By Donald Zuhn —
 On March 4, the U.S. Patent and Trademark Office issued a guidance memorandum, entitled "Guidance For Determining Subject Matter Eligibility Of Claims Reciting Or Involving Laws of Nature, Natural Phenomena, & Natural Products" (or "Myriad-Mayo Guidance"), to implement a new procedure for determining the subject matter eligibility of claims under 35 U.S.C. § 101 in view of the Supreme Court's decisions in Association for Molecular Pathology v. Myriad Genetics, Inc. (2013), and Mayo Collaborative Services v. Prometheus Laboratories, Inc. (2012). At a public forum hosted at the USPTO in May, the Office encouraged shareholders to submit written comments on the Guidance. Since the forum, the Office has encouraged the patent community to provide further feedback regarding the Guidance (despite the Office's announced deadline of July 31 for submitting comments).
On March 4, the U.S. Patent and Trademark Office issued a guidance memorandum, entitled "Guidance For Determining Subject Matter Eligibility Of Claims Reciting Or Involving Laws of Nature, Natural Phenomena, & Natural Products" (or "Myriad-Mayo Guidance"), to implement a new procedure for determining the subject matter eligibility of claims under 35 U.S.C. § 101 in view of the Supreme Court's decisions in Association for Molecular Pathology v. Myriad Genetics, Inc. (2013), and Mayo Collaborative Services v. Prometheus Laboratories, Inc. (2012). At a public forum hosted at the USPTO in May, the Office encouraged shareholders to submit written comments on the Guidance. Since the forum, the Office has encouraged the patent community to provide further feedback regarding the Guidance (despite the Office's announced deadline of July 31 for submitting comments).
Comments regarding the Guidance, including five received after the July 31 deadline, have been posted on the USPTO website. The comments have been divided into seven groups (with the number of submissions in each group also provided): Intellectual property organizations and other associations (21), government agencies (0), academic and research institutions (4), law firms (7), companies (8), individuals (43), and comments received after August 1, 2014 (5). Today, while applicants and practitioners await promised revisions to the Guidance (now more than a month overdue based on initial estimates from Office representatives), we present the first part of an examination of the comments submitted on August 6, 2014 by The Coalition for 21st Century Medicine (note: the Coalition also was a signatory on joint comments submitted on October 14, 2014 by the Biotechnology Industry Organization, five companies, and six individuals (see "Examination of Myriad-Mayo Guidance Comments — BIO Joint Comment")).
According to the Coalition's letter, the group represents more than two dozen diagnostic technology companies, clinical laboratories, researchers, physicians, venture capitalists, and patient advocacy groups (a list of Coalition members can be found on the group's website). The Coalition's 45-page letter provides proposed examination processes for analyzing composition claims and method claims (pages 3-12) and more than 30 pages of examples providing 41 exemplary claims and an analysis of each claim under the Coalition's proposed "practical framework." The Coalition divides its examples into four categories (with the number of claims in each category also provided):
A. Composition/Manufacture Claim Reciting A Natural Product (4 claims)
B. Composition vs. Method Claims, Each Reciting A Small Organic Molecule (11 claims)
C. Composition vs. Method Claims, Each Reciting Two Natural Products (12 claims)
D. Process Claim Involving A Natural Principle (14 claims)
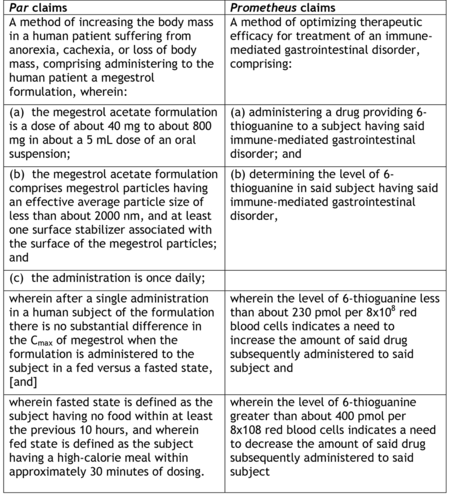
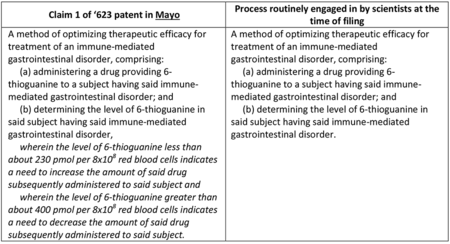
By comparison, the Office's Myriad-Mayo Guidance ran 19 pages and provided eight examples with only fourteen exemplary claims (see "USPTO Issues Guidance for Analyzing Subject Matter Eligibility of Claims Reciting Laws of Nature/Natural Principles, Natural Phenomena or Natural Products").
The Coalition's letter begins by asserting that its "proposed practical framework" and analysis in each of the four examples "are supported by binding case law precedent from the Supreme Court and the Federal Circuit," while acknowledging "the Office's challenge in creating a practical framework for everyday examination that harmonizes decades of court decisions under changing statutory provisions, especially when such court decisions appear to have intentionally avoided any clear-cut analytical rules." That being said, the Coalition contends that:
The Courts have made clear that the default under § 101 is eligibility. Only if subject matter clearly fits within a category of excluded matter is it ineligible for patenting. And those categories should be interpreted faithfully but narrowly to exclude only that which is clearly a product, law or phenomenon of nature or an abstract idea.
The Coalition stresses that "[t]he importance of the Office correctly instructing its examiners on how to analyze subject matter eligibility cannot be overstated," stating that:
The United States is currently a global leader in biotechnology, emerging technologies, and medicine. Restricting the scope of patent-eligibility in the U.S. beyond that required by binding case law may cause a slowing of the growth of human capital, investment, and even academic advancement in those areas, especially as compared to other markets such as China, Europe, Australia, Japan, Russia, and South Korea that continue to offer a superior degree of protection for inventions in these industries.
The Coalition next outlines proposed processes for analyzing composition claims and method claims. With respect to composition claims, the letter suggests that the following questions be addressed in examining composition claims for subject matter eligibility:
1. Does the claim appear, on its face, to potentially encompass a product of nature?
2. What is the product of nature potentially encompassed by the claim?
3. Is the claimed composition structurally identical to a natural product, including any discrete natural unit?
4. Does the claimed composition possess a new or enhanced function or utility as compared to the natural product?
According to the Coalition, "[t]he overarching question that should guide examination for subject matter eligibility under 35 U.S.C. § 101 is 'Who/what produced the composition as claimed?'" Citing Diamond v. Chakrabarty, the letter states that:
"Congress thus recognized that the relevant distinction was […] between products of nature […] and human-made inventions." Chakrabarty, 447 U.S. at 313. That which is made by nature unaided by the hand of man is "part of the storehouse of knowledge of all men" and ineligible for patenting. "Anything under the sun that is made by man" is eligible for patenting.
In addressing the first question of its proposed process for analyzing composition claims, the Coalition, sounding somewhat prescient, argues that "[e]xamination must recognize the critical distinction between 'encompassing' and 'comprising,'" adding that "[a] claim 'comprising' a product of nature could well be eligible since everything at its core 'comprises' a product of nature." The Office has recently indicated that the revised Guidance would focus on claims that are directed to a judicial exception rather than those that merely recite or involve a judicial exception (see "USPTO Provides Update on Status of Revised Myriad-Mayo Guidance").
According to the Coalition, the next step in addressing the first question of its proposed analysis involves looking for a potential natural counterpart in the specification or common knowledge in the art. The letter contends, however, that "[a] claim to anything whose chemical structure as a whole is facially not found or not reasonably suspected of being in nature does not raise the question of subject matter eligibility."
Moving on to the second question, the letter declares that "[t]his is arguably the most important part of the analysis," adding that "[a] careful and correct identification and definition of what the product of nature is (and is not) will generally determine the outcome." Following a discussion of the difference between a molecule's chemical and physical properties and its specific biological functions, the letter states that:
A claim does not encompass a natural product simply because the claimed composition shares some or many properties with a natural product. If those properties are put to new, different, or enhanced function or use in the particular composition of the claim, then the claim is to a patent eligible human invention.
The letter next addresses the third question in the analysis of composition claims, noting that "[i]f care has been taken in correctly identifying a natural product against which to compare the claim[ed] composition, then structural identity should be fairly straightforward to assess." While suggesting that relatively few or minor structural differences may confer subject matter eligibility, the letter acknowledges that:
Not just any change will be sufficient to make a composition patent eligible. For example, the Court in AMP recognized that the claimed isolated DNA was structurally different from native DNA ("broken covalent bonds") but nevertheless determined the claimed compositions were ineligible for patenting. This was because the claims were not defined in terms of any structural change that yielded an identified functional change and were, instead, defined expressly according to the natural properties and functions of the genes.
However, the Coalition's letter explains that:
[A]ny structural change may potentially confer eligibility, no matter how relatively minor, if it imparts some functional change. In Chakrabarty, the structural differences between the claimed bacterium and the natural bacteria were comparatively minor. These changes represented a relatively small insertion of genetic material that produced a relatively miniscule additional complement of proteins. But the important thing for the Court was the impact these small changes had on the function of the bacterium. "Here, by contrast, the patentee has produced a new bacterium with markedly different characteristics from any found in nature and one having the potential for significant utility." Chakrabarty, 447 U.S. at 310 (emphasis added). The real question under this point is: Has the patentee created something (rather than merely finding something in nature) and has the patentee's activity (the "hand of man") given that something new utilities?
The letter lays out three possible scenarios following analysis of composition claims under the third prong of the Coalition's proposed process:
1. The claimed composition as a whole is structurally identical to a natural product as a whole — at which point the claim should be rejected under 35 U.S.C. § 101 for being directed to a natural product.
2. The claimed composition as a whole is not structurally identical to and does not share substantial structural similarity with a natural product as a whole – at which point the composition should be found to be patent eligible.
3. The claimed composition as a whole is not structurally identical to, but shares substantial structural similarity with, a natural product as a whole — at which point examination should proceed to the fourth prong of analysis.
With respect to the fourth question in the Coalition's proposed process of analyzing composition claims, the letter argues that "[e]xamination should focus on determining whether the claimed composition possesses at least one new or enhanced function or utility, either recited in the specification or in the claim language itself, which is not found in the natural product." The letter explains that "[t]he new or enhanced function or utility must be sufficient under traditional § 101 utility analysis (i.e., specific, substantial, credible)," adding that:
If the composition is structurally differentiated from a natural product in only a general, negative way (e.g., merely "isolated") and is claimed according to its natural function, then the composition is patent ineligible. If on the other hand the composition is distinguished from the natural product in a positive, specific way (e.g., a specific structural feature) and is claimed according to that function, then the composition is eligible.
With respect to the "special problem" of a combination of two or more natural products, the letter suggests that "[t]he composition can be patent eligible if (a) the combination is recited at some level of specificity to exclude reasonably foreseeable natural combinations and (b) the combination as claimed possesses at least one function not found in any of the natural components alone or any natural combination of the natural components."
In subsequent posts, we will examine the Coalition's proposed process for analyzing method claims and some of the examples and exemplary claims set forth in the Coalition's letter.
For additional information regarding this topic, please see:
• "BIO IPCC Panel Discusses Impact of Myriad-Mayo Guidance," November 13, 2014
• "USPTO Provides Update on Status of Revised Myriad-Mayo Guidance," November 12, 2014
• "Examination of Myriad-Mayo Guidance Comments — University Community Joint Comment," October 28, 2014
• "Examination of Myriad-Mayo Guidance Comments — BIO Joint Comment," October 23, 2014
• "Sherry Knowles Addresses Real World Impact of Myriad-Mayo Guidance at BIO Symposium," October 15, 2014
• "Guest Post: Myriad — A Direct and Unexceptional Approach," October 12, 2014
• "USPTO Outlines Changes to Myriad-Mayo Guidance at BIO Symposium," September 30, 2014
• "USPTO Expected to Issue Revised Myriad-Mayo Guidance in October," September 17, 2014
• "Guest Post: Myriad-Mayo Guidance — Consistency With International Harmonization and TRIPS," August 26, 2014
• "Examination of Myriad-Mayo Guidance Comments — AUTM, COGR, AAU, and APLU," August 21, 2014
• "Examination of Myriad-Mayo Guidance Comments — International Bioindustry Associations," August 11, 2014
• "Examination of Myriad-Mayo Guidance Comments — ACLU," August 5, 2014
• "Guest Post: Overview of First Published Comments on Myriad-Mayo Patent Eligibility Guidance," July 13, 2014
• "Guest Post: USPTO Public Forum on Patent Guidance: My Thoughts as a Speaker and Attendee," June 11, 2014
• "USPTO Holds Forum on Subject Matter Eligibility — Part IV," May 22, 2014
• "USPTO Holds Forum on Subject Matter Eligibility — Part III," May 15, 2014
• "USPTO Holds Forum on Subject Matter Eligibility — Part II," May 14, 2014
• "Guest Post: How to Patent Grapefruit Juice — The New USPTO Guidance for Patent Eligible Subject Matter Is Both Sticky and Sour," May 13, 2014
• "USPTO Holds Forum on Subject Matter Eligibility — Part I," May 12, 2014
• "USPTO Tries to Address Public Misunderstandings Regarding Myriad-Mayo Guidance," April 16, 2014
• "USPTO Issues Guidance for Analyzing Subject Matter Eligibility of Claims Reciting Laws of Nature/Natural Principles, Natural Phenomena or Natural Products," March 4, 2014
 Strafford will be offering a webinar/teleconference entitled "Evolving PTAB Trial Practice: Navigating Complex Procedural Rules — Strategically Using Routine and Additional Discovery, Requests for Joinder, and Motions to Amend" on December 15, 2014 from 1:00 to 2:30 pm (EST). Greg H. Gardella and Scott A. McKeown of Oblon Spivak McClelland Maier & Neustadt will provide guidance to counsel on Patent Trial and Appeal Board (PTAB) trial practice, focusing on discovery, joinder and motions to amend, and examine guidance issued by the PTAB, as well as lessons from recent PTAB decisions. The webinar will review the following questions:
Strafford will be offering a webinar/teleconference entitled "Evolving PTAB Trial Practice: Navigating Complex Procedural Rules — Strategically Using Routine and Additional Discovery, Requests for Joinder, and Motions to Amend" on December 15, 2014 from 1:00 to 2:30 pm (EST). Greg H. Gardella and Scott A. McKeown of Oblon Spivak McClelland Maier & Neustadt will provide guidance to counsel on Patent Trial and Appeal Board (PTAB) trial practice, focusing on discovery, joinder and motions to amend, and examine guidance issued by the PTAB, as well as lessons from recent PTAB decisions. The webinar will review the following questions: