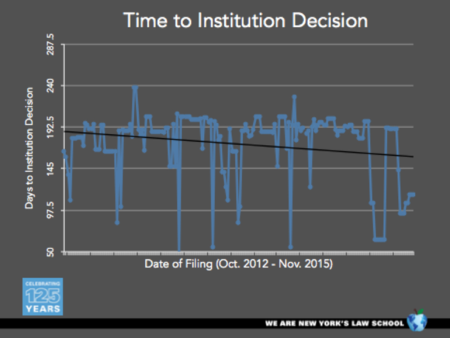
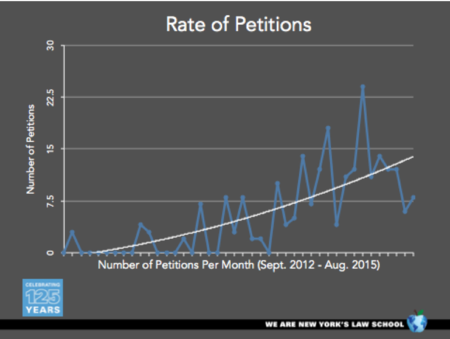
By Sherri Oslick —
 About Court Report: Each week we will report briefly on recently filed biotech and pharma cases.
About Court Report: Each week we will report briefly on recently filed biotech and pharma cases.
Forest Laboratories, LLC et al. v. Amerigen Pharmaceuticals, Inc. et al.
1:15-cv-00966; filed October 23, 2015 in the District Court of Delaware
• Plaintiffs: Forest Laboratories, LLC; Forest Laboratories Holdings, Ltd.; Adamas Pharmaceuticals, Inc.
• Defendants: Amerigen Pharmaceuticals, Inc.; Amerigen Pharmaceuticals Ltd.
Infringement of U.S. Patent Nos. 8,039,009 ("Modified Release Formulations of Memantine Oral dosage Forms," issued October 18, 2011), 8,058,291 ("Methods and Compositions For the Treatment of CNS-Related Conditions," issued November 15, 2011), 8,168,209 ("Method and Composition for Administering an NMDA Receptor Antagonist to a Subject," issued May 1, 2012), 8,173,708 (same title, issued May 8, 2012), 8,283,379 ("Method and Compositions for the Treatment of CNS-Related Conditions," issued October 9, 2012), 8,293,794 (same title, issued October 23, 2012), 8,329,752 ("Composition for Administering an NMDA Receptor Antagonist to a Subject," issued December 11, 2012), 8,338,485 ("Compositions for the Treatment of CNS-Related Conditions," issued December 25, 2012), 8,338,486 ("Methods for the Treatment of CNS-Related Conditions," issued December 25, 2012), 8,362,085 ("Method for Administering an NMDA Receptor Antagonist to a Subject," issued January 29, 2013), 8,580,858 ("Compositions For the Treatment of CNS-Related Conditions," issued November 12, 2013), and 8,598,233 ("Method for Administering an NMDA Receptor Antagonist To A Subject," issued December 3, 2013) following a Paragraph IV certification as part of Amerigen's filing of an ANDA to manufacture a generic version of Forest's Namzaric ® (memantine hydrochloride extended-release and donepezil hydrochloride, used for the treatment of moderate to severe dementia of the Alzheimer's type). View the complaint here.
Tris Pharma, Inc. v. Actavis Laboratories FL, Inc. et al.
1:15-cv-00969; filed October 23, 2015 in the District Court of Delaware
• Plaintiff: Tris Pharma, Inc.
• Defendants: Actavis Laboratories FL, Inc.; Andrx Corp.; Actavis, Inc.; Actavis Pharma, Inc.
Tris Pharma, Inc. v. Actavis Laboratories FL, Inc. et al.
2:15-cv-07684; filed October 23, 2015 in the District Court of New Jersey
• Plaintiff: Tris Pharma, Inc.
• Defendants: Actavis Laboratories FL, Inc.; Andrx Corp.; Actavis, Inc.; Actavis Pharma, Inc.
The complaints in these cases are substantially identical. Infringement of U.S. Patent No. 9,040,083 ("Orally Effective Methylphenidate Extended Release Powder And Aqueous Suspension Product," issued May 26, 2015) following a Paragraph IV certification as part of Actavis' filing of an ANDA to manufacture a generic version of Tris' Quillivant XR® (methylphenidate hydrochloride oral suspension, used to treat attention deficit hyperactivity disorder, distributed by Pfizer). View the Delaware complaint here.
Takeda Pharmaceutical Co. Ltd. et al. v. Aurobindo Pharma Ltd. et al.
1:15-cv-09398; filed October 23, 2015 in the Northern District of Illinois
• Plaintiffs: Takeda Pharmaceutical Co. Ltd.; Takeda Pharmaceuticals USA Inc.; Takeda Pharmacticals America, Inc.
• Defendants: Aurobindo Pharma Ltd.; Aurobindo Pharma U.S.A., Inc.; Aurolife Pharma LLC
Infringement of U.S. Patent Nos. 6,328,994 ("Orally Disintegrable Tablets," issued December 11, 2001), 7,431,942 ("Orally Disintegrable Tablets," issued October 7, 2008), 7,875,292 ("Orally Disintegrable Tablets," issued January 25, 2011), and 7,399,485 ("Rapidly Disintegrable Solid Preparation," issued July 15, 2008) following a Paragraph IV certification as part of Aurobindo's filing of an ANDA to manufacture a generic version of plaintiffs' Prevacid® SoluTab (lansoprazole delayed release orally disintegrating tablets, used to treat ulcers, gastroesophageal reflux disease, erosive esophagitis, and pathological hypersecretory conditions, including Zollinger-Ellison syndrome). View the complaint here. [NB: The complaint was later voluntarily dismissed.]
Mallinckrodt LLC et al. v. Par Pharmaceutical Inc.
2:15-cv-07694; filed October 23, 2015 in the District Court of New Jersey
• Plaintiffs: Mallinckrodt LLC; Mallinckrodt Inc.; Depomed, Inc.
• Defendant: Par Pharmaceutical Inc.
Infringement of U.S. Patent Nos. 6,488,962 ("Tablet Shapes to Enhance Gastric Retention of Swellable Controlled-Release Oral Dosage Forms," issued December 3, 2002), 8,597,681 ("Methods of Producing Stabilized Solid Dosage Pharmaceutical Compositions Containing Morphinans," issued December 3, 2013), 8,658,631 ("Combination Composition Comprising Oxycodone and Acetaminophen for Rapid Onset and Extended Duration of Analgesia," issued February 25, 2014), 8,741,885 ("Gastric Retentive Extended Release Pharmaceutical Compositions," issued June 3, 2014), 8,980,319 ("Methods of Production Stabilized Solid Dosage Pharmaceutical Composition Containing Morphinans," issued March 17, 2015), 8,992,975 ("Methods of Producing Stabilized Solid Dosage Pharmaceutical Compositions Containing Morphinans," issued December 3, 2013), 7,976,870 ("Gastric Retentive Oral Dosage Form with Restricted Drug Release in the Lower Gastrointestinal Tract," issued July 12, 2011), 8,668,929 ("Gastric Retentive Extended-Release Dosage Forms Comprising Combinations of a Non-Opioid Analgesic and an Opioid Analgesic," issued March 11, 2014), 8,372,432 (""Gastric Retentive Extended-Release Dosage Forms Comprising Combinations of a Non-Opioid Analgesic and an Opioid Analgesic," issued February 12, 2013), 8,377,453 (same title, issued February 19, 2013), 8,394,408 (same title, issued March 12, 2013), and 9,050,335 ("Pharmaceutical Compositions for Extended Release of Oxycodone and Acetaminophen Resulting in a Quick Onset and Prolonged Period of Analgesia," issued June 9, 2015) following a Paragraph IV certification as part of Par's filing of an ANDA to manufacture a generic version of Mallinckrodt's Xartemis XR (acetaminophen and oxycodone, used for the management of acute pain severe enough to require opioid treatment and for which alternative treatment options are inadequate). View the complaint here.
Pfizer Inc. et al. v. Mylan Inc. et al.
1:15-cv-00188; filed October 23, 2015 in the Northern District of West Virginia
• Plaintiffs: Pfizer Inc.; Wyeth LLC; Pfizer Pharmaceuticals LLC; PF Prism C.V.; Pfizer Manufacturing Holdings LLC
• Defendants: Mylan Inc.; Mylan N.V.; Mylan Laboratories Ltd.; Mylan Pharmaceuticals Inc.
Infringement of U.S. Patent Nos. 7,879,828 ("Tigecycline Compositions and Methods of Preparation," issued February 1, 2011), 8,372,995 ("Crystalline Solid Forms of Trigecycline and Methods of Preparing Same," issued February 12, 2013), and 8,975,242 ("Tigecycline Compositions and Methods of Preparation," issued March 10, 2015), licensed to Pfizer, following a Paragraph IV certification as part of Mylan's filing of an ANDA to manufacture a generic version of Pfizer's Tygacil® (tigecycline injectible, used for the treatment of complicated skin and skin structure infections, complicated intra-abdominal infections, and community-acquired bacterial pneumonia). View the complaint here.
 The Federal Circuit Bar Association (FCBA), in conjunction with the John Marshall Law School, the Notre Dame Law School, and DePaul University College of Law will be offering a program on "Patent Litigation 2016: The Courts and the Patent Trial and Appeal Board" on April 28, 2016 from 8:45 am to 5:00 pm (CT) at the John Marshall Law School in Chicago, Illinois. The program will include presentations on the following topics:
The Federal Circuit Bar Association (FCBA), in conjunction with the John Marshall Law School, the Notre Dame Law School, and DePaul University College of Law will be offering a program on "Patent Litigation 2016: The Courts and the Patent Trial and Appeal Board" on April 28, 2016 from 8:45 am to 5:00 pm (CT) at the John Marshall Law School in Chicago, Illinois. The program will include presentations on the following topics: