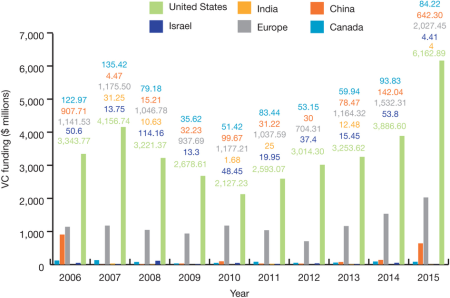
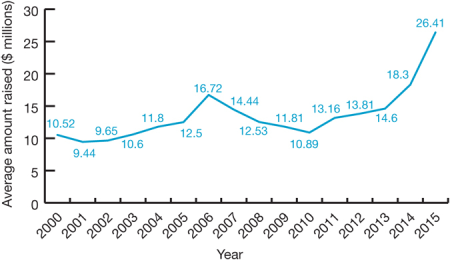
By Kevin E. Noonan —
 Many of the complaints from patent holders over the PTO's inter partes review process under the Leahy-Smith America Invents Act (codified in pertinent part at 35 U.S.C. §§ 311-319) stem from how the Office has implemented these proceedings (at §§ 42.1-42.80 and 42.100-42.123). Genzyme has addressed their complaints about the process, in the context of a challenge under the Administrative Procedures Act (5 U.S.C. § 500 et seq.) in a petition for panel rehearing or en banc review of the Federal Circuit's decision affirming invalidation of the claims of Genzyme's U.S Patent Nos. 7,351,410 and 7,655,226, in Genzyme Therapeutic Products, Inc. v. Biomarin Pharmaceutical, Inc.
Many of the complaints from patent holders over the PTO's inter partes review process under the Leahy-Smith America Invents Act (codified in pertinent part at 35 U.S.C. §§ 311-319) stem from how the Office has implemented these proceedings (at §§ 42.1-42.80 and 42.100-42.123). Genzyme has addressed their complaints about the process, in the context of a challenge under the Administrative Procedures Act (5 U.S.C. § 500 et seq.) in a petition for panel rehearing or en banc review of the Federal Circuit's decision affirming invalidation of the claims of Genzyme's U.S Patent Nos. 7,351,410 and 7,655,226, in Genzyme Therapeutic Products, Inc. v. Biomarin Pharmaceutical, Inc.
 The case arose over Biomarin's IPR of claims directed to methods for treating Pompe's disease, a deficiency in the lysosomal enzyme acid α-glucosidase (GAA), which is expressed as an inability to break down glycogen, particularly in muscle, causing glycogen buildup in muscle tissue. Claim 1 of the '410 patent is representative of the claims involved in the IPR:
The case arose over Biomarin's IPR of claims directed to methods for treating Pompe's disease, a deficiency in the lysosomal enzyme acid α-glucosidase (GAA), which is expressed as an inability to break down glycogen, particularly in muscle, causing glycogen buildup in muscle tissue. Claim 1 of the '410 patent is representative of the claims involved in the IPR:
A method of treating a human patient with Pompe's disease, comprising intravenously administering biweekly to the patient a therapeutically effective amount of human acid alpha glucosidase, whereby the concentration of accumulated glycogen in the patient is reduced and/or further accumulation of glycogen is arrested.
Biomarin requested an IPR in 2013 based on four grounds for the '410 patent, with the PTAB instituting on two of them: under § 103 based on the combination of the Duke press release and two prior art references (the Barton reference and van der Ploeg '88 references); and also under § 103 for the combination of the another reference, the Reuser reference, with the Barton and van der Ploeg '88 references. For the '266 patent, the PTAB instituted under § 103 for claims 1 and 3 based on the combination of the Duke press release, the van der Ploeg '88 reference, and the van Hove reference, and for claims 4-6 based on the combination of the Duke press release, the van der Ploeg '88 reference, the Barton reference, and the Reuser reference.
Genzyme responded to Biomarin's request by arguing that none of the cited references disclosed the results of in vivo tests in humans or animals. In response, Biomarin asserted two additional references that disclosed in vivo administration of M6P-modified GAA in mouse (van der Ploeg '91) and Japanese quail (Kikuchi).
The Board found the challenged claims to be obvious, based on disclosure in the Reuser reference of all elements of the claimed invention except the dosing interval which, according to the PTAB, was the result of "routine optimization." With regard to whether there was a reasonable expectation of success, the Board stated that "all that remained to be achieved over the prior art was the determination that a specific dose within a previously suggested dose range, and its corresponding dosing schedule, would have been safe and effective for the treatment of human patients," basing this portion of its determination on the cited art, including the Kikuchi and van der Ploeg art submitted by Biomarin during the trial phase of the IPR proceeding (i.e., after the Board had rendered its decision to initiate the IPR).
 The Federal Circuit affirmed, in an opinion by Judge Bryson, joined by Judges Moore and Reyna. Relevant to the rehearing petition, the panel rejected Genzyme's procedural challenge under the Administrative Procedures Act (APA), that the Board had erred in relying on "facts and legal arguments" not asserted in the request, as a violation of the Act's notice and opportunity to respond requirements. While acknowledging that "formal adjudication" such as an IPR imposes "certain procedural requirements" on the PTO under the APA, including "timely notice" and an opportunity to "submit facts and argument" under 5 U.S.C. §§ 554(b)-(c), 557(c), the Court stated that these provisions were intended to prevent an agency from "chang[ing] theories in midstream" without giving a respondent the opportunity to address the changed theory, citing Belden v. Berk-Tek LLC, 805 F.3d 1064, 1080 (Fed. Cir. 2015) (quoting Rodale Press, Inc. v. FTC, 407 F.2d 1252, 1256-57 (D.C. Cir. 1968). This was not the case here, according to the opinion, because Genzyme had ample notice and opportunity to rebut Biomarin's obviousness case against its claims. Support for this conclusion was had by noting that the Board based its final determination of obviousness on the same references it used for deciding to institute the IPR. The fact that the Board cited references (the in vivo references, van der Ploeg '91, and Kikuchi) in its final determination that were not included in the combination used to institute the IPR was not to the contrary, because "the introduction of new evidence in the course of the trial is to be expected in inter partes review trial proceedings and, as long as the opposing party is given notice of the evidence and an opportunity to respond to it, the introduction of such evidence is perfectly permissible under the APA."
The Federal Circuit affirmed, in an opinion by Judge Bryson, joined by Judges Moore and Reyna. Relevant to the rehearing petition, the panel rejected Genzyme's procedural challenge under the Administrative Procedures Act (APA), that the Board had erred in relying on "facts and legal arguments" not asserted in the request, as a violation of the Act's notice and opportunity to respond requirements. While acknowledging that "formal adjudication" such as an IPR imposes "certain procedural requirements" on the PTO under the APA, including "timely notice" and an opportunity to "submit facts and argument" under 5 U.S.C. §§ 554(b)-(c), 557(c), the Court stated that these provisions were intended to prevent an agency from "chang[ing] theories in midstream" without giving a respondent the opportunity to address the changed theory, citing Belden v. Berk-Tek LLC, 805 F.3d 1064, 1080 (Fed. Cir. 2015) (quoting Rodale Press, Inc. v. FTC, 407 F.2d 1252, 1256-57 (D.C. Cir. 1968). This was not the case here, according to the opinion, because Genzyme had ample notice and opportunity to rebut Biomarin's obviousness case against its claims. Support for this conclusion was had by noting that the Board based its final determination of obviousness on the same references it used for deciding to institute the IPR. The fact that the Board cited references (the in vivo references, van der Ploeg '91, and Kikuchi) in its final determination that were not included in the combination used to institute the IPR was not to the contrary, because "the introduction of new evidence in the course of the trial is to be expected in inter partes review trial proceedings and, as long as the opposing party is given notice of the evidence and an opportunity to respond to it, the introduction of such evidence is perfectly permissible under the APA."
Genzyme's argument to the contrary, according to the Court, was based on "misunderstanding of the role of the institution decision in inter partes review proceedings before the Board":
There is no requirement, either in the Board's regulations, in the APA, or as a matter of due process, for the institution decision to anticipate and set forth every legal or factual issue that might arise in the course of the trial. See Boston Carrier, Inc. v. ICC, 746 F.2d 1555, 1560 (D.C. Cir. 1984) (even when adjudicating charges of misconduct, an agency "is not burdened with the obligation to give every applicant a complete bill of particulars as to every allegation that carrier will confront"). Because the institution decision comes at the outset of the proceedings and the patentee is not obligated to respond before the Board makes its institution decision, it is hardly surprising that the Board cannot predict all the legal or factual questions that the parties may raise during the litigation.
Indeed, the opinion goes on to say that "development of evidence in the course of the trial is in keeping with the oppositional nature of an inter partes review proceeding," with the requestor asserting its invalidity case to the PTAB and the patent owner presenting amendments (sic), and with the Board then deciding whether the challenger has borne the burden of proving invalidity (citing the Senate legislative history of the IPR provisions, despite the differences in how Congress may have thought it was providing for IPRs and the PTAB's decisions on how to implement the statute).
The "critical question" under the APA is "whether Genzyme received 'adequate notice of the issues that would be considered, and ultimately resolved, at that hearing'" the Court opined, citing Pub. Serv. Comm'n of Ky. v. FERC, 397 F.3d 1004, 1012 (D.C. Cir. 2005). The panel concluded that Genzyme had not shown that there were any issues of fact or law used by the PTAB as the basis of its obviousness determinations for which Genzyme had not had adequate notice or opportunity to be heard. Finally, the opinion also noted that PTO procedures provide means for moving to exclude evidence (such as 37 C.F.R. § 42.64(c)) or to file a surreply when Biomarin introduced this evidence during the institution proceedings, neither of which procedural avenues Genzyme used.
In its petition for rehearing, Genzyme poses what it characterizes as a "precedent-setting" question:
Whether, to comply with the Administrative Procedure Act in an inter partes review proceeding, the Patent Trial and Appeal Board must provide a patent owner notice of the components of the legal and factual analysis that are necessary to a finding of unpatentability.
In support of its answer to this question (i.e., that the Board is compelled to provide express notice under circumstances arising in this IPR), petitioner asserts that the panel decision was contrary to Belden Inc. v. Berk-Tek LLC, 805 F.3d 1064 (Fed. Cir. 2015); In re Biedermann, 733 F.3d 329 (Fed. Cir. 2013); and Rambus Inc. v. Rea, 731 F.3d 1248 (Fed. Cir. 2013) (the latter two cases in the context of ex parte examination).
The petition asserts that the panel contravened this precedent because it held that the notice provision could be satisfied when the initiation decision included "(1) the prior art references that establish the claim limitations and (2) the statutory basis of invalidity asserted" but could omit the legal argument relied upon in the Final Written Decision provided that it was "in some way brought to the patent owner's attention" during the course of the trial. Here, according to Genzyme, there were two omissions: any discussion of the "reasonable expectation of success" prong of the obviousness test, and art disclosing in vivo data of effectiveness of the claimed method, at the time the IPR was initiated. Genzyme argues that this failure to provide notice under 5 U.S.C. § 554(b)(3) was outcome-determinative since Genzyme could have sworn behind the references if given timely notice but could not because they didn't have notice at a time when such an act would have been effective.
In addition, the petition characterizes the Board's reliance on the "reasonable expectation of success" argument as the Board "changing course" — somewhat paradoxically, Genzyme argues that the Board and the parties acknowledged that a § 103 challenge must establish that there would have been a reasonable expectation of success, but then criticizes the decision for not including the argument in the institution decision. Petitioner's rationale in support of this argument is that it matters not that the parties knew generically that the expectation of success question would be germane to obviousness generally. The notice Genzyme believes was required (and that it did not get) was that this issue would be considered by the Board in rendering its decision. In its view:
The panel's cramped reading of the APA's notice provision departs from this Court's precedent establishing, in the examination context, that "reliance on the same statutory basis and the same prior art references, alone, is insufficient to avoid making a new ground of rejection"—and thus violating the APA—"when the Board relies on new facts and rationales." In re Biedermann, 733 F.3d at 337 (quoting In re Leithem, 661 F.3d 1316, 1319 (Fed. Cir. 2011)). "The ultimate criterion," this Court has explained, "is whether the appellant has had before the PTO a 'fair opportunity to react to the thrust of the rejection.'" Rambus, 731 F.3d at 1255 (quoting In re Jung, 637 F.3d 1356, 1365 (Fed. Cir. 2011)). And the "thrust of the rejection change[s] when," for example, the Board finds "a new factual basis for the reason to combine," In re Biedermann, 733 F.3d at 338; see Rambus, 731 F.3d at 1256—even if the references being combined do not change.
The petition characterizes the panel decision as being contrary to this precedent, which the petition asserts stands (according to petition) for the proposition that the Board must only provide the statutory basis for invalidity — obviousness — and the prior art that supports the conclusion to satisfy the notice provisions. Not only is this not a proper interpretation of the law, in Genzyme's view, but the precedent relied upon seemingly has a higher notice standard for ex parte examination than the panel used in this case; the standard for IPR should be even more stringent than this precedent, according to the petition, in view of the fact that an IPR extinguishes an existing rather than a nascent property right.
The panel's position was that there were no "new grounds" of rejection and thus that the precedent was not contravened. The petition rebuts that assertion, stating that "the substance of the panel's analysis is incompatible with that precedent. There is no plausible dispute that the Board's final written decisions here relied on "new facts" (i.e., new in vivo references) "and rationales" (i.e., reasonable expectation of success) compared to the institution decisions," citing In re Biedermann, 733 F.3d at 337.
The petition also states that the APA itself does not provide any basis for "applying different notice standards in IPRs" or for making a distinction between those references "used to establish claim limitations" (for which there is no dispute that they must be noticed) and those that are used to set forth the state of the art (which the panel decision asserts need not be noticed). For petitioners, there is no distinction in effect between the "matters of fact and law" needed to establish that the elements recited by the claims are in the prior art and those needed to show that the skilled worker had a reasonable expectation of success. And in view of the importance of these issues, the petition asserts that if notice does not apply under the APA then the notice provision is "meaningless in the IPR context."
Petitioners also distinguish the panel's reliance on Ariosa Diagnostics v. Verinata Health, which stands for the proposition that the Board may rely on a reference to show the state of the art even if that reference was not part of the institution decision. Petitioners don't dispute that — here their argument is that the Board did not indicate that the in vivo references were relevant to the obviousness question and then had its legal determination of obviousness rest on those references. It is this legal analysis that petitioners say is new and for which they had no notice. The crux:
The panel's decision thus allows petitioners not only to "build a record by introducing evidence" (Slip Op. 10) but to cure fundamental defects in the institution decision after the patent owner points them out—and after the patent owner has any right to respond [emphasis added].
The real argument appears to be that if the institution decision is flawed, the Board should not get to cure during the trial; however, neither the Board nor the Federal Circuit may look favorably on such a procedural avenue to permit the patentee to prevail without addressing the substance.
The petition raises a secondary point: the need for "notice" to come from the Board and not from the record, i.e., the trial or the other party. This argument is based on a due process argument supported by precedent in other contexts that the agency must provide notice under the APA, citing Pub. Serv. Comm'n of Ky. v. F.E.R.C., 397 F.3d 1004, 1012 (D.C. Cir. 2005). It does not seem to be in dispute that the Board relied on Biomarin's introduction of the in vivo evidence and argument during the trial phase to provide notice, which petitioner says is improper.
Again, relating to the right to respond:
The petitioner is "expected" to introduce new evidence at the reply stage, Slip Op. 9—when the patent owner has no right to respond, Belden, 805 F.3d at 1081 ("no rule provides patent owners the right to file surreplies to a petitioner's Reply")—and once the petitioner has done so, the APA's notice requirement is satisfied, see Slip Op. 10-11. But at that point, it is too late.
In this regard the petition notes that the panel apparently believes that petitioner could have objected to introduction of this evidence during the IPR trial, but states that this is a question of admissibility not notice, and there is little to no basis to object on admissibility grounds. Also deficient was the panel's suggestion that this evidence could have been subject to a motion to exclude, because inter alia this would shift the burden to the patentee (plus running afoul of the PTO's overarching public interest concerns).
The petition raises the crux of many of the procedural objections patentees have raised against how the PTO has implemented IPRs: the Board's focus on speedy resolution within the 12 month period deforms many parts of the process, here by shortening the time for a patentee to respond to new evidence to its detriment and prejudice. The same argument could be made (and has) with regard to other procedural decisions (such as patentee's motions to amend) with some force; perhaps the Office should reconsider whether expeditiousness (or the perception of the overriding importance it has placed in compliance with the timing requirements of the Act) is worth the perception that the Office is not treating patentees and their patent property rights fairly. And it should escape no one's notice that in casting the issue in an administrative law context, the issues are ripe for Supreme Court review.
 The U.S. Patent and Trademark Office will be offering the next webinar in its Patent Quality Chat webinar series from 12:00 to 1:00 pm (ET) on September 13, 2016. In the latest webinar, which will provide a discussion on "Future Information Disclosure Practice in a Global Dossier Environment," the Office will explore whether to leverage electronic resources (e.g., Global Dossier and USPTO internal databases) to automatically import relevant information into pending U.S. applications from sources such as domestic parent and counterpart foreign applications. The webinar will also consider the potential benefits from the perspectives of examiners and applicants.
The U.S. Patent and Trademark Office will be offering the next webinar in its Patent Quality Chat webinar series from 12:00 to 1:00 pm (ET) on September 13, 2016. In the latest webinar, which will provide a discussion on "Future Information Disclosure Practice in a Global Dossier Environment," the Office will explore whether to leverage electronic resources (e.g., Global Dossier and USPTO internal databases) to automatically import relevant information into pending U.S. applications from sources such as domestic parent and counterpart foreign applications. The webinar will also consider the potential benefits from the perspectives of examiners and applicants.