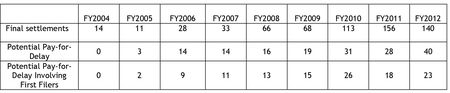
Looking at these data dispassionately,
the number of settlements of ANDA litigation has decreased, while the number of
settlements having some economic incentive for generic drug makers has
increased. However, the data also show
that this economic incentive in the form of direct payments has actually
decreased, and the Commission's statistics show an increase because it has
included agreements with "no authorized generic" provisions as "potential
pay-for-delay" agreements.In a press release
accompany the Report, the Commission highlighted these increases,
characterizing them as having "significantly increased" and
comprising a "record number" of such agreements. (As is its wont, the Commission characterizes
these agreements as "pay for delay," even for agreements with no
express payments from the branded to the generic drugmaker.) But the Commission considers even these
agreement to be "potentially anticompetitive," saying that "[s]uch 'no-AG' [authorized generic] promises are valuable to generic firms,
as they significantly reduce the level of competition the new generic entrant
will face, allowing the generic firm to secure greater market share and extract
higher prices from consumers." The
latter statement is, of course, at least unlikely to be untrue: the value of a "no-AG"
promise is that the generic drug company will make more money, of course, but
not by charging higher prices but by recouping more of the total generic market
as a consequence of not having to split the market with the authorized generic
product.FTC Chairman Jon Leibowitz added his own
ideological opinion on these results, saying:Sadly, this year's report makes it clear that
the problem of pay-for-delay is getting worse, not better. . . . More
and more brand and generic drug companies are engaging in these sweetheart
deals, and consumers continue to pay the price. Until this issue is
resolved, we will all suffer the consequences of delayed generic entry — higher
prices for consumers, businesses, and the U.S. taxpayer.This sentiment is
continued in the remainder of the press release, which correctly notes that the
average "delay" for generic drug entry is 17 months, but ignores the
reality that this is a delay from a putative successful ANDA challenge. It does not take into effect the acceleration
of generic drug entry that results from settlement of ANDA litigation rather
than that proportion of these cases where the generic drug challenger loses and
is barred from the marketplace until the challenged Orange Book listed patents
expire. While this emphasis on delay is
understandable in view of the Commission's ideological biases (and its
unsupported and unrealistic belief that only holders of "bad" patents
settle ANDA litigation), it does not give the public a clear picture of the
actual circumstances behind the data the Commission presents.This may be the last
Report from the Commission on reverse payment settlement agreement frequency,
should the FTC prevail in its challenge to these agreements before the Supreme
Court (FTC v. Watson Pharmaceuticals). That would be the culmination of a steady
stream of Commission, district court, and appellate court challenges to these
agreements, as well as several stalled legislative efforts. The Commission's position has been rejected
often by several appellate courts (including the 2nd, 9th,
11th and Federal Circuit courts of appeal) based on an analysis of
the business and legal realities not overburdened with the Commission's
ideological bias (i.e., that any delay in generic drug entry is anticompetitive
per se). The Supreme Court should have the last word
on this issue. -
By Donald
Zuhn — Last month, the World Intellectual
Last month, the World Intellectual
Property Organization (WIPO) issued a report indicating that worldwide
intellectual property filings showed strong growth in 2011 despite a global
economy that continued to underperform. In
particular, the report on 2012 World Intellectual Property Indicators
notes that patent filings grew by 7.8% in 2011, the second consecutive year
with more than 7% growth.The report points out that in 2011, China's
State Intellectual Property Office (SIPO) became the largest patent office in
the world in terms of the number of patent applications received. SIPO overtook the U.S. Patent and Trademark (USPTO)
in 2011 after passing the Japan Patent Office (JPO) in 2010. The final tallies for 2011 show that SIPO
received 526,412 applications, the USPTO received 503,582 applications, and the
JPO received 342,610 applications. The
report notes that over the past 100 years, only three patent offices — the
USPTO, JPO, and Deutsches Patent- und Markenamt (DPMA) or German Patent Office
— have occupied the top spot. In
addition to China's milestone, worldwide patent filings exceeded the 2 million
mark for the first time in 2011, with some 2.14 million applications being
filed. The worldwide backlog of
unprocessed applications, which numbers some 4.8 million applications, dropped
by 4.9% in 2011 after falling by 3.3% in 2010. With respect to country of origin,
With respect to country of origin,
residents of Japan filed the largest number of patent applications worldwide
(472,417), followed by Chinese residents, and then U.S. residents. As for subject matter, the report indicates
that applications directed to digital communications experienced the
highest average annual growth rate (+8.1%) between 2006 and 2010. Over the same period, biotech applications
showed 3.0% growth and pharmaceutical applications dropped 1.6%. With respect to 2011 patent
grants, the report indicates that Japan took the top spot with 238,323 patents
issued and the U.S. placed second with 224,505 patents. -
By Donald Zuhn —
 The U.S. Patent and
The U.S. Patent and
Trademark Office published a notice in the Federal Register (78 Fed. Reg.
2960) earlier today requesting
public comment regarding potential practices that applicants can employ in the
drafting of patent applications to facilitate examination and bring more
certainty to the scope of issued patents.
Although the instant notice is directed to potential practices that
applicants can employ, the notice indicates that the Office intends to issue a
separate notice identifying potential practices that the Office can employ to
also facilitate examination and bring more certainty to the scope of issued
patents.Today's notice sets forth a
list of nine "potential practice changes that applicants can employ to augment
the quality of issued patents" and seeks input as to whether any of the
practices should be used by applicants during the preparation of an application
to place the application in a better condition for examination. The list of practices is divided into two
groups, one group related to clarifying the scope of the claims and one group
related to clarifying the meaning of claim terms in the specification. The first group of potential practices includes:1. Presenting claims in a multi-part format by
way of a standardized template that places each claim component in separate,
clearly marked, and designated fields (e.g.,
preamble, transitional phrase, and individual claim limitations).2. Identifying corresponding support in the
specification for each of the claim limitations (e.g., using a claim chart).3. Indicating whether
examples in the specification are intended to be limiting or merely
illustrative.4. Identifying whether the claim preamble is
intended to be a limitation on claim scope.5. Expressly identifying clauses within
particular claim limitations for which the inventor intends to invoke 35 U.S.C.
§ 112(f) (which pertains to means-plus-function limitations for applications
filed on or after September 16, 2012) and pointing out where in the
specification corresponding structures, materials, or acts are disclosed that
are linked to the identified § 112(f) claim limitations.6. Using textual and graphical notation systems
known in the art to disclose algorithms in support of computer-implemented
claim limitations, such as C-like pseudo-code or XML-like schemas for textual notation
and Unified Modeling Language (UML) for graphical notation.The second group of practices includes:
1. Indicating whether terms of degree — such as
substantially, approximately, about, essentially — have a lay or technical
meaning and explaining the scope of such terms.2. Including in the specification a glossary of
potentially ambiguous, distinctive, and specialized terms used in the
specification and/or claims.3. Designating, at the time of filing the application,
a default dictionary or dictionaries to be used in ascertaining the meaning of
the claim terms.Comments can be sent by
e-mail to QualityApplications_Comments@uspto.gov, or by regular mail
addressed to: Mail Stop Comments — Patents,
Commissioner for Patents, P.O. Box 1450,
Alexandria, VA 22313–1450, marked to the attention of Nicole D. Haines. The deadline for submitting comments in
response to the notice is March 15, 2013. -
By Kevin E. Noonan —
 The
The
tendency for members of the House of Representatives to exhibit ignorance of
the patent system, so amply demonstrated in the "debate" over H.R.
6621 at the end of the last Congress (see "Congressional Misunderstandings (Apparently) Motivate H.R. 6621"), persists in the 113th
Congress. The most recent example is
H.R. 193, the ''Seed Availability and Competition Act of 2013''
introduced on January 4th by Representative Marcy Kaptur, D-OH
(9th District, which includes Toledo). The seeming intent of the bill is to
substitute government bureaucrats from the Departments of Agriculture and the
Treasury for private patentees in enforcing patents on genetically engineered
seed. In addition to amounting to a
compulsory license, Rep. Kaptur has evidently never considered that, sometimes,
the scariest sentence in the English language is "I'm from the Federal
government, and I'm here to help." This is not surprising; Rep. Kaptur was recently elected to her 15th
term, and her background is not in patent law or agriculture: her professional
training is in urban planning.The bill requires "persons [farmers] who seek
to retain seed harvested from the planting of patented seeds to register with
the Secretary of Agriculture and pay fees set by the Secretary for retaining
such seed, and for other purposes." Specifically, any farmer who plants patented seed or
seed "derived from" patented seed to retain the seed for replanting
is subject to two requirements. The farmer
must submit a notice to the Secretary of Agriculture of the intent to retain
patented seed for replanting, (§ 2(a)(1)) and to pay a fee
established by the Secretary and dependent on the "type and quantity"
of seed that can be retained and "any other information" the
Secretary determines to be "appropriate" (§ 2(a)(2)). The fee is paid to the Federal government (§ 2(b)) and
deposited into a "Patented Seed Fund" established in the Treasury (§ 2(e)(1)(2)). The fees will
be refunded to any farmer who can establish that, after paying the fees,
natural disaster or "related" circumstances prevented them from
replanting the seed (§ 2(c)).The
monies collected from the farmers is then paid to the patent holders by the
goverment (§ 2(d)). The motivation for all these
seeming legal and economic inefficiencies finally becomes evident in § 2(f), which provides that any farmer who complies with the
provisions of the bill (if enacted) "shall not be bound" by any
contractual, patent royalty, or licensing fees. Not wishing to be considered
Not wishing to be considered
un-American, Rep. Kaptur (at left) included in the bill a provision (§ 3) whereby the Treasury Department will collect a tariff
amounting to the difference between the royalties or licensing fees imposed
upon purchasers of patented seed in the U.S. and any lower royalties or
licensing fees paid by purchasers of patented seeds in a foreign country and
then imported into the U.S. Those
tariffs are paid into the Patented Seed Fund, presumably for payment to patent
holders who have negotiated the lesser fees in the foreign country in the first
place.It is hard to know where to
begin. Clearly the political controversy
engendered by the many patented seed cases brought by patentees against farmers
over the past decade, and the recent kerfuffle produced by the Public Patent
Foundation in its suit on behalf or "organic" farmers came to Rep.
Kaptur's attention. And in some ways
this is a classic "split the baby" result, where the government (and
its sensitivity to political considerations) can determine the "appropriate"
royalty for replanted seed and distribute it to the patent holders. The tariff provisions might also be seen as a
boon to those patentees who were unable to obtain royalties or patent licensing
fee abroad commensurate with the fees they collect from U.S. farmers.But in reality, of course,
the bill imposes a compulsory license on holders of genetically engineered seed
patents, who have resisted the political pressure to permit farmers to replant
seed instead of purchasing seed for each planting. Although this has imposed legal and public
relations costs on these patentees, the patent grant permits them to impose
these restrictions (unless and until the Supreme Court decides otherwise in Bowman v. Monsanto). But regardless of which side has the better policy
argument in that debate, Rep. Kaptur's bill is not a remedy required by the
politics or economics of the situation. Indeed, it would just impose another
government bureaucracy on U.S. agriculture that would not promote either
agriculture or technological progress.The bill has been referred to
the House subcommittee on Rural Development, Research,
Biotechnology, and Foreign Agriculture for further consideration.Text
of the bill:A BILL
To require persons who seek to retain seed
harvested from the planting of patented seeds to register with the Secretary of
Agriculture and pay fees set by the Secretary for retaining such seed, and for
other purposes.Be it enacted by the Senate and House of Representatives
of the United States of America in Congress assembled,SECTION 1. SHORT TITLE.
This Act may be cited as the ''Seed Availability
and 5 Competition Act of 2013''.SEC.
2. RETAINING PATENTED SEED.(a) REGISTRATION.—Any person who plants patented
seed or seed derived from patented seed may retain seed from the harvest of the
planted seed for replanting by that person if that person—
(1) submits to the Secretary of Agriculture notice,
in such form as the Secretary may require, of the type and quantity of seed to
be retained and any other information the Secretary determines to be appropriate;
and
(2) pays the fee established by the Secretary pursuant
to subsection (b) for the type and quantity of seed retained.
(b) FEES.—The Secretary of Agriculture shall establish
a fee to be paid by a person pursuant to subsection (a)(2) based on the type
and quantity of seed retained. The Secretary shall deposit amounts
collected pursuant to subsection (a)(2) in the Patented Seed Fund established
under subsection (e)(1).
(c) REFUNDS.—The Secretary of Agriculture may refund
or make an adjustment of the fee paid pursuant to subsection (a)(2) when the
person is unable to plant or harvest the retained seed as a result of a natural
disaster or related condition and under such other circumstances as the
Secretary considers such refund or adjustment appropriate.
(d) DISTRIBUTIONS.—The Secretary of
Agriculture shall pay the collected fees to the appropriate patent holders, at
a frequency that the Secretary determines is appropriate, from the Patented
Seed Fund established under subsection (e)(1), taking into consideration the
possibility of refunds pursuant to subsection (c).
(e) PATENTED SEED FUND.—
(1) ESTABLISHMENT.—There is established in the
Treasury of the United States a fund to be known as the ''Patented Seed Fund'',
consisting of such amounts as may be received by the Secretary and deposited
into such Fund as provided in this section.
(2) ADMINISTRATION.—The Fund shall be administered
by the Secretary of Agriculture and all moneys in the Fund shall be distributed
solely by the Secretary in accordance with this section and shall not be
distributed or appropriated for any other purpose. Amounts in the Fund are
available without further appropriation and until expended to make payments to
patent holders.
(f) INAPPLICABILITY OF CONTRACTS AND PATENT FEES.—A person who retains seed under
subsection (a) from the harvest of patented seed or seed derived from patented
seed shall not be bound by any contractual limitation on retaining such seed,
or by any requirement to pay royalties or licensing or other fees, by reason of
the patent, for retaining such seed.
(g) DEFINITION.—In this section, the term ''patented
seed'' means seed for which a person holds a valid patent.SEC. 3. TARIFF ON CERTAIN IMPORTED PRODUCTS.
(a) TARIFF.—In any case in which—
(1) genetically modified seed on which royalties or
licensing or other fees are charged by the owner of a patent on such seed to
persons purchasing the seed in the United States is exported, and
(2) no such fees, or a lesser amount of such fees,
are charged to purchasers of the exported seed in a foreign country, then there
shall be imposed on any product of the exported seed from that foreign country
that enters the customs territory of the United States a duty determined by the
Secretary of the Treasury, in addition to any duty that otherwise applies, in
an amount that recovers the difference between the fees paid by purchasers of
the seed in the United States and purchasers of the exported seed in that
country.
(b) DEPOSIT OF DUTIES.—There shall be deposited in
the Patented Seed Fund established under section 2(e)(1) the amount of all
duties collected under subsection (a) for distribution to the appropriate
patent holders in accordance with section 2(d).
(c) DEFINITIONS.—In
this section—
(1) the term ''genetically modified seed'' means any seed that
contains a genetically modified material, was produced with a genetically
modified material, or is descended from a seed that contained a genetically
modified material or was produced with a genetically modified material; and
(2) the term ''genetically modified material''
means material that has been altered at the molecular or cellular level by
means that are not possible under natural conditions or processes (including
recombinant DNA and RNA techniques, cell fusion, microencapsulation,
macroencapsulation, gene deletion and doubling, introducing a foreign gene,
and changing the positions of genes), other than a means consisting exclusively
of breeding, conjugation, fermentation, hybridization, in vitro fertilization,
tissue culture, or mutagenesis. -
By Sherri Oslick —
 About Court Report: Each week we will report briefly on recently filed biotech and pharma cases.
About Court Report: Each week we will report briefly on recently filed biotech and pharma cases.Allergan, Inc. v. Apotex Inc et al.
1:13-cv-00016; filed January 8, 2013 in the Middle District of North
Carolina• Plaintiff: Allergan, Inc.
• Defendants: Apotex Inc.; Apotex Corp.; Sandoz, Inc.; Hi-Tech Pharmacal
Co., Inc.; Watson Pharmaceuticals, Inc.; Watson Laboratories, Inc.; Watson
Pharma, Inc.Infringement of U.S. Patent No. 8,263,054 ("Method of Enhancing
Hair Growth," issued September 11, 2012) based on defendants' ANDA to
manufacture a generic version of Allergan's Latisse® (bimatoprost topical
solution/drops, 0.03%, used to treat inadequate or not enough eye lashes). View the complaint here.
Xenetic Biosciences plc v. Kappos
1:13-cv-00022; filed January 7, 2013 in the Eastern District of VirginiaReview and correction of the patent term adjustment calculation made by
the U.S. Patent and Trademark Office for U.S. Patent No. 8,217,154 ("Activated
Sialic Acid Derivatives for Protein Derivatisation and Conjugation,"
issued July 10, 2012). View the complaint here.
Novartis Pharmaceuticals Corp. et al. v. Alvogen Pine Brook Inc. et
al.
1:13-cv-00052; filed January 4, 2013 in the District Court of Delaware• Plaintiffs: Novartis Pharmaceuticals
Corp.; Novartis AG; Novartis Pharma AG; Novartis International
Pharmaceutical Ltd.; LTS Lohmann Therapie-Systeme AG
• Defendants: Alvogen Pine Brook Inc.; Alvogen Group Inc.Infringement of U.S. Patent Nos. 6,316,023 ("TTS Containing an
Antioxidant," issued November 13, 2001) and 6,335,031 (same title, issued
January 1, 2002) following a Paragraph IV certification as part of Alvogen's
filing of an ANDA to manufacture a generic version of Novartis' Exelon® Patch
(rivastigmine tartrate, used to treat mild to moderate dementia of the
Alzheimer's type, and mild to moderate dementia associated with Parkinson's
disease). View the complaint here.
Genetic Technologies Ltd. v. General Genetics Corp.
1:13-cv-00055; filed January 4, 2013 in the District Court of DelawareInfringement of U.S. Patent No. 5,612,179 ("Intron Sequence
Analysis Method for Detection of Adjacent Locus Alleles as Haplotypes,"
issued March 18, 1997) based on Natera's manufacture, use, sale, and offer for
sale of paternity, predisposition, and forensics testing. View the complaint here.
Astrazeneca AB et al. v. Dr. Reddy's Laboratories, Inc. et al.
3:13-cv-00091; filed January 4, 2013 in the District Court of New Jersey• Plaintiffs: Astrazeneca AB; Astrazeneca
LP; KBI-E Inc; Pozen Inc.
• Defendants: Dr. Reddy's
Laboratories, Inc.; Dr. Reddy's Laboratories, Ltd.Infringement of U.S. Patent Nos. 5,714,504 ("Compositions,"
issued February 3, 1988), 6,369,085 ("Form of S-omeprazole," issued
April 9, 2002), 7,411,070 ("same title," issued August 12 ,2008),
7,745,466 (same title, issued June 29, 2010), and 6,926,907 ("Pharmaceutical
Compositions for the Coordinated Delivery of NSAIDs," issued August 9,
2005) following a Paragraph IV certification as part of Dr. Reddy's filing of
an ANDA to manufacture a generic version of AstraZeneca's Vimovo® (naproxen and
esomeprazole magnesium, used to relieve the signs and symptoms of
osteoarthritis, rheumatoid arthritis, and ankylosing spondylitis, and to
decrease the risk of stomach (gastric) ulcers in patients at risk of developing
stomach ulcers from treatment with non-steroidal anti-inflammatory drugs
(NSAIDs)). View the complaint here.
Noven Pharmaceuticals, Inc. v. Kappos
1:13-cv-00017; filed January 4, 2013 in the Eastern District of VirginiaReview and correction of the patent term adjustment calculation made by
the U.S. Patent and Trademark Office for U.S. Patent No. 8,216,606 ("Dermal
Composition for Controlling Drug Flux Comprising Two Acrylic Adhesive Polymers
Having Different Functionalities and Different Solubility Parameters,"
issued July 10, 2012). View the complaint here.
Celldex Therapeutics Inc. v. Kappos
1:13-cv-00018; filed January 4, 2013 in the Eastern District of VirginiaReview and correction of the patent term adjustment calculation made by
the U.S. Patent and Trademark Office for U.S. Patent No. 8,236,318 ("Antibodies
That Bind Human Dendritic and Epithelial Cell 205 (DEC-205)," issued
August 7, 2012). View the complaint here.
Transcept Pharmaceuticals, Inc. v. Kappos
1:13-cv-00019; filed January 4, 2013 in the Eastern District of VirginiaReview and correction of the patent term adjustment calculation made by
the U.S. Patent and Trademark Office for U.S. Patent No. 8,242,131 ("Methods
of Treating Middle-of-the-Night Insomnia," issued August 14, 2012). View
the complaint here. -

January 16, 2013 – European biotech patent law update (D
Young & Co) – 4:00 am, 7:00 am, 12:00 pm (ET)January 17,
2013 – Global IP Protection and the Transition to the AIA (Intellectual Property Owners Association) – 1:00 – 2:00 pm (ET)January 17-18, 2013 – The Future of Patent Law (University of
San Diego School of Law) – San Diego, CAJanuary 22, 2013 – Top Patent Law Stories of 2012 (McDonnell
Boehnen Hulbert & Berghoff LLP) – 10:00 to
11:15 am (CT)January 22, 2013 – Patent Term Adjustments and Extensions: Recent Developments —
Leveraging Exelixis, Other Court
Decisions and Recent USPTO Rule Changes to Maximize PTAs and PTEs (Strafford) – 1:00 – 2:30 pm (EST)January 23-24, 2013 – The Comprehensive Guide to Patent Reform*** (American Conference
Institute) – New York, NYJanuary 24, 2013 – Canadian
IP Law Update — Practical Tips and Information for U.S. Attorneys (Smart & Biggar) – Chicago, ILJanuary 27, 2013 – Canadian
IP Law Update — Practical Tips and Information for U.S. Attorneys (Smart & Biggar) – Minneapolis, MNJanuary 29, 2013 – Combined Customer Partnership Meeting
of TC3700 and TC1600 (U.S. Patent and Trademark Office) – Alexandria, VAJanuary
29-30, 2013 – Biotech & Pharma Patent Litigation*** (C5) – Amsterdam, NetherlandsFebruary
4-5, 2013 – Naples Midwinter Patent Law Experts Conference (Akron University Law School) – Naples, FLFebruary 13, 2013 – Corporate Intellectual
Property Law Conference (Law Bulletin
Publishing Company) – Chicago, ILFebruary 14, 2013 – Last Chance! The Top Ten Things
In-House Counsel Need to Do to Prepare for The AIA’s March 2013 Deadline (McDonnell
Boehnen Hulbert & Berghoff LLP) – 10:00 am to 11:15 am (CST)February 20, 2013 – AIA Strategy Considerations for Patent Filings in
2013: Should You File on March 15 or March 16 (or Both)? (Intellectual Property Owners Association) – Washington, DCFebruary 20-22, 2013 – Intensive Patent Law Seminar (Chisum Patent Academy) – New York, NY
February
26-27, 2013 – Biotech & Pharmaceutical Patenting (IBC Legal) – Munich, GermanyFebruary 27-28, 2013 – Life Sciences
Collaborative Agreements and Acquisitions*** (American Conference
Institute) – New
York, NYMarch 5-6, 2013 – Medical Device
Patents*** (American Conference
Institute) – Chicago, IL***Patent Docs is a media partner of this conference or CLE
-
 The
The
Intellectual Property Owners Association (IPO) will offer a one-hour webinar entitled
"Global IP Protection and the Transition to the AIA" on January 17,
2013 beginning at 1:00 pm (ET). A panel
consisting of David Arthur, Associate
General Patent Counsel at Xerox Corporation; Richard Treanor of Oblon Spivak;
and Anthony Tridico, managing
partner of Finnegan's European office in Brussels will discuss the special
difficulties that IP owners who regularly file for patent protection overseas
may run into when new U.S. patent application rules come into effect in March,
as well as consider how the change in the definition of prior art in the AIA
will impact international patenting strategy.The
registration fee for the webinar is $120 (government and academic rates are
available upon request). Those
interested in registering for the webinar can do so here. -
 McDonnell
McDonnell
Boehnen Hulbert & Berghoff LLP will be offering a live webinar entitled
"Last Chance! The Top Ten Things
In-House Counsel Need to Do to Prepare for The AIA’s March 2013 Deadline"
on February 14, 2013 from 10:00 am to 11:15 am (CST). In a follow-up to their December 12, 2012
webinar, Grantland Drutchas and Patent
Docs author Dr. Donald Zuhn will discuss some of the things that you can do
while you still have time before the March 16, 2013 deadline, including:•
Prepare for the pitfalls of first-to file
•
Establish new priorities with your employees and with outside counsel
•
Assess commercialization/trade secret strategies
•
Consider the impact on your budgets
•
Review the changes from the AIA corrections bill (passed by the U.S. Congress
on January 1, 2013)While
there is no fee to participate, attendees must register in advance. Those wishing to register can do so here. CLE credit is pending for the states of
California, Georgia, Illinois, North Carolina, New Jersey, New York, and
Virginia. -
 The
The
first Naples Midwinter Patent Law Experts Conference will be held on February
4-5, 2013 at the Naples Ritz Carlton in Naples, FL. The conference, which is being sponsored by
the Akron University Law School, has been planned as a small, intimate
gathering of leaders of the profession including corporate, private and
academic sectors with a maximum participation
of no more than eighty persons. An
opening reception will be held from 8:30 to 10:00 pm on Monday, with an all day
working session to be held on Tuesday.
The working session on Tuesday will include presentations on the following topics:•
The AIA and Changes at the Patent
Office; New Legislation? — includes a morning keynote address by The Hon. Robert
Stoll, former Commissioner for Patents
for the U.S. Patent and Trademark Office
•
The "Unified" European Patent
•
The Supreme Court and the Federal
Circuit — includes a keynote luncheon speech by The Hon. Richard Linn, Circuit Judge, U.S. Court of Appeals for the
Federal Circuit
•
Obviousness/Inventive Step at the PTAB,
A Comparative View
•
A Corporate Management Perspective on
Global IP Litigation — panel includes The Hon. Sharon R. Barner, former Deputy Under Secretary of Commerce
and Deputy Director for the U.S. Patent and Trademark OfficeAdditional information regarding conference,
including a list of the speakers and panelists and detailed descriptions of the
conference sessions, can be found here or downloaded here. Those interested in attending
the conference should contact Dean Margaret E. Matejkovic, Associate Dean,
Akron University Law School at ema16@uakron.edu.