By Sherri Oslick —
 About Court Report: Each week we will report briefly on recently filed biotech and pharma cases.
About Court Report: Each week we will report briefly on recently filed biotech and pharma cases.
Pfizer Inc. et al. v. Mylan Inc. et al.
1:15-cv-00026; filed January 8, 2015 in the District Court of Delaware
• Plaintiffs: Pfizer Inc.; Wyeth LLC; Pfizer Pharmaceuticals LLC; PF Prism C.V.; Pfizer Manufacturing Holdings LLC
• Defendants: Mylan Inc.; Agila Specialties Private Ltd.; Mylan Pharmaceuticals Inc.
Infringement of U.S. Patent Nos. 7,879,828 ("Tigecycline Compositions and Methods of Preparation," issued February 1, 2011) and 8,372,995 ("Crystalline Solid Forms of Trigecycline and Methods of Preparing Same," issued February 12, 2013), licensed to Pfizer, following a Paragraph IV certification as part of Mylan's filing of an ANDA to manufacture a generic version of Pfizer's Tygacil® (tigecycline injectible, used for the treatment of complicated skin and skin structure infections, complicated intra-abdominal infections, and community-acquired bacterial pneumonia). View the complaint here.
Sanofi-Aventis U.S. LLC et al. v. Accord Healthcare, Inc.
1:15-cv-00018; filed January 7, 2015 in the Middle District of North Carolina
• Plaintiffs: Sanofi-Aventis U.S. LLC; Aventis Pharma S.A.; Sanofi
• Defendant: Accord Healthcare, Inc.
Sanofi-Aventis U.S. LLC et al. v. Accord Healthcare, Inc.
3:14-cv-08079; filed December 29, 2014 in the District Court of New Jersey
• Plaintiffs: Sanofi-Aventis U.S. LLC; Aventis Pharma S.A.; Sanofi
• Defendant: Accord Healthcare, Inc.
The complaints in these cases are substantially identical. Infringement of U.S. Patent No. 5,847,170 ("Taxoids, Their Preparation and Pharmaceutical Compositions Containing Them" issued December 8, 1998) following a Paragraph IV certification as part of Accord's filing of an ANDA (for one dosage form) and an NDA (under § 505(b)(2) of the Food, Drug and Cosmetic Act) (for another dosage form) to manufacture a generic version of Sanofi's Jevtana® (cabazitaxel injection, used in combination with prednisone for the treatment of patients with hormonerefractory metastatic prostate cancer previously treated with a docetaxel-containing treatment regimen). View the New Jersey complaint here.
Forest Laboratories LLC et al. v. Apotex Corp. et al.
1:15-cv-00018; filed January 5, 2015 in the District Court of Delaware
• Plaintiffs: Forest Laboratories LLC; Forest Laboratories Holdings Ltd.; Cerexa Inc.; Takeda Pharmaceutical Company Ltd.
• Defendants: Apotex Corp.; Apotex Inc.; Sandoz Inc.
Infringement of U.S. Patent Nos. 6,417,175 ("Phosphonocephem Derivatives," issued July 9, 2002), 6,906,055 ("Phosphonocephem Compound," issued June 14, 2005), 7,419,973 (same title, issued September 2, 2008), and 8,247,400 ("Cephem Compounds Useful for the Treatment of Bacterial Infections," issued August 21, 2012) following a Paragraph IV certification as part of Apotex's filing of an ANDA to manufacture a generic version of Forest's Teflaro® (ceftaroline fosamil injection, used for the treatment of community-acquired bacterial pneumonia and for the treatment of acute bacterial skin and skin structure infections). View the complaint here.
Reckitt Benckiser Pharmaceuticals Inc. et al. v. Par Pharmaceutical Inc. et al.
1:14-cv-01573; filed December 31, 2014 in the District Court of Delaware
• Plaintiffs: Reckitt Benckiser Pharmaceuticals Inc.; MonoSol Rx LLC
• Defendants: Par Pharmaceutical Inc.; IntelGenX Technologies Corp.
Reckitt Benckiser Pharmaceuticals Inc. et al. v. Watson Laboratories Inc.
1:14-cv-01574; filed December 31, 2014 in the District Court of Delaware
• Plaintiffs: Reckitt Benckiser Pharmaceuticals Inc.; MonoSol Rx LLC
• Defendant: Watson Laboratories Inc.
The complaints in these cases are substantially identical. Infringement of U.S. Patent Nos. 8,900,497 ("Process for Making a Film Having a Substantially Uniform Distribution of Components," issued December 2, 2014) and 8,906,277 ("Process for Manufacturing a Resulting Pharmaceutical Film," issued December 9, 2014) in conjunction with defendants' filing of an ANDA to manufacture a generic version of Reckitt Benckiser's Suboxone® (buprenorphine hydrochloride and naloxone hydrochloride sublingual film, used for the maintenance treatment of opioid dependence). View the Par complaint here.
Sanofi-Aventis U.S. LLC et al. v. Fresenius Kabi USA LLC
1:14-cv-01533; filed December 30, 2014 in the District Court of Delaware
• Plaintiffs: Sanofi-Aventis U.S. LLC; Aventis Pharma S.A.; Sanofi
• Defendant: Fresenius Kabi USA, LLC
Sanofi-Aventis U.S. LLC et al. v. Fresenius Kabi USA, LLC
3:14-cv-08082; filed December 29, 2014 in the District Court of New Jersey
• Plaintiffs: Sanofi-Aventis U.S. LLC; Aventis Pharma S.A.; Sanofi
• Defendant: Fresenius Kabi USA, LLC
The complaints in these cases are substantially identical. Infringement of U.S. Patent Nos. 5,847,170 ("Taxoids, Their Preparation and Pharmaceutical Compositions Containing Them" issued December 8, 1998) and 7,241,907 ("Acetone Solvate of Dimethoxy Docetaxel and its Process of Preparation" issued July 10, 2007) following a Paragraph IV certification as part of Fesenius' filing of an ANDA to manufacture a generic version of Sanofi's Jevtana® (cabazitaxel injection, used in combination with prednisone for the treatment of patients with hormonerefractory metastatic prostate cancer previously treated with a docetaxel-containing treatment regimen). View the New Jersey complaint here.
Sanofi-Aventis U.S. LLC et al. v. BPI Labs, LLC et al.
8:14-cv-03233; filed December 30, 2014 in the Middle District of Florida
• Plaintiffs: Sanofi-Aventis U.S. LLC; Aventis Pharma S.A.; Sanofi
• Defendants: BPI Labs, LLC; Belcher Pharmaceuticals, LLC
Sanofi-Aventis U.S. LLC et al. v. BPI Labs, LLC et al.
3:14-cv-08081; filed December 29, 2014 in the District Court of New Jersey
• Plaintiffs: Sanofi-Aventis U.S. LLC; Aventis Pharma S.A.; Sanofi
• Defendants: BPI Labs, LLC; Belcher Pharmaceuticals, LLC
The complaints in these cases are substantially identical. Infringement of U.S. Patent No. 5,847,170 ("Taxoids, Their Preparation and Pharmaceutical Compositions Containing Them" issued December 8, 1998) following a Paragraph IV certification as part of BPI's filing of an ANDA to manufacture a generic version of Sanofi's Jevtana® (cabazitaxel injection, used in combination with prednisone for the treatment of patients with hormonerefractory metastatic prostate cancer previously treated with a docetaxel-containing treatment regimen). View the New Jersey complaint here.
Novartis Pharmaceuticals Corp. et al. v. Ranbaxy Inc. et al.
1:14-cv-01526; filed December 29, 2014 in the District Court of Delaware
• Plaintiffs: Novartis Pharmaceuticals Corp.; Novartis AG
• Defendants: Ranbaxy Inc.; Ranbaxy Laboratories Ltd.
Infringement of U.S. Patent Nos. 6,894,051 ("Crystal Modification of a N-phenyl-2-pyrimidineamine Derivative, Processes for Its Manufacture and Its Use," issued May 17, 2005) and RE43,932 ("Crystal Modification of a N-phenyl-2-pyrimidineamine Derivative, Processes for Its Manufacture and Its Use," issued January 15, 2013) following a Paragraph IV certification as part of Ranbaxy's filing of an ANDA to manufacture a generic version of Novartis' Gleevec® (imatinib mesylate, used for various indications, including treatment of myeloid leukemia). View the complaint here.
AstraZeneca AB et al. v. Andrx Labs, LLC et al.
3:14-cv-08030; filed December 24, 2014 in the District Court of New Jersey
• Plaintiffs: AstraZeneca AB; Aktiebolaget Hassle; AstraZeneca LP; Zeneca Inc.
• Defendants: Andrx Labs, LLC; Andrx Corp.; Actavis, Inc.
Infringement of U.S. Patent Nos. 6,369,085 ("Form of S-omeprazole," issued April 9, 2002) and 7,411,070 (same title, issued August 12, 2008) following a Paragraph IV certification as part of Andrx's filing of an ANDA to manufacture a generic version of AstraZeneca's Nexium 24HR® (esomeprazole magnesium, used for the treatment of gastroesophageal reflux disease). View the complaint here.
Otsuka Pharmaceutical Co., Ltd. v. Apotex Corp. et al.
1:14-cv-08074; filed December 24, 2014 in the District Court of New Jersey
• Plaintiff: Otsuka Pharmaceutical Co., Ltd.
• Defendants: Apotex Corp.; Apotex Inc.; Apotex Pharmachem Inc.
Infringement of U.S. Patent Nos. 8,017,615 ("Low Hygroscopic Aripiprazole Drug Substance and Process for the Preparation Thereof," issued September 13, 2011), 8,580,796 (same title, issued November 12, 2013), 8,642,760 (same title, issued February 4, 2014), and 8,759,350 ("Carbostyril Derivatives and Serotonin Reuptake Inhibitors for Treatment of Mood Disorders," issued June 24, 2014) following a Paragraph IV certification as part of Apotex's filing of an ANDA to manufacture a generic version of Otsuka's Abilify® (aripiprazole, used to treat bipolar disorder and schizophrenia). View the complaint here.
Otsuka Pharmaceutical Co., Ltd. v. Sciegen Pharmaceuticals Inc. et al.
1:14-cv-08077; filed December 22, 2014 in the District Court of Delaware
• Plaintiff: Otsuka Pharmaceutical Co., Ltd.
• Defendants: Sciegen Pharmaceuticals Inc.; Bactolac Pharmaceutical, Inc.
Infringement of U.S. Patent Nos. 8,017,615 ("Low Hygroscopic Aripiprazole Drug Substance and Process for the Preparation Thereof," issued September 13, 2011), 8,580,796 (same title, issued November 12, 2013), and 8,642,760 (same title, issued February 4, 2014) following a Paragraph IV certification as part of Sciegen's filing of an ANDA to manufacture a generic version of Otsuka's Abilify® (aripiprazole, used to treat bipolar disorder and schizophrenia). View the complaint here.
 If innovation drives progress, then 2014 was the year of progress! During the past year the U.S. Food and Drug Administration approved 41 new drugs. That is 14 more than 2013 and the highest number in 18 years! What spurred this increase? And what challenges do these new drugs face in the 2015 market?
If innovation drives progress, then 2014 was the year of progress! During the past year the U.S. Food and Drug Administration approved 41 new drugs. That is 14 more than 2013 and the highest number in 18 years! What spurred this increase? And what challenges do these new drugs face in the 2015 market?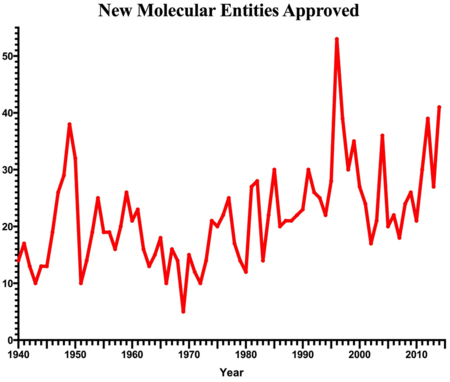 Chart of New Molecular Entities approved: Annual approved NMEs based on data from [1] and [3].
Chart of New Molecular Entities approved: Annual approved NMEs based on data from [1] and [3].







