By Sherri Oslick —
 About Court Report: Each week we will report briefly on recently filed biotech and pharma cases.
About Court Report: Each week we will report briefly on recently filed biotech and pharma cases.
Alpex Pharma
S A et al. v. Zydus Pharmaceuticals USA Inc. et al.
1:13-cv-01143;
filed June 26, 2013 in the District Court of Delaware
• Plaintiffs:
Alpex Pharma S A; Citius Pharmaceuticals LLC; Prenzamax LLC
• Defendants:
Zydus Pharmaceuticals USA Inc.; Cadila Healthcare Ltd.
Infringement
of U.S. Patent No. 6,149,938 ("Process
for the Preparation of a Granulate Suitable to the Preparation of Rapidly
Disintegrable Mouth-Soluble Tablets and Compositions Obtained Thereby,"
issued November 21, 2000) following a Paragraph IV certification as part
of Zydus' filing of an ANDA to
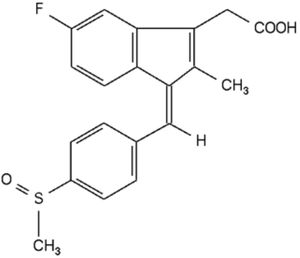
manufacture a generic version of Alpex's Suprenza® (phentermine hydrochloride,
used to treat obesity). View the
complaint here.
Alza
Corp. et al. v. Osmotica Kereskedelmi es Szolgaltato Kft
1:13-cv-04668;
filed June 26, 2013 in the Northern District of Illinois
• Plaintiffs:
Alza Corp.; Janssen Pharmaceuticals, Inc.
• Defendant:
Osmotica Kereskedelmi es Szolgaltato Kft
Alza
Corporation et al. v. Osmotica Kereskedelmi es Szolgaltato Kft et al.
1:13-cv-01126;
filed June 24, 2013 in the District Court of Delaware
• Plaintiffs:
Alza Corp.; Janssen Pharmaceuticals Inc.
• Defendants:
Osmotica Kereskedelmi es Szolgaltato Kft; Osmotica Pharmaceutical Corp.;
Norwich Pharmaceuticals Inc.
The
complaints in these cases are substantially identical. Infringement of U.S. Patent No. 8,163,798 ("Methods
and Devices for Providing Prolonged Drug Therapy," issued April 24, 2012)
following a Paragraph IV certification as part of Osmotica's filing of an ANDA
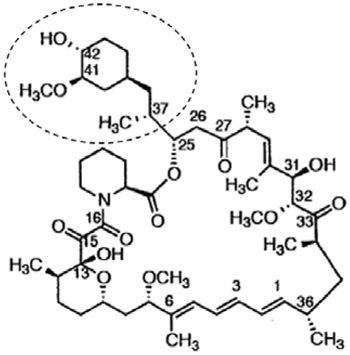
to manufacture a generic version of Alza's Concerta® (methylphenidate
hydrochloride, used to treat attention deficit hyperactivity disorder). View the Delaware complaint here.
Sandoz Inc.
v. Amgen Inc. et al.
3:13-cv-02904;
filed June 24, 2013 in the Northern District of California
• Plaintiff:
Sandoz Inc.
• Defendants:
Amgen Inc.; Hoffman-La Roche Inc.
Declaratory
judgment of non-infringement, invalidity, and unenforceability of U.S. Patent
Nos. 8,063,182 ("Human TNF Receptor Fusion Protein," issued November
22, 2011) and 8,163,522 ("Human TNF Receptor," issued April 24, 2012)
based on Sandoz's development of a biologic drug containing etanercept that
would compete with Amgen's Enbrel® (etanercept, used to treat rheumatoid
Arthritis, polyarticular juvenile idiopathic arthritis, psoriatic arthritis,
ankylosing spondylitis, and plaque psoriasis).
View the complaint here.
Pegasus
Laboratories, Inc. v. Wedgewood Village Pharmacy, Inc.
4:13-cv-00630;
filed June 24, 2013 in the Western District of Missouri
• Plaintiff:
Pegasus Laboratories, Inc.
• Defendants:
Wedgewood Village Pharmacy, Inc.
Infringement
of U.S. Patent Nos. 5,747,476 ("Treatment of Equine Protozoal Myeloencephalitis"
issued May 5, 1998), 6,255,308 (same title, issued July 3, 2001), and 6,448,252
(same title, issued September 10, 2002), licensed to Pegasus, based on
Wedgewood's marketing and sale of a sulfadiazine and pyrimethamine product for
treating horses with Equine Protozoal Myeloencephalitis caused by sarcocystic
neurona. View the complaint here.
Janssen
Products, L.P. et al. v. Lupin Ltd. et al.
2:13-cv-03891;
filed June 24, 2013 in the District Court of New Jersey
• Plaintiffs:
Janssen Products, L.P.; Janssen R&D Ireland
• Defendants:
Lupin Ltd.; Lupin Pharmaceuticals Inc.
Infringement
of U.S. Patent Nos. 7,700,645 ("Pseudopolymorphic Forms of a HIV Protease
Inhibitor," issued April 20, 2010), 7,126,015 ("Method for the
Preparation of Hexahydro-furo-[2,3-b]furan-3-ol," issued October 24, 2006),
and 7,595,408 ("Methods for the Preparation of
(3R,3aS,6aR)hexahydro-furo[2,3-b]furan-3-ol," issued September 29, 2009) following
a Paragraph IV certification as part of Lupin's filing of an ANDA to manufacture
a generic version of Janssen's Prezista® (darunavir, used to treat human
immunodeficiency virus (HIV-1) infection).
View the complaint here.
Pfizer et al.
vs. Alkem Laboratories, LTD
1:13-cv-04628;
filed June 24, 2013 in the Northern District of Illinois
• Plaintiffs:
Pfizer Inc.; UCB Pharma GmbH
• Defendant:
Alkem Laboratories, Ltd.
Pfizer Inc. et
al. v. Alkem Laboratories Ltd.
1:13-cv-01110;
filed June 21, 2013 in the District Court of Delaware
• Plaintiffs:
Pfizer Inc.; UCB Pharma GmbH
• Defendants:
Alkem Laboratories Ltd.
Pfizer Inc. et
al. v. Sandoz Inc.
1:13-cv-01111;
filed June 21, 2013 in the District Court of Delaware
• Plaintiffs:
Pfizer Inc.; UCB Pharma GmbH
• Defendants:
Sandoz Inc.
The
complaints in these cases are substantially identical. Infringement of U.S. Patent Nos. 6,858,650 ("Stable
Salts of Novel Derivatives of 3,3-Diphenylpropylamines," issued February
22, 2005), 7,384,980 ("Derivatives of 3,3-Diphenylpropylamines,"
issued June 10, 2008), 7,855,230 (same title, issued December 21, 2010),
7,985,772 (same title, issued July 26, 2011), and 8,338,478 (same title, issued
December 25, 2012) following a Paragraph IV certification as part of Alkem's
filing of an ANDA to manufacture a generic version of Pfizer's Toviaz®
(fesoterodine fumarate, used to treat the symptoms of overactive bladder). View the Delaware Alkem complaint here.
Par
Pharmaceutical Inc. et al. v Breckenridge Pharmaceutical Inc.
1:13-cv-01114;
filed June 21, 2013 in the District Court of Delaware
• Plaintiffs:
Par Pharmaceutical Inc.; Alkermes Pharma Ireland Ltd.
• Defendant:
Breckenridge Pharmaceutical Inc.
Infringement
of U.S. Patent Nos. 6,592,903 ("Nanoparticulate Dispersions Comprising a
Synergistic Combination
of a Polymeric Surface Stabilizer and Dioctyl Sodium Sulfosuccinate,"
issued July 15, 2003) and 7,101,576 ("Nanoparticulate Megestrol
Formulations," issued September 5, 2006) following a Paragraph IV
certification as part of Breckenridge's filing of an ANDA to manufacture a
generic version of Par's Megace ES® (megestrol acetate, used for the treatment
of appetite loss, severe malnutrition, or unexplained, significant weight loss
in AIDS patients). View the complaint here.
Endo
Pharmaceuticals Inc. v. Ranbaxy Laboratories Ltd. et al.
1:13-cv-04343;
filed June 21, 2013 in the Southern District of New York
• Plaintiff:
Endo Pharmaceuticals Inc.
• Defendants:
Ranbaxy Laboratories Ltd.; Ranbaxy Inc.; Ranbaxy Pharmaceuticals Inc.
Infringement
of U.S. Patent Nos. 7,851,482 ("Method for Making Analgesics," issued
December 14, 2010), 8,309,122 ("Oxymorphone Controlled Release
Formulations," issued November 13, 2012), and 8,329,216 (same title,
issued December 11, 2012) following a Paragraph IV certification as part of
Ranbaxy's filing of an ANDA to manufacture a generic version of Endo's Opana®
ER CRF (oxymorphone hydrochloride, crush-resistant formulation, used to treat
moderate to severe pain in patients requiring continuous, around-the-clock
opioid treatment for an extended period of time). View the complaint here.
Kremers Urban
Pharmaceuticals, Inc. v. Rea et al.
3:13-cv-00399;
filed June 21, 2013 in the Eastern District of Virginia
• Plaintiff:
Kremers Urban Pharmaceuticals, Inc.
• Defendants:
Teresa Stanek Rea; United States Patent and Trademark Office
Reversal of
the revival by the U.S. Patent and Trademark Office of the application that
granted as U.S. Patent No. 6,469,035 ("Methods of Pretreating
Hyperlipidemic Individuals with a Flush Inhibiting Agent Prior to the Start of
Single Daily Dose Nicotinic Acid Therapy to Reduce Flushing Provoked by
Nicotinic Acid," issued October 22, 2002), listed in the Orange Book for AbbVie's
Niaspan® (niacin extended-release tablets, used to treat hypercholesterolemia),
and therefore a declaration that the patent is invalid. View the complaint here.
 On June 13, the Genetic
On June 13, the Genetic The Genetic Alliance, which
The Genetic Alliance, which According to the Genetic
According to the Genetic