This is the second article in a series on advancements in microbiome research and development. This installment reviews US patents issued in 2015 in "Microbiomics." Upcoming articles will continue to review important topics in this technology area, and patents of interest.
By Jessica Miles* and Anthony D. Sabatelli** —
Microbiome-related therapies typically involve compositions containing bacteria — often called probiotics — that treat disease or promote health. Some therapies change the composition of the gut microbiome by providing desirable bacterial species, nutrients that promote the growth of desirable microbiome members, or bacterial species that displace bacterial pathogens. Other therapies comprise bacteria or bacterial components that interact with the patient's own organs, tissues, and systems. Many of these therapies stimulate the immune system, and therapies are being developed to treat various metabolic, inflammatory, and infectious diseases.
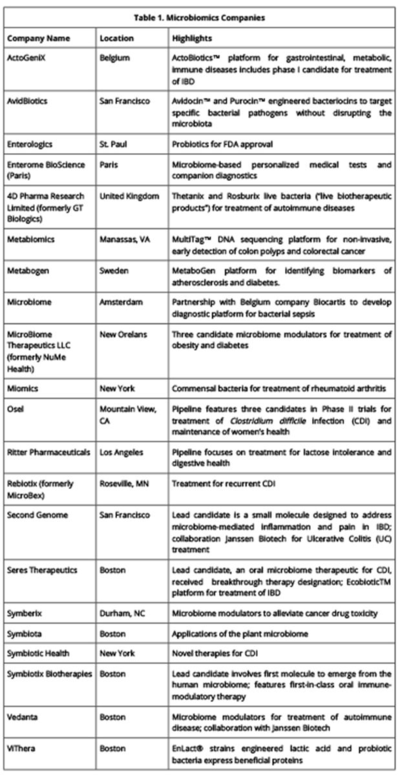
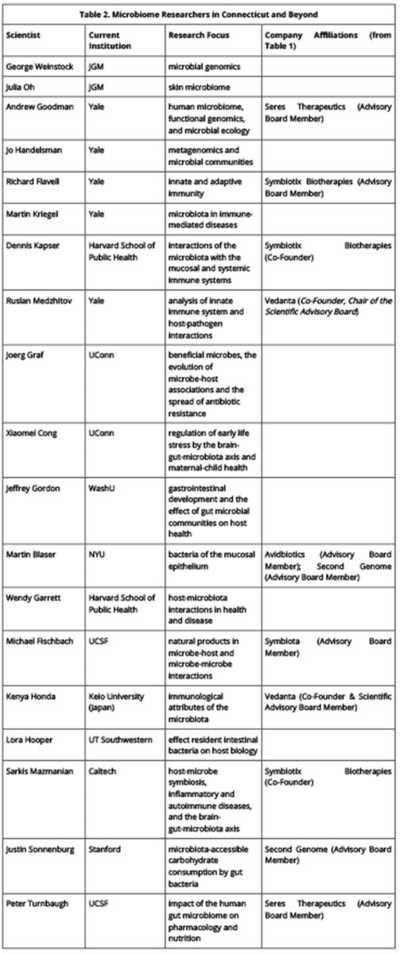
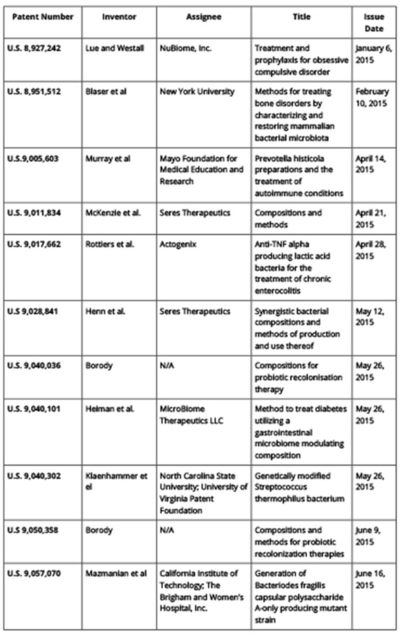
There has been continuous patent activity in this field in 2015. Each issued U.S. patent is listed in Table 1 (click on table to enlarge) and summarized in more detail below. Notably, these patents relate to a variety of research areas within microbiomics. Patent No. 8,927,242 relates to the "gut-brain axis," an emerging area of research that will be discussed more fully in a next installment. The term describes the connection between the gastrointestinal tract and the brain, and research in this area seeks to identify how microbiota influence brain function. Patent No. 8,951,512 relates to the relationship between the composition of the microbiome and diseases such as obesity and related metabolic syndromes. The patent covers therapies for osteoporosis and other disorders of bone formation and mineralization. Patent No. 9,005,603 describes treatments for autoimmune disease.
Other patents focus on therapies for gastrointestinal disorders. Patent 9,011,834 describes the use of bacterial spores for modulating the composition of the gut microbiota. Patent 9,017,662 relates to a therapeutic, engineered strain for the treatment inflammatory bowel disease. Patent 9,028,841 describes a method for treating Clostridium difficle infection. Patent 9,040,036 relates to a probiotic combination of bacterial spores and non-pathogenic bacteria. Patent 9,040,101 describes a method for treating metformin-induced diarrhea while improving insulin sensitivity in diabetic patients. Patent 9,040,302 relates to the use of an engineered bacterial strain for targeted delivery of therapies to the gastrointestinal tract. Patent 9,050,358 describes a probiotic combination of bacterial species for the treatment of irritable bowel syndrome and gastrointestinal distress. Patent 9,057,070 relates to the use of an engineered strain for treatment of inflammatory bowel disease.
There are both public and private assignees represented among these newly-granted patents. New York University, North Carolina State University, the University of Virginia Patent Foundation, and CalTech are universities and university affiliates. A non-profit medical practice and research center, the Mayo Clinic, and the Brigham and Women's Hospital, Inc., a teaching and research hospital, were also awarded patents. Several of the companies profiled in the first installment of this series as leaders in microbiome R&D continue to develop their portfolios with these recent patents: NuBiome, Inc., Actogenix, Seres Therapeutics, and MicroBiome Therapeutics LLC were all granted patents this year. There are also two patents with the same sole inventor, Thomas Borody. Dr. Borody is the founder and director of the Sydney-based Centre for Digestive Diseases, which researches, diagnoses, and treats gastrointestinal disorders.
As this review of patent activity indicates, microbiome research and development is thriving. The patents discussed here cover a wide range of applications of the microbiome, targeting gastrointestinal, autoimmune, bone, and psychiatric disorders. Moreover, the patents come from a variety of institutions, including academia, industry, and non-profit research centers. The diversity of subject matter and origin of these inventions speaks to the strength and importance of microbiome R&D, and it is likely that the remainder of 2015 will see more innovation in this field.
Table 1: 2015 Patents in Microbiomics
U.S. Patent No. 8,927,242 generally relates to a bacterial therapy for treatment of a subset of psychiatric disorders in children. The patent covers administration of the bacteria Streptococcus oralis 89a to treat mental illness. Pediatric Autoimmune Neuropsychiatric Disorders Associated with Streptococcal Infections (PANDAS) include Obsessive Compulsive Disorder (OCD) and/or Tourette Syndrome, whose symptoms can worsen following infection. Autoimmunity from inappropriate immune responses to bacterial antigens is thought to impair function of the basal ganglia, which controls the brain structure involved in emotion and movement. Embodiments of this invention use a combination of therapeutic bacteria, which reduces populations of the antigen-inducing species, with supplemented enzymes (such as PepO, PepO2 and subtilisin), which break down the antigens that cause disease.
U.S. Patent No. 8,951,512 generallyrelates to a bacterial therapy for treatment of osteoporosis or related bone disorders. The gut microbiota modulates metabolism and immunity. Changes to the composition of the gut microbiota can disrupt these critical functions and lead to changes in bone mass. The patent claims bacterial strains, including Helicobacter pylori, which restore gut microbiome composition; prebiotics that stimulate the growth of therapeutic bacterial strains or improve gut microbiome composition; and methods for administering these compositions to patients.
U.S. Patent No. 9,005,603 generally relates to a bacterial therapy containing Prevotella histicola for treatment of autoimmune diseases such as arthritis and multiple sclerosis. This species reduces inflammation, a hallmark of autoimmune disease.
U.S. Patent 9,011,834 generally relates to a bacterial therapy for treatment or prevention changes to the composition of the gut microbiota, such as those that accompany Clostridium difficile infection, irritable bowel syndrome, and Crohn's. The patent claims the production, isolation, and administration of germination-competent bacterial spores. These spores are intended to modulate the gut microbiota.
U.S. Patent 9,017,662 generally relates to a bacterial therapy for treatment of inflammatory bowel disease (IBD). The patent claims oral administration of a strain of Lactococcus lactis engineered to produce antibodies that neutralize tumor necrosis factor-α (TNF).
U.S. Patent 9,028,841 generally relates to a bacterial therapy for treatment or prevention of changes to the composition of the gut microbiota, such as those that accompany C. difficile infection. The patent claims a composition containing a combination of Collinsella aerofaciens and Clostridiaceae family members. These bacteria are intended to inhibit the growth of pathogenic species and populate the gut.
U.S. Patent 9,040,036 generally relates to a bacterial therapy for treatment or prevention of changes to the composition of the gut microbiota. The patent claims a composition containing a combination of bacterial spores from anaerobic cocci, non-pathogenic Clostridiaceae family members, members of the genus Bacteroides, Collinsella, Fusobacteria, Propionibacteria, Lactobacillus, Gemmiger, Desulfomonas, Peptostreptococcus; and a non-pathogenic Escherichia coli species.
U.S. Patent 9,040,101 generally relates to the treatment of diarrhea in diabetic patients taking metformin, a treatment for type II diabetes. Metformin treatment causes diarrhea in some patients and has been shown to change the composition of the gut microbiome in mice. The patent claims a treatment containing inulin, beta-glucan, and polyphenolic compounds from blueberries. Inulin is an undigestible carbohydrate that can be utilized by Bacteriodetes species, promoting their growth and function. Beta-glucan is intended not only to protect members of the gut microbiome from the antimicrobial action of bile, but also to provide polysaccharide-derived nutrients for the members of the gut microbiota.Polyphenolic compounds have been shown to improve insulin sensitivity in diabetic patients.
U.S. Patent 9,040,302 generally relates to the use of an engineered strain of Streptococcus thermophiles as a drug delivery vehicle. The patent claims a composition containing the engineered strain that expresses an alanine-glutamine or alanine-glutamine-arginine rich bioagent after being exposed to bile in the intestine. Glutamine helps promote intestinal integrity. The addition of alanine is intended to improve stability of the peptide, while arginine may increase glutamine's effects.
U.S. Patent 9,050,358 generally relates toa bacterial therapy for treatment of gastrointestinal disorders associated with changes to the composition of the gut microbiome. The patent claims a composition containing bacterial spores from Clostridium bifermentans, Clostridium butyricum, Clostridium difficile, Clostridium ramosum, Clostridium innocuum and a Collinsella species. This composition can also be combined with a number of other Clostridia and Bacteroides, members of the genus, Bifidobacterium Pseudoflavonifractor capillosus, Odoribacter splanchnicus, anaerobic cocci, Fusobacteria, Propionibacteria, Lactobacillus, Gemmiger, Desulfomonas, Peptostreptococcus, Escherichia coli, Peptostreptococcus productus II, Parabacteroides distasonis Fusobacterium prausnitzii, Coprococcus eutactus, , Peptostreptococcus productus I, Ruminococcus bromii, Eubacterium siraeum, Ruminococcus torques, Eubacterium rectale, Eubacterium eligens, Eubacterium biforme, Eubacterium rectale III-F, Coprococcus comes, Ruminococcus albus, Dorea formicigenerans, Eubacterium haffii, Eubacterium ventriosum I, Ruminococcus obeum, Eubacterium rectale, , Ruminococcus callidus, Butyrivibrio crossotus, Acidaminococcus fermentans, Eubacterium ventriosum, Coprococcus catus, Aerostipes hadrus, Eubacterium cylindroides, Eubacterium ruminantium, Eubacterium CH-1, Staphylococcus epidermidis, Peptostreptococcus BL, Eubacterium limosum, Tissirella praeacuta, Fusobacterium mortiferum I, Fusobacterium naviforme, Ruminococcus flavefaciens, Ruminococcus AT, Peptococcus AU-1, Fusobacterium nucleatum, Fusobacterium mortiferum, Gemella morbillorum, Finegoldia magnus, Peptococcus G, -AU-2; Streptococcus intermedius, Ruminococcus lactaris, Ruminococcus CO Gemmiger X, Coprococcus BH, –CC; Eubacterium tenue, Eubacterium ramulus, , Prevotella oralis, Prevotella ruminicola, Odoribacter splanchnicus, Desuifomonas pigra, Succinivibrio A, and a non-pathogenic fungi.
U.S. Patent 9,057,070 generally relates to a bacterial therapy for treatment of inflammatory bowel disease. The patent claims administration of a strain of Bacillus fragilis engineered to lack all but one of its native capsular polysaccharides (capsular polysaccharide A, or PSA). The inventors have shown that PSA reduces intestinal inflammation in mouse models of multiple sclerosis and inflammatory bowel disease. This invention is intended to facilitate the purification of PSA, but not other polysaccharides, from B. fragilis.
* Jessica Miles is a Technology Specialist at Dilworth IP
** Dr. Sabatelli is a Partner with Dilworth IP
 Strafford will be offering a webinar/teleconference entitled "Trade Secrets and Cybersecurity: Protecting Intellectual Property, Mitigating Loss and Navigating Legal Responses" on September 24, 2015 from 1:00 to 2:30 pm (EDT). Matthew F. Prewitt of Schiff Hardin will provide guidance to IP counsel on protecting trade secrets from cyberattack, discuss what action to take if trade secrets are stolen in a cyberattack and to mitigate the loss, and examine the legal responses available to respond to a cyberattack. The webinar will review the following questions:
Strafford will be offering a webinar/teleconference entitled "Trade Secrets and Cybersecurity: Protecting Intellectual Property, Mitigating Loss and Navigating Legal Responses" on September 24, 2015 from 1:00 to 2:30 pm (EDT). Matthew F. Prewitt of Schiff Hardin will provide guidance to IP counsel on protecting trade secrets from cyberattack, discuss what action to take if trade secrets are stolen in a cyberattack and to mitigate the loss, and examine the legal responses available to respond to a cyberattack. The webinar will review the following questions: