By
Andrew Williams —
 Can
Can
the claim term "substantially pure" mean two different things when
the specification uses the same term to refer to both an intermediate compound
and a final drug product in a specification? The Federal Circuit recently answered the question in the affirmative,
in Aventis Pharma. Inc. v. Amino
Chemicals Ltd., depending of course on the context that the term is used in
the claims. The Court also focused on
the claim term "substantially pure regioisomer," instead of the
truncated "substantially pure" from the lower court's decision, and determined
that the term required "largely but not wholly the para-regioisomer of the
intermediate of the structure shown, as compared to the meta isomer." Judge Bryson in dissent, however, found it
more compelling that the patentees appeared to use the term "substantially
pure" interchangeably in both the specification and the prosecution
history when referring to both the intermediate and final drug products. Therefore, he would have affirmed the lower
court's claim construction of "substantially pure" to require "at
least 98% purity with respect to all impurities."
The
technology at issue in this case was the process for producing large quantities
of fexofenadine, the active ingredient of Allegra® and Allegra-D®
24 hour. The prior art process of making
piperidine derivatives such as fexofenadine was inefficient because it required
purification steps after the derivatives were fully formed, and this added to
the both the cost and the time to obtain a pharmaceutically acceptable final
product. Dr. Thomas E. D'Ambra overcame
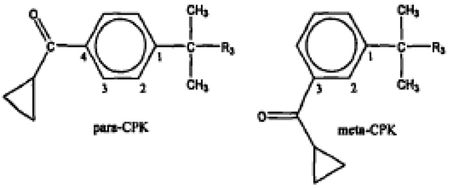
this problem by using piperidine and cyclopropylketone ("CPK")
intermediates earlier in the reaction. The CPK intermediate could take on at least two conformations, para-CPK
and meta-CPK:
but
it was the para-CPK that gave rise to the biologically active piperidine
derivatives. The molecule could also
adopt the ortho-CPK form, but it was rarely produced and was of little
biological efficacy. These different conformations
are referred to as regioisomers. The
prior art processes used a "Friedel-Crafts" reaction, which produced
a statistical admixture for the "second mixture of aromatic regioisomers"
of 67% meta-isomers and 33% para-isomers. The final products (third mixture) were then obtained by converting
these intermediates, although the ratio of the regioisomers persisted. It was therefore nearly impossible to
completely separate the para-isomers to pharmaceutical purity. Dr. D'Ambra's use of CPK intermediates made
it possible to obtain "substantially pure regioisomers" of para-CPK,
and consequently a subsequent "substantially pure" para-piperdine
derivative end product.
Claim
1 of the patent-in-suit, U.S. Patent No. 5,750,703 ("the '703 patent"),
provides (with the claim term highlighted):
1. A process of
preparing a piperidine derivative compound of the formula:
 wherein
wherein
R1 is
hydrogen or hydroxy;
R2 is
hydrogen;
or R1 and R2
taken together form a second bond between the carbon atoms bearing R1
and R2;
R3 is –COOH or –COOR4
;
R4 has 1 to 6 carbon atoms;
A, B, and D are
the substituents of their aromatic rings, each of which may be different or the
same, and are selected from the group consisting of hydrogen, halogens, alkyl, hydroxy,
alkoxy, or other substituents,
said process comprising:
providing a substantially pure regioisomer of the following formula:
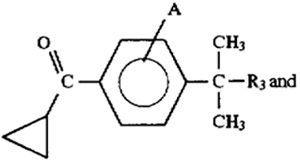 converting the substantially pure regioisomer to the
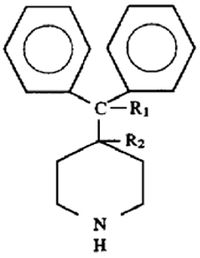
converting the substantially pure regioisomer to the
piperidine derivative compound with a piperidine compound of the formula:
 Even
Even
a casual reading of this claim revels that the term "substantially pure"
only modifies the intermediate regioisomer and not the final product. However, the specification uses the same term
in reference to the final product of the prior art process, and a divisional
patent, U.S. Patent No. 5,578,610 ("the '610 patent") has claims
drawn to substantially pure piperdine derivative compounds.
 The
The
procedural history of this case was complex and involved dozens of parties in
twenty different lawsuits. The
defendants had filed a Drug Master File that was referenced in the Abbreviated
New Drug Applications of Mylan Pharmaceuticals Inc. and Teva Pharmaceuticals
USA Inc. to market generic versions of antihistamines containing
fexofenadine. Upon submission of the
ANDAs, Aventis timely brought suit alleging, among other things, infringement
of the '703 patent. At the claim
construction phase, the lower court construed the term "substantially pure"
to mean "at least 98% purity with respect to all impurities." As a result, Aventis stipulated that it could
not prove infringement, and so the District Court entered final judgment to
allow Aventis to appeal the claim construction ruling.
In
reversing the lower court's decision, the Federal Circuit highlighted that
claims "must be construed in light of the appropriate context in which the
claim term is used" (citing Toro
Co. v. White Consol. Indus., Inc., 199 F.3d 1295 (Fed. Cir. 1999)). In fact, the Court pointed out that the same
claim term can have different constructions within the claims and the
specification. However, for the patent-in-suit,
the term "substantially pure" was only used in the claims to refer to the CPK intermediate. The Court apparently discounted the use of
the term in the claims of the related '610 case, in reference to the final
piperidine derivative end products. Nevertheless, what was important was how one skilled in the art would
understand that term in the context of the claims, and such a skilled artisan
would not have expected that an intermediate would have the same purity as the
final end product. After all, the
improvement was to provide a piperidine derivative end product of higher
regioisomeric purity with less extensive purification, and requiring a
consistent construction of this term ignored the distinct contexts in which the
terms are used. As a result, the Court
held that the proper construction of "substantially pure" required
different interpretations depending on whether it was modifying the intermediate
or the end product.
In
coming to this conclusion, the Court had to address the District Court's
reliance on the intrinsic record that Judge Bryson found persuasive in dissent. First, as Judge Bryson pointed out, the
specification stated:
Although the second mixture of regioisomers [an intermediate] and the third
mixture of regioisomers [the final piperidine derivative product] can be
analyzed by HPLC experiments, a practical separation to obtain gram quantities
of substantially pure regioisomers has not been achieved.
Each mixture (including the first [also an intermediate]), would be expected to
contain 33% of the para isomer and 67% of the meta isomer. Since these
components are inseparable, it has not been possible to obtain either of the
regioisomers in each mixture in substantially pure form.
'703
patent at col. 4 ll. 16-24. This was at
least part of the evidence that the patentee supposedly used the terms
interchangeably. However, as was pointed
out, this use was only in reference to the prior art — the patentee did not
refer to his own piperidine derivative end products as substantially pure in
the specification.
More
difficult to reconcile were comments made by the patentee during an
interference proceeding in the related '610 patent case. In a filing in that case, the patentee wrote "[w]hen
read in light of the specification, one skilled in the art would have
understood that the phrase 'substantially pure,' as used in claims
1-17 of [the '610 patent], to mean that the subject compound has a pharmaceutical
grade purity and is in a form purer than that attained by the prior art." Most of claims 1-17 recite a substantially
pure piperidine derivative end product. However, claim 12 of the '610 patent recites "a piperidine
derivative compound produced by a process comprising: providing a substantially
pure regioisomer . . . ." Therefore, different claims from claims 1-17 of the '610 patent contain
the "substantially pure" claim term to refer to both the intermediate
and the end product. Nevertheless, the
Federal Circuit's opinion found this to be of little help. It pointed out that the focus of the
interference proceeding was interpreting claims in reference to the piperidine
end product, and so the comments were only in regards to the "subject compound"
of the interference — the end product. And, since the Court had already determined that the "substantially
pure" should have different interpretations, it found no justification for
applying the definition provided for this term from the interference.
Finally,
having found the lower court's construction incorrect, the Federal Circuit
performed its own claim construction. It
noted that the term "substantially" is often not amenable to
numerical boundaries. It was clear from
the '703 patent that the regioisomeric purity should be greater than 67%
(because this was the purity in the prior art process), but it did not need to
be as high as 98%. So, the Court
borrowed a construction of the term from Ecolab,
Inc. v. Envirochem, Inc., 264 F.3d 1358 (Fed. Cir. 2001), in which "largely
but not wholly" was used as a flexible approach. Therefore, the Court adopted the definition of
"largely but not wholly the para regioisomer of the intermediate of the
structure shown, as compared to the meta isomer." The case was reversed and remanded to
determine the other issues in the case consistent with this claim construction.
MBHB
represented Aventis in the above appeal. To the extent that this case
summary contains any opinions, the opinions would be of Dr. Williams and not
Aventis or MBHB.
Aventis Pharmaceuticals
Inc. v. Amino Chemicals Ltd. (Fed. Cir.
2013)
Panel:
Circuit Judges Newman, Bryson, and Reyna
Opinion
by Circuit Judge Reyna; dissenting opinion by Circuit Judge Bryson
 Strafford
Strafford














