By Nicholas Vincent* and Anthony D. Sabatelli** —
Many research efforts into the microbiome have focused primarily on the human microbiome, i.e., microorganisms within and on the body, and how changes in these microbial communities correlate with changes in health and disease. Less attention, however, has been paid to the microbial communities external to humans and how changes in these communities can affect health. These communities have a broad range, from the microbiome of indoor spaces, also called the microbiology of the built environment (MoBE), to microbial communities found outdoors. Microbial communities that give certain foods, such as San Francisco sour dough bread, various wines, beers, and even cheeses, characteristic qualities like taste and texture are also examples of external microbiomes. As with the human microbiome, scientists do not yet fully understand how changes in external or indoor microbiomes could alter human health, but we see plentiful possibilities for further research and intellectual property protection of subsequent innovation, especially with regards to the MoBE. This article concludes with a sampling of some of the patenting activity in this area.
Efforts to Understand the Environmental and Built Microbiomes
Initial research efforts into external microbiomes centered mostly around environmental microbial communities. The Earth Microbiome Project (EMP) is a large-scale research endeavor that has attempted to categorize and classify microbial communities in different biomes across the globe in an effort to elucidate the "Earth's microbiome" using a variety of metagenomic and metatranscriptomic approaches. These approaches focus on large-scale and often high-throughput data collection and analyses. Projects such as the EMP emphasize that the composition of complex environmental microbial communities that surround us are largely unknown to researchers.
Although projects such as the EMP look at environmental microbial communities, they leave out an important aspect of human life—the indoor microbiome, or the microbiology of the built environment (MoBE). Various research efforts are currently being undertaken to foster growth in this area. The National Academies of Science and Engineering have begun a consensus study to understand issues related to the MoBE including its formation and effects on human health. The end goal of the study is to gather enough information to apply the findings to novel building materials and architectural approaches. Although a report is expected in 2017, efforts to understand the indoor microbiome are still in their infancy. As this field expands, so will, we believe, opportunities for patentable innovation related to it.
Implications of an Altered Microbiome
It is largely unclear how an altered external or built microbiome will affect human health. Several initial investigations have sought to understand the role potential pathogens play as part of the indoor microbiome. Handorean, et al. determined that aerosolized particles from soiled linens in healthcare settings could contribute to pathogen exposure. Additionally Rhoads, et al. noted that temperature setting is critical in control of pathogens such as Legionella pneumophila in water heaters and water taps. Both of these findings suggest that environmental changes and the introduction of pathogens into external or indoor microbiomes can easily lead to potential exposure, suggesting the need for novel filtration and sterilization techniques and approaches.
Many researchers are also beginning to explore whether exposure to an altered indoor microbiome in infancy or childhood could lead to the development of adverse health outcomes like asthma and allergies. Several studies have further investigated the connection between an altered indoor environment and immunological development in children. Avershina, et al. found that high vacuum cleaning frequency is associated with an altered gut microbiome during pregnancy and for 2-year old children. In even younger subjects, Shin et al. found that operating room microbes may seed C-section-born children with microbes, and therefore the indoor microbiome of the birth room can play an especially important role in immune development and health in a subset of newborn children. Furthermore, King, et al. determined that shotgun sequencing could reveal normal hospital microbiomes and detect any fluctuations in microbial community composition, including the emergence of opportunistic pathogens. Research into MoBEs has not been restricted to hospitals: Hewitt, et al. found that bacterial diversity in offices varies from city to city.
Although research efforts have begun to elucidate the external and indoor microbiomes, many questions remain. In September 2016, Adams, et al. proposed ten questions regarding indoor microbiomes and data collection related to them, not only in an effort to advance the field, but also in an effort to standardize its methods, approaches, and interpretations. In short, much research is needed, but as we learn more, we see the opportunity emerging for novel patents.
Intellectual Property and Patent Considerations of External and Built Microbiomes
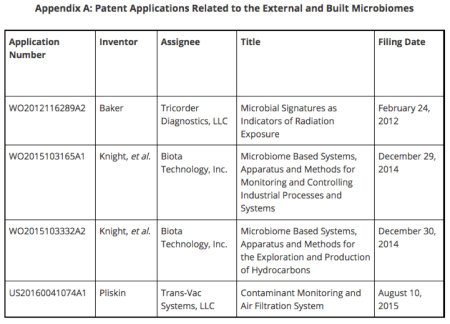
Although initial patent applications have been filed that are relevant to monitoring, sampling, and detecting external microbial communities, we expect the space to expand rapidly as more basic research is completed. In 2012, patent application WO2012116289A2 (for this and all other mentioned patent applications, see Appendix A) was filed describing microbial population changes in response to radiation, and using those changes as identifiers of the type of radiation that has occurred.
In 2015, patent application WO2015103165A1 was filed by Biota Technology, Inc. describing novel and unique methods, systems, and processes for monitoring, analyzing, and controlling the microbial presence in industrial activities. The company also filed patent application WO2015103332A2 describing novel and unique methods for characterizing the microbiome associated with hydrocarbon processes, including production and exploration.
Recently, Trans-Vac Systems LLC filed patent application US20160041074A1, which recognizes how humans alter the built microbiome both in living areas and medical settings, and proposes novel air monitoring and filtration system inventions to remove potential pathogens from MoBEs.
Closing
As research efforts rapidly expand, we expect growth in the need for patents to protect novel advancements related to the built environment. As we understand more with regards to how the indoor environment affects human health, we expect an increased need to patent novel inventions related to sterilization and filtration techniques and apparatuses. Further, as we learn how building materials and approaches can affect the indoor microbiome, we expect an increased need for patents ranging from actual building material products to architectural approaches. Additionally, previous patent applications (WO2015103165A1 and WO2015103332A2, for example) have highlighted the potential of microbiome-related intellectual property advances in the fields of fuel and energy exploration.
* Nicholas Vincent is a Technology Specialist at Dilworth IP
** Dr. Sabatelli is a Partner with Dilworth IP
For additional information regarding this topic, please see:
• "The Emergent Microbiome: A Revolution for the Life Sciences – Part V, Patents Relating to Obesity and Metabolic Disorders," February 28, 2016
• "The Emergent Microbiome: A Revolution for the Life Sciences – Part IV, Obesity and other Metabolic Disorders," February 18, 2016
• "Jackson Laboratory Hosts Microbiome Symposium Related to Cancer and Immunology," January 19, 2016
• "The Emergent Microbiome: A Revolution for the Life Sciences – Part III, Psychobiotics," October 13, 2015
• "The Emergent Microbiome: A Revolution for the Life Sciences – Part II, 2015 Patent Trends," August 11, 2015
• "The Emergent Microbiome: A Revolution for the Life Sciences – Part I, R&D Leaders," August 10, 2015