By Andrew Williams —
 Do you want the good news or the bad news first? Well, the good news is that the Federal Circuit has begun reversing PTAB decisions on the merits for IPR proceedings. To be fair, in the Microsoft case, the Federal Circuit remanded because the claim construction was not reasonable — and therefore could not be the broadest reasonable interpretation. But on November 5, 2015, in Belden Inc. v. Berk-Tek LLC, the appeals court reversed the trend of affirming every Board decision related to the prior art and reversed a PTAB's obviousness determination.
Do you want the good news or the bad news first? Well, the good news is that the Federal Circuit has begun reversing PTAB decisions on the merits for IPR proceedings. To be fair, in the Microsoft case, the Federal Circuit remanded because the claim construction was not reasonable — and therefore could not be the broadest reasonable interpretation. But on November 5, 2015, in Belden Inc. v. Berk-Tek LLC, the appeals court reversed the trend of affirming every Board decision related to the prior art and reversed a PTAB's obviousness determination.
That brings us to the bad news, at least for Patent Owners. The Federal Circuit held as obvious two claims that had been affirmed by the PTAB as patentable. In other words, despite having the reputation of being a patent "death squad," the Board was told by the Federal Circuit that it was not invalidating enough claims (at least in this case). Moreover, the Federal Circuit sanctioned the introduction of new affidavit testimony by the Petitioner after the Patent Owner had submitted its last response and expert testimony. The Court did explain that the Board did not err in determining that the new evidence was limited to responding to assertions of the Patent Owner's expert. Nevertheless, in view of the fact that no expert testimony accompanied the original petition, this case amounts to an all-around unwelcome decision for Patent Owners.
As this case turns on obviousness, at least a cursory understanding of the technology is important. The patent at issue, Belden's U.S. Patent No. 6,074,503, discloses and claims "a method of making a cable by passing a core and conducting wires through one or more dies, bunching the wires into grooves on the core, twisting the bunch to close the cable, and jacketing the entire assembly." The problems solved by this invention included how to align the conducting wires to the core, and how to prevent the core from twisting independently during the process. Claim 1 reads:
1. A method of producing a cable, comprising steps of:
passing a plurality of transmission media and a core through a first die which aligns the plurality of transmission media with surface features of the core and prevents twisting motion of the core;
bunching the aligned plurality of transmission media and core using a second die which forces each of the plurality of transmission media into contact with the surface features of the core which maintain a spatial relationship between each of the plurality of transmission media;
twisting the bunched plurality of transmission media and core to close the cable; and
jacketing the closed cable.
Fig. 4 from the patent shows an assembly containing three dies (the two squares and the cone-like structure), and Figure 2 represents a cross-section of the resulting cable.
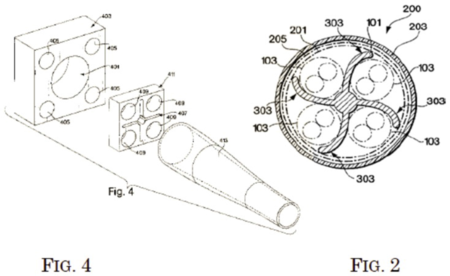
Claim 4 was grouped with claim 1 because it only contained the additional limitation that the core be "extruded," such that the surface features of the core align with the plurality of transmission media. Claims 2 and 3 were grouped together because they both required the third die (represented as the biggest square in Fig. 4 above). Finally, claims 5 and 6 were grouped because they both required that the transmission media be "twisted pairs of insulated conductors."
Obviousness
The Board instituted trial after Berk-Tek's predecessor in interest (Nexans, Inc.) filed a petition asserting anticipation and obviousness based on seven prior art references. For the four original claims (claims 1-4), the Patent Office sided with the Petitioner and invalidated the claims as obvious. For the remaining 2 claims, however, the Board concluded that, even though all of the elements were known in the art, the petitioner had not established that there was a reason to combine them.
On appeal, the Federal Circuit found that there was substantial evidence for the Board's conclusion that claims 1-4 were obvious. One of the primary references was a Japanese patent (Japanese Patent No. 19910, or JP '910). Belden argued that this reference was only related to making transmission lines, but the Board noted that, among other things, the title of the JP '910 was "A method of manufacturing plastic insulated communication cables." The PTAB also concluded that a particular board in the Japanese reference could have been used to prevent the twisting of the core. Moreover, the Board cited to the expert report of Mr. Baxter, which included reasons why a skilled artisan would have so modified the board. The Federal Circuit agreed that there was substantial evidence to make this conclusion.
With regard to claims 2 and 3, Belden did not dispute that two additional prior art references disclosed a third die. Instead, it asserted that there was no reason to combine this third die with JP '910. However, the Board had cited to the reference in the KSR decision to predictable variations. In other words, the Supreme Court had required consideration of factors such as "design incentives and other market forces" in determining whether a skilled artisan would have recognized the potential benefits and pursued the particular variations. Because the PTAB made the appropriate inquiry, including considering the expert report of Mr. Baxter, the Federal Circuit was satisfied that there was substantial evidence for the Board's conclusion.
As mentioned, the Federal Circuit disagreed with the Board with regard to claims 5 and 6, even giving it the deference that it deserved. The PTAB had found that two pieces of prior art could be combined to teach or suggest these claims. However, it did not think that there was any motivation to so combine these teachings. The Federal Circuit disagreed, stating that the conclusion in the Final Written Decision did not "withstand[] scrutiny through the lens of governing law." First, the Board had concluded that JP '910 only taught conductors that were not insulated. However, the Federal Circuit noted that the Board had already determined that JP '910 disclosed aligning the wires with the core, and how to do so. This alignment problem, the Court explained, did not depend on whether the wires were insulated. A prior art reference must be considered for everything it teaches, and is not limited to the particular invention described. In addition, the Board had found compelling the patent owner's argument that if the conductors were insulted, the final jacketing step would have been redundant. However, the Federal Circuit pointed out that the final step did not require that the jacketing be insulted. Finally, the patent owner had made an argument that modifying JP '910 to included twisted pairs would destroy the circular shape of the wire. Again, the Federal Circuit pointed out that the claims did not require a circular shape.
The most significant aspect of the Federal Circuit's decision with regard to claims 5 and 6 is that it did not vacate and remand for the Board to make the appropriate finding. Instead, it found that the record was already sufficient to reverse the Board because the petitioner had apparently proven by a preponderance of the evidence that the claims were obvious. For good reason, the opinion is silent about whether the same would have been true under the clear and convincing standard used by district courts. A patent owner found in a similar situation would be wise to consider this case and convince the Federal Circuit why the Board had substantial evidence to reach the conclusion that it did.
Motion to Exclude
Belden also lost its challenge of the Board's denial to exclude the Baxter declaration submitted after the Patent Owner's response. Belden had argued that the information presented in the declaration could have been, and should have been, included with the petition. Instead, as the argument goes, Berk-Tek essentially "sandbagged" the patent owner with its reply. Correspondingly, the patent owner complained that it lacked a fair opportunity to respond. Nevertheless, the Federal Circuit rejected all of these arguments.
For the first argument, the Court noted that Mr. Baxter responded to the Patent Owner response, and to the declaration submitted by Berk-Tek's expert, Mr. Clark. Of course, little mention was made of the fact that the same arguments could have been presented before the patent owner was required to file a response, and why therefore did Mr. Clark's declaration essentially white-wash the omission.
With regard to the second argument, the Federal Circuit pointed out that the Patent Owner had plenty of opportunity to respond. These opportunities included, for example:
• It had the ability to cross-examine the expert and filed observations;
• It could have moved to exclude the declaration;
• It could have disputed the substance of the declaration at the oral hearing;
• It could have moved to submit a surreply;
• It could have requested that the Board waive or suspend the regulations preventing the Patent Owner an opportunity to respond.
For its part, the Board did hold the petitioner to a "response-only" standard for the new evidence, and ensured that the prima facie obviousness case did not rely on any new evidence. It also provided for cross-examination and submission of observations. Moreover, Belden moved to exclude the declaration (which the Board considered but obviously rejected). Because the Patent Owner took advantage of several of the opportunities, it was irrelevant that the other potential opportunities did not occur.
The take away lesson for a patent owner is to argue for as many of these alternatives as possible should a petitioner in the future undertake a similar strategy. Of course, this does set up a potential gamesmanship opportunity for petitioners. And, unfortunately for Belden, the guidance provided by the Federal Circuit was of little use for the patent owner in this case.
Belden Inc. v. Berk-Tek LLC (Fed. Cir. 2015)
Panel: Circuit Judges Newman, Dyk, and Taranto
Opinion by Circuit Judge Taranto











