By Donald Zuhn —
 The U.S. Patent and Trademark Office recently released its Performance and Accountability Report Fiscal Year 2016. In describing the USPTO's strategic and performance-planning framework, the 2016 report reminds readers that the Office issued a new strategic plan in 2014, the 2014-2018 Strategic Plan. The report reiterates that the 2014-2018 Strategic Plan "demonstrates the progress made to date by building on the tangible successes of recent years with a focus on achieving the USPTO's vision as a global IP leader by:
The U.S. Patent and Trademark Office recently released its Performance and Accountability Report Fiscal Year 2016. In describing the USPTO's strategic and performance-planning framework, the 2016 report reminds readers that the Office issued a new strategic plan in 2014, the 2014-2018 Strategic Plan. The report reiterates that the 2014-2018 Strategic Plan "demonstrates the progress made to date by building on the tangible successes of recent years with a focus on achieving the USPTO's vision as a global IP leader by:
• Establishing the optimal pendency and quality levels for both patents and trademarks that will enable the USPTO to operate efficiently and effectively within the expectations of the IP community;
• Administering effectively the provisions of the AIA;
• Continuing to transform the USPTO with next-generation technology and services;
• Maintaining a strong and diverse leadership team, agile management structure, and a diverse and engaged cadre of employees in achieving the agency’s mission and vision;
• Continuing to work with other government agencies, Congress, and USPTO’s global partners to establish IP systems that benefit innovation, create jobs, and lead to strong economies around the world; and
• Recruiting and retaining the highest quality employees to accomplish the agency’s important work."
The 2016 Report specifies ten key performance outcome measures for which the Office has developed annual performance targets. According to the report, the Office met its annual performance targets for eight of the ten performance measures. Missing from this year's report was the Patent Quality Composite Score performance measure (for FY 2015, the Office did not meet its target for this measure, hitting eight of eleven performance targets). With regard to the omitted performance measure, the 2016 report explains in a footnote that:
The USPTO is working with internal and external stakeholders to reevaluate the entire quality process at the USPTO by engaging in public forums and roundtables to increase the effectiveness, clarity, and simplicity of the USPTO's quality review process by focusing on excellence in work products, excellence in measuring patent quality, and excellence in customer service. As part of this effort, the USPTO aims to define and introduce revised quality metrics based on stakeholder input by October 2016. Because the precise contours of the metric will likely change in the upcoming years, it is not useful to portray trends for the current measure.
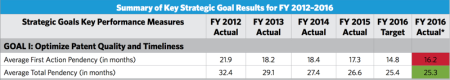
With respect to the remaining performance measures, two of the ten measures fall within the Office's first strategic goal, which concerns optimizing patent quality and timeliness. In particular, average first action pendency was 16.2 months (higher than the 14.8-month target, but lower than the 17.3-month average first action pendency of FY 2015), and average total pendency was 25.3 months (lower than the 25.4-month target and lower than the 26.6-month average total pendency of FY 2015).
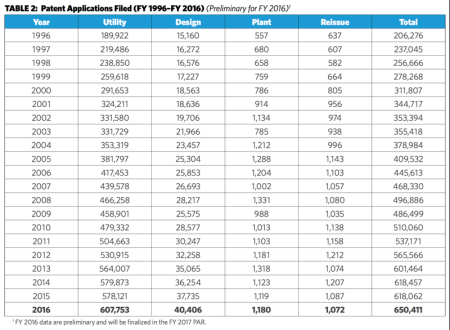
Table 2 of the report provides data for the patent-related performance targets for FY 2012 to FY 2016 (click on any table to expand):
The report also notes that the number of applications filed increased from 618,062 in FY 2015 to 650,411 in FY 2016, which constituted a 5.2% increase in filings (see Table 1 below). This followed a slight decrease in application filings in FY 2015 and a 2.8% increase in application filings in FY 2014.

The report also indicates that while the Office accepted more than 600,000 patent applications for the fourth straight year and topped 500,000 applications for the seventh consecutive year (see Table 2 below), it was able to reduce the number of applications awaiting action from 592,417 in FY 2015 to 570,074 in FY 2016 (see Table 3 below). The total number of pending applications also decreased from 1,099,468 in FY 2015 to 1,070,163 in FY 2016. It was the Office's third consecutive reduction in number of applications awaiting action and sixth consecutive reduction in total number of pending applications.

After dropping from 303,930 utility patent issuances in FY 2014 to 295,459 in FY 2015, patent issuances were once again on the rise in FY 2016, hitting 334,107 (see Table 6 below).

As noted above, the results for first action and total pendency were mixed, with first action pendency coming in above the Office's target, and total pendency coming in below the Office's target (see Tables 4 and 5 below). And the goals for FY 2017 present even tougher challenges: for first action pendency, the annual performance target drops from 14.8 months for FY 2016 (which the Office failed to meet) to 14.5 months for FY 2017, and for total pendency, the annual performance target drops from 25.4 months in FY 2016 (which the Office met) to 23.5 months in FY 2017.

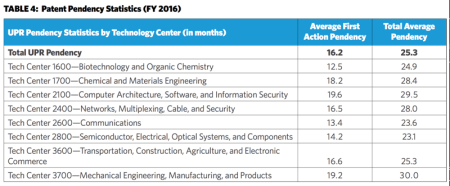
When comparing pendency statistics by Technology Center, Tech Center 1600 (biotechnology and organic chemistry) produced the best average first action pendency (12.5 months), and Tech Center 2100 (computer architecture, software, and information security) produced the worst average first action pendency (19.6 months) (see Table 4 below). As for total average pendency, Tech Center 2800 (semiconductor, electrical, optical systems, and components) produced the best total average pendency (23.1 months), and Tech Center 3700 (mechanical engineering, manufacturing, and products) produced the worst (30.0 months).
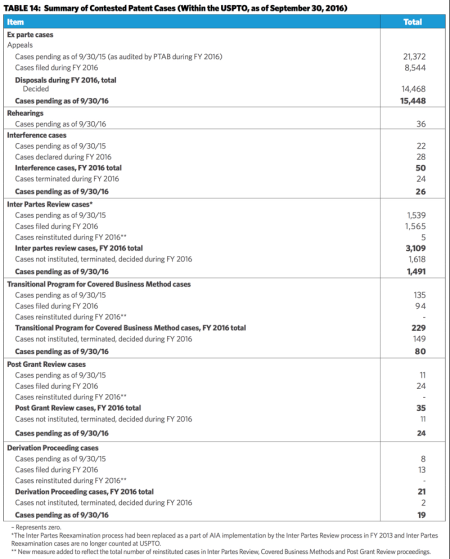
Finally, the report indicates that 1,565 inter partes review cases were filed in FY 2016, down from 1,737 in FY 2015, and 24 post grant review cases were filed in FY 2016, up from 11 in FY 2015 (see Table 14 below).
For additional information regarding this and other related topics, please see:
• "USPTO Issues Performance and Accountability Report for FY 2015," March 3, 2016
• "USPTO Issues Performance and Accountability Report for FY 2014," April 7, 2015
• "USPTO Releases Performance and Accountability Report for FY 2013," January 9, 2014
• "USPTO Releases Performance and Accountability Report for FY 2012," November 28, 2012
• "USPTO Releases Performance and Accountability Report for FY 2011," November 30, 2011
• "USPTO Releases 2010 Performance and Accountability Report," November 17, 2010
• "USPTO Announces 'Highest Performance Levels in Agency's History' in 2008," November 18, 2008
• "USPTO Announces 'Record Breaking' 2007 Performance," November 15, 2007
• "Patent Office Announces Record-Breaking Year," December 27, 2006