 Grünecker will be offering a two-day seminar on European Patent Law, including the UPC and Unitary Patent, on September 21-22, 2017 in Munich, Germany. The seminar will offer strategic and practical information on the "opt-out" and will walk through a UPC litigation (mock trial). Those wishing to register can do so here.
Grünecker will be offering a two-day seminar on European Patent Law, including the UPC and Unitary Patent, on September 21-22, 2017 in Munich, Germany. The seminar will offer strategic and practical information on the "opt-out" and will walk through a UPC litigation (mock trial). Those wishing to register can do so here.

Patent Law Weblog
recent posts
- USPTO Moves to Protect Design Rights for Digital Innovations
- Judicial Conduct and Disability Committee Has Its Say, Denies Judge Newman’s Latest Request for Review
- PTAB Issues Judgment on Priority in CRISPR Interference
- Parties File Supplemental Priority Statements in CRISPR Interference
- Why the Alice Test is Stupid, Part V: The Goalposts Keep Moving
about
-
-
Technology Transfer Tactics will be offering a webinar entitled "Best Practices for Safeguarding University IP When Structuring Deals in China" on September 19, 2017 from 1:00 to 2:30 pm (Eastern). Dan Harris of Harris Bricken will how to protect your technology and other IP interests from being counterfeited, pirated, or otherwise misappropriated by Chinese companies. The webinar will cover the following topics:
• How to choose a good Chinese partner
• Identifying your IP assets that need protection
• How to structure and draft your China deal:
– Licensing IP
– Selling IP
– Producing goods
• How to structure your China technology and IP licensing deals with concern for:
– Export issues
– Ownership rights
• Drafting China employee and third party contracts to protect your IP
• IP registrations: What you should know about trademarks, patents, copyrights, and licensing agreements
• Understanding the wants and needs of Chinese entities and universities and how to meet them
• Cultural nuancesThe registration fee for the webinar is $197. Those interested in registering for the webinar, can do so here.
-
 By Kevin E. Noonan –
By Kevin E. Noonan –The first scene in the iconic dystopian 1985 sci-fi movie Brazil by Terry Gilliam (one of the Monty Python troop) is of a peaceful citizen being snatched from his comfy chair by jackbooted, black-clad members of a SWAT team, who unceremoniously stuff him into a body bag-restraint as they take him away. As a final insult, they leave something behind for his wife and family — a bill, for the cost of taking him into custody.
This scene came to mind when a panel of the Federal Circuit held, on June 23rd of this year, that the provision of 35 U.S.C. § 145 (Civil Action to Obtain a Patent) providing that all expenses of the proceedings be borne by the plaintiff/appellant be construed to mean that a court can award attorney's fees to the Patent Office. This decision came as somewhat of a surprise to many at the time, and it seems to have raised eyebrows to a sufficient number of Federal Circuit judges that the Court today issued a per curiam, sua sponte order that the question be heard en banc. The order requests the parties submit briefs addressing this question:
Did the panel in NantKwest, Inc. v. Matal, 860 F.3d 1352 (Fed. Cir. 2017) correctly determine that 35 U.S.C. § 145's "[a]ll the expenses of the proceedings" provision authorizes an award of the United States Patent and Trademark Office's attorneys' fees?
Appellant's brief is due Monday, October 16th, and appellee's brief is due Tuesday, November 14th. Amicus briefs "will be entertained" on the question, without requiring consent of the parties or leave from the Court but subject to Fed. R. App. P. 29 and Federal Circuit Rule 29.
-
By Kevin E. Noonan –
 Patent law can be apparently inconsistent, particularly where claim construction is concerned. For example, claim construction standards that apply in district court are not the same standards that the U.S. Patent and Trademark Office applies during examination; the rationale for the difference is a mixture of a patent having a presumption of validity that claims undergoing examination (or the various flavors of re-examination or review) do not, as well as the gatekeeping function of the Office in granting claims commensurate in scope with an applicant's disclosure.
Patent law can be apparently inconsistent, particularly where claim construction is concerned. For example, claim construction standards that apply in district court are not the same standards that the U.S. Patent and Trademark Office applies during examination; the rationale for the difference is a mixture of a patent having a presumption of validity that claims undergoing examination (or the various flavors of re-examination or review) do not, as well as the gatekeeping function of the Office in granting claims commensurate in scope with an applicant's disclosure.Another area where there are differences between the Office and district courts is in applying the law of indefiniteness, i.e. determining whether claims satisfy the requirements of 35 U.S.C. § 112, second paragraph (renamed § 112(b) under the Leahy-Smith American Invents Act). The Supreme Court opined on the standard district courts should apply in Nautilus, Inc. v. Biosig Instruments, Inc., 572 U.S._, 134 S. Ct. 2120 (2014); in this precedential opinion, the Patent Trial and Appeal Board (PTAB) provided the Board's view on how the statute should be applied by the Office and the impact vel non of the Court's Nautilus decision on the Office with regard to the indefiniteness standard.
The claims of the application on appeal, U.S. Serial No. 13/435,655 to McAward et al., were directed to "water leakage detectors . . . which are easily connectable to flexible water hoses, and, can be coupled together to monitor leakage from hot and cold supplies." All claims were rejected; claim 1 being representative (with italics added):
1. A water detector comprising:
a housing;
flow connectors carried by the housing including a spin on female pipe connector at an inflow end and a spin-on male pipe connector on an outflow end;
at least one water presence sensor carried by the housing;
an electrically actuatable valve, carried by the housing, and having first and second states; and
control circuits, carried by the housing, coupled to the sensor and valve, and, responsive to a leakage indicating signal from the sensor, the circuits actuate the valve causing it to change state wherein the control circuits detect flood conditions, shut off a malfunctioning water branch to a home appliance and communicate to a premises alarm communication device or home automation system via a wireless link and wherein the water detector is configured to be reliably installed by an untrained installer or a homeowner and to not require the services of a plumber or electrician to perform installation, thereby permitting widespread and cost effective adoption.The italicized limitation was recited by the Examiner as being indefinite under 35 U.S.C. § 112, second paragraph. The Examiner asserted that the metes and bounds of this claim could not be understood by the person of ordinary skill in the art because "the claim language does not provide any structure to the apparatus or system that would allow it to be 'configured' to function as described in the claims."
This rejection was affirmed by the PTAB in a precedential decision in which the Board reaffirmed its application of the indefiniteness requirement in light of the Supreme Court's decision in Nautilus. In the opinion, the PTAB reiterates the PTO's role in having the initial burden of presenting a prima facie case of indefiniteness to reject claims on this basis, citing Hyatt v. Dudas, 492 F.3d 1365, 1369-70 (Fed. Cir. 2007), and In re Oetiker, 977 F.2d 1443, 1445 (Fed. Cir. 1992). This exercise requires claim construction (In re Zietz, 893 F.2d 319, 321-22 (Fed. Cir. 1989)) and that raised the issue of the claim construction standard the Office should use. The Board reiterated that the proper standard is the broadest reasonable interpretation (BRI) construction (In re Am. Acad. of Sci. Tech Ctr., 367 F.3d 1359, 1364 (Fed. Cir. 2004) (cited with approval in Phillips v. A WH Corp., 415 F.3d 1303, 1316 (Fed. Cir. 2005) (en banc)). Citing In re Packard, 751F.3d1307, 1311-12 (Fed. Cir. 2014) (per curiam), the opinion sets forth the rationale for this standard:
During prosecution, an examiner's indefiniteness rejection begins what is intended to be an interactive process in which the applicant has the opportunity to respond to the examiner by amending the claims or by providing evidence or explanation that shows why the claims are not indefinite.
This reasoning supports the Office's role in ensuring that the claims are "clear, unambiguous, and drafted as precisely as the art allows" so that "the rights afforded by patents [are] commensurate with the invention's contribution to the art," citing. Packard, 751 F.3d at 1313. Using BRI permits the Office to "fulfill this important role" according to the opinion, citing In re Buszard, 504 F.3d 1364, 1366 (Fed. Cir. 2007). An applicant has the opportunity to respond to such a rejection by amending claims to satisfy the § 112 requirements, citing In re Yamamoto, 740 F.2d 1569, 1571 (Fed. Cir. 1984) (and quoting In re Prater, 415 F.2d 1393, 1405 (CCPA 1969)). The Board further asserted that the Office's use of BRI was approved by the Supreme Court in Cuozzo Speed Techs., LLC v. Lee, 136 S. Ct. 2131, 2146 (2016), as being authorized under the Office's rulemaking authority. In Cuozzo, the Court's approval was grounded on the understanding that such a system protects the public by avoiding "overly broad claims" and "tying up too much knowledge."
And In re Morris, 127 F.3d 1048, 1054 (Fed. Cir. 1997), is consistent with this view:
We note that the patent drafter is in the best position to resolve the ambiguity in the patent claims, and it is highly desirable that patent examiners demand that applicants do so in appropriate circumstances so that the patent can be amended during prosecution rather than attempting to resolve the ambiguity in litigation.
Citing In re Packard, the opinion characterizes this regime as an approach that "fundamentally differs" from a court's role — one that "effectively results in a lower threshold for ambiguity than a court's":
The different approaches to indefiniteness before the Office and the courts stem not from divergent interpretations of § 112, but from the distinct roles that the Office and the courts play in the patent system. The lower threshold makes good sense during patent examination because the patent record is in development and not fixed, the Office construes claims broadly during that period, and an applicant may freely amend claims.
Nautilus, which was decided after Packard, is not contrary: "We do not understand Nautilus, however, to mandate a change in the Office's approach to indefiniteness in patent-examination matters in which, as discussed above, the claims are interpreted under the broadest reasonable interpretation standard and an opportunity to amend the claims is afforded." The government's position has been consistent, in its amicus in Nautilus and in opposing a writ of certiorari in Packard.
The PTO standard from Packard controls: "[a] claim is indefinite when it contains words or phrases whose meaning is unclear." 751 F.3d at 1310, 1314. Put differently, "claims are required to be cast in clear-as opposed to ambiguous, vague, indefinite-terms":
[W]hen the USPTO has initially issued a well-grounded rejection that identifies ways in which language in a claim is ambiguous, vague, incoherent, opaque, or otherwise unclear in describing and defining the claimed invention, and thereafter the applicant fails to provide a satisfactory response, the USPTO can properly reject the claim as failing to meet the statutory requirements of § 112(b). MPEP § 2173.02(1).
In applying these principles, the Board found that the claims are indefinite because "the claim language attempts to further define the water detector's structure by the skill level required to install the water detector" rather than "further defining the water detector's structure," which the opinion terms "unusual." For this reason, the claim language "fails to provide adequate clarity to the required structure" because the skill level of "an untrained installer or a homeowner" is "ambiguous and vague." The definitions the Applicant argued on appeal for the meaning of the term were not found in the specification according to the Board's opinion, particular with regard to the knowledge or tools needed for installing the claimed water detector. As with many such indefiniteness problems, here the wide variability in skill levels among "an untrained installer or a homeowner" robbed the phrase of the clarity required by the statute. In addition, the Board found that the specification did not provide a basis for determining what the term "configured" meant in the context of the disputed limitation. These deficiencies extended to both the plumbing and electrical components of the claimed device according to the Board.
The Board also affirmed rejection of all pending claims on obviousness grounds, construing narrowly the terms determined to be indefinite where, even under such a construction the claims were found to be obvious.
-
By Joseph Herndon —
 On July 27, 2017, the Federal Circuit issued an opinion in Audatex North America, Inc. v. Mitchell International, Inc., upholding the U.S. Patent and Trademark Office Patent Trial and Appeal Board's (PTAB) decision in which all claims of U.S. Patent Nos. 7,912,740 and 8,468,038 were held patent ineligible under 35 U.S.C. § 101 and invalid as obvious under 35 U.S.C. § 103.
On July 27, 2017, the Federal Circuit issued an opinion in Audatex North America, Inc. v. Mitchell International, Inc., upholding the U.S. Patent and Trademark Office Patent Trial and Appeal Board's (PTAB) decision in which all claims of U.S. Patent Nos. 7,912,740 and 8,468,038 were held patent ineligible under 35 U.S.C. § 101 and invalid as obvious under 35 U.S.C. § 103.Audatex initially sued Mitchell for infringement of the '740 and '038 patents. Mitchell then filed petitions requesting CBM review of the patents. The PTAB issued final written decisions in the CBMs holding that all challenged claims were found to be drawn to a patent ineligible abstract idea. A summary of one of the decisions can be found here.
Upon instituting both CBM proceedings, the Board held all claims ineligible under § 101 and invalid as obvious under § 103. Audatex filed a motion to amend, cancelling claims 1–29 of the '740 patent and proposing substitute claims 30–58. It similarly moved to amend the '038 patent, cancelling claims 1–31 and proposing substitute claims 32–62.
Claims 30 and 37 of the '740 patent are representative and are reproduced below.
30. A method for obtaining an automobile insurance claim valuation report of a damaged vehicle in association with the processing of an insurance claim, comprising:
transmitting a uniform resource locator over an electronic communication network from a client computer;
connecting with a web site that corresponds to the uniform resource locator, the web site provides a plurality of web pages that allows an operator to input data relating to an insurance claim for the damaged vehicle, the insurance claim being a request to recover market value or repair cost in association with an insurance policy;
entering data relating to the insurance claim;
providing a parts list and calculated estimate data through the web site; processing the entered data to generate a valuation report for the damaged vehicle, the valuation report provides a market value for the damaged vehicle, before the damaged vehicle was damaged, based on factors including mileage, condition, and geographic location; and,
transmitting the valuation report to the client computer over the electronic communication network through the web site.37. The method of claim 30, further comprising transmitting the valuation report from a valuation server to a web server before transmitting the valuation report to the client computer, the valuation report being generated by the valuation server with a database of vehicle values that is called by a first active server page, the parts list and calculated estimate data being provided by a program called by a second active server page.
Turning to the patents at issue, the '038 patent is a continuation of the '740 patent. Entitled "system and method for processing work products for vehicles via the world wide web," these patents relate to systems for entering data associated with an insurance claim for damaged vehicles. The systems process these data into a valuation report transmitted via the Internet, allowing insurance claims adjusters to access a vehicle valuation database more readily.
According to Audatex, the substituted claims solve problems associated with utilizing data from constantly changing databases for multiple customers through the use of Active Server Page ("ASP") files. Claim 37, for example, employs two such ASP files: the first for calling a database of vehicle values; the second to call a program for providing vehicle parts lists and calculated estimate data. These ASP calls allow the valuation server to generate valuation reports that adjusters use for their calculations. The proposed claims thus describe methods and systems for obtaining automobile insurance claim valuation reports by combining calls to multiple databases that provide customized insurance estimate tools for insurance adjusters based on their specific needs.
After considering these amendments, the Board maintained its ineligibility and obviousness grounds for the proposed claims. Audatex appealed on both grounds.
Patent Eligibility
The Federal Circuit followed the two-step process under Alice to determine patent eligibility, where step one analyzes for an "abstract idea", and step two analyzes for an "inventive concept".
Turning to step one, Audatex argued that the proposed claims are not abstract, but rather are directed to specific improvements of computer-related technology. It contended that the claims provide "a very specific way of generating a valuation report, including a special-purpose valuation server that processes the data and generates the valuation report with a database of vehicles values." It emphasized that the system employs a first ASP to call this database and requires a second ASP to call the parts lists and calculated estimate data. It concluded that these features, combined with other elements such as an electronic network, a client computer, and a web server that allows for the input of data relating to an insurance claim and transmission of a valuation report, render the claims non-abstract.
The Federal Circuit, however, found that the proposed claims are directed to the abstract idea of "providing a vehicle valuation through the collection and use of vehicle information." Here, the Federal Circuit noted that the proposed claims recite nothing more than the collection of information to generate a valuation report for a damaged vehicle with the aid of well-known technology.
The claims were found to embody an abstract idea that merely uses a computer and generic components as tools to collect these data and generate reports.
Regarding step two, the Federal Circuit found that the proposed claims neither improve the technological infrastructure nor provide solutions to challenges particular to the Internet. Rather, the claims simply add computer functionality and recite use of the Internet to increase the speed and efficiency of an abstract process.
Audatex relied heavily on the two ASPs recited in the claims. But Audatex itself conceded that it did not invent ASPs, and the claims do not recite them in a manner that produces "a result that overrides the routine and conventional" use of these known features. Thus, the Federal Circuit concluded that when viewed as an ordered combination, the proposed claims recite no more than the sort of "perfectly conventional" generic computer components employed in a customary manner that the Court has held insufficient to transform the abstract idea into a patent-eligible invention.
As a result, the Federal Circuit concluded that the proposed claims recite patent-ineligible subject matter under § 101. The Federal Circuit did not reach the obviousness grounds of Audatex's appeal.
The Federal Circuit's analysis is short and concise, and simply rejected or disagreed with the position taken by Audatex.
Looking at the substitute claims, one issue to consider is whether the claims recite how the functions are performed. In situations where the functional terms are generic and the claim lacks details of how to carry out the specific function, courts are more likely to find the claims patent-ineligible. Here, the claims recite "transmitting," "connecting," "entering," "providing," and "transmitting," but none of the method steps really recite how to perform those functions. Without such details, courts generally will determine that the claim is directed to the benefit of the invention rather than the invention itself. The invention needs to be recited in the claims, and the invention is how you achieve the benefit. Thus, with broadly recited functions as seen here, the claims are more likely to be considered patent-ineligible.
Audatex North America, Inc. v. Mitchell International, Inc. (Fed. Cir. 2017)
Nonprecedential disposition
Panel: Chief Judge Prost and Circuit Judges O'Malley and Chen
Opinion by Chief Judge Prost -
District Court Finds Claims Directed to Method for Analyzing Enoxaparin Sample to Be Patent Eligible
By Donald Zuhn —
 Last month, in Momenta Pharmaceuticals, Inc. v. Amphastar Pharmaceuticals, Inc., District Judge Nathaniel M. Gorton of the U.S. District Court for the District of Massachusetts allowed a motion for judgment as a matter of law filed by Plaintiffs Momenta Pharmaceuticals, Inc. and Sandoz Inc. ("Momenta") contending that two affirmative defenses asserted by Defendants Amphastar Pharmaceuticals, Inc. and International Medication Systems, Ltd. ("Amphastar") were inapplicable. Amphastar had asserted the affirmative defenses of patent ineligible subject matter and indefiniteness in response to Momenta's claim that Amphastar's sale of generic enoxparain products infringed U.S. Patent No. 7,575,886. The instant post addresses Amphastar's affirmative defense of patent ineligible subject matter.
Last month, in Momenta Pharmaceuticals, Inc. v. Amphastar Pharmaceuticals, Inc., District Judge Nathaniel M. Gorton of the U.S. District Court for the District of Massachusetts allowed a motion for judgment as a matter of law filed by Plaintiffs Momenta Pharmaceuticals, Inc. and Sandoz Inc. ("Momenta") contending that two affirmative defenses asserted by Defendants Amphastar Pharmaceuticals, Inc. and International Medication Systems, Ltd. ("Amphastar") were inapplicable. Amphastar had asserted the affirmative defenses of patent ineligible subject matter and indefiniteness in response to Momenta's claim that Amphastar's sale of generic enoxparain products infringed U.S. Patent No. 7,575,886. The instant post addresses Amphastar's affirmative defense of patent ineligible subject matter.The '886 patent is directed at a set of manufacturing quality control processes that ensure that each batch of generic enoxaparin includes the individual sugar chains characteristic of enoxaparin sodium, which is marketed under the brand name LOVENOX. Representative claim 1 of the '886 patent recites:
1. A method for analyzing an enoxaparin sample for the presence or amount of a non naturally occurring sugar associated with peak 9 of FIG. 1 that results from a method of making enoxaparin that included β-eliminative cleavage with a benzyl ester and depolymerization, comprising:
providing an enoxaparin sample that has been exhaustively digested with two or more heparin degrading enzymes;
using a separation method to determine, in the enoxaparin sample that has been contacted with two or more heparin degrading enzymes, the presence of a structural signature associated with the non naturally occurring sugar associated with peak 9 of FIG. 1 that results from a method of making enoxaparin that includes β-eliminative cleavage with a benzyl ester and depolymerization;
making a determination about the enoxaparin sample based upon a comparison of the determination of the presence of a structural signature associated with the non naturally occurring sugar associated with peak 9 to a reference standard for enoxaparin, and
determining the presence of the structural signature associated with the non naturally occurring sugar associated with peak 9 of FIG. 1 in a second batch of enoxaparin, to thereby analyze the enoxaparin sample.Two days after Amphastar received FDA approval to market its generic enoxaparin product, Momenta filed a complaint alleging that Amphastar infringed the '886 patent by manufacturing generic enoxaparin for commercial sale using its patented method. After twice reaching the Federal Circuit (see "Momenta Pharmaceuticals, Inc. v. Amphastar Pharmaceuticals, Inc. (Fed. Cir. 2012)" and "Momenta Pharmaceuticals, Inc. v. Teva Pharmaceuticals USA Inc. (Fed. Cir. 2015)"), the case returned to the District Court where Amphastar moved for summary judgment of invalidity (on the grounds of lack of patent eligible subject matter and indefiniteness) and non-infringement, and Momenta cross-moved for summary judgment of dismissal of the equitable defenses of waiver and estoppel. In a decision issued in June (see Memonrandum & Order), the District Court denied the motions filed by both Amphastar and Momenta.
With regard to Amphastar's motion for summary judgment of lack of patent eligible subject matter, Amphastar argued that each of the four steps of the claimed method for analyzing an enoxaparin sample involves patent ineligible subject matter. In particular, Amphastar contended that the first step of the claimed process involves the digestion of enoxaparin, which is a law of nature, the second step involves the identification and separation of that natural process, and the third and fourth steps involve the comparison between the tested product and a standard product, which are comparisons of abstract ideas. In response, Momenta countered that the exhaustive digestion of enoxaparin in the first step, which requires exposure of the enoxaparin sample to a chemical cocktail for a certain time and at a specific temperature, does not occur naturally, and that the process results in a mixture of long and short sugar chains that do not individually exist in nature. Momenta also disputed Amphstar's assertion that the second, third, and fourth steps of the claimed process rendered the claimed process patent ineligible. In denying Amphastar's motion, the District Court determined that after "[v]iewing the facts in the light most favorable to Momenta," Momenta had persuaded the Court "that the '886 patent '[is] directed to a new and useful method' of ensuring the quality of enoxaparin."
Turning to Momenta's motion for judgment as a matter of law on Amphastar's affirmative defense of patent ineligible subject matter, the District Court once again faced the issue of the patent eligibility of asserted claims. The Court began by outlining the two-step framework for determining patent eligible subject matter set forth in Mayo Collaborative Servs. v. Prometheus Labs., Inc., 132 S. Ct. 1289, 1293 (2012). In the first step, the Court must determine whether the claims are "directed" to a patent ineligible concept, such as a natural law, natural phenomenon or abstract idea, and if the claims are so directed, then the Court must determine whether the elements of the invention "transform" the claims into an application eligible for a patent. In allowing Momenta's motion, the District Court determined that:
Because the '886 patent "[is] directed to a new and useful method" of ensuring the quality of enoxaparin and involves a series of laboratory steps rather than a law of nature or abstract idea, this Court concludes that the asserted claims involve patentable subject matter. See CellzDirect, 827 F.3d at 1048.
Momenta Pharmaceuticals, Inc. v. Amphastar Pharmaceuticals, Inc. (D. Mass. 2017)
Memorandum & Order by District Judge Gorton -
By Juan Luis Serrano* —
 In the first and second installments of this series, we've explained the general purpose and the patents and marketing authorizations sections of the study published on August 9, 2017 by the Federal Commission for Economic Competition[1]. In this third installment we take a look at the sections on strategic behaviour and the analysis on litigation cases.
In the first and second installments of this series, we've explained the general purpose and the patents and marketing authorizations sections of the study published on August 9, 2017 by the Federal Commission for Economic Competition[1]. In this third installment we take a look at the sections on strategic behaviour and the analysis on litigation cases.For the purposes of this entry, we'll refer to Mexican authorities by their acronyms; COFECE for the antitrust body, COFEPRIS for the regulatory authority, and IMPI for the patent office.
The section on Strategic Behavior of the study by COFECE (3.4), is heavy with criticism to the innovative pharmaceutical industry; it starts with references to evegreening practices, and patent clustering as a way to block generic entrances.
Whereas the study does mention the main argument in favor of allowing several patents related to a single drug product (inventive merit on new formulations or uses), it quotes the policy decisions taken in countries such as Argentina and India which have limited this possibility, mentioning that such policies have required countries to strike a balance between national interests and TRIPS. There is also a reference to the practice in Brazil requiring the regulatory authority to provide a green light before a patent is issued.
The angle of the study in favor of the generic side of industry is clear in the section related to product switching (explained as a marketing strategy leading physicians to a new formulation of an existing product to take advantage of a more recent patent); there is a direct reference to the position of ANAFAM (a major association of generic drug manufacturers) on the issue without a counter-argument from the innovator's association.
This section concludes with a reference to pay for delay agreements which have been subject of antitrust analysis and litigation in the U.S. and Europe, but does not indicate whether any such agreement has taken place in Mexico or not. The Mexican legal system does not force companies to automatically report such an agreement, but if one takes place it could be subject of review by COFECE.
Overall, this section leads the reader to think that the current environment in Mexico is slanted in favor of innovator companies, with an underlying message that the authority will start to look at actions that have been found as anticompetitive in other markets to study them here.
Special relevance comes from the fact that the message is issued by a Government office; whereas other factors would certainly be in play before any major policy decision is taken (the position by Congress and the Patent Office, and current NAFTA renegotiations which will include IP related discussions), it seems clear that we should expect additional activity from COFECE on the review of practices which have been sanctioned abroad.
In the section "litigation related to patents and marketing authorizations" (3.5), the study lists some of the major judicial and policy decisions that have shaped the current regulatory environment for generic drug entry in the past 20 years, as follows:
a) The enactment of linkage regulations in 2003, which were initially interpreted by authorities as limited to patents covering active pharmaceutical ingredients;
b) The creation of a specialized IP Court in 2008;
c) The Supreme Court decision of 2010 establishing that linkage should be broadly interpreted to include formulation patents;
d) The addition of an opposition system to patent applications in the IP Law, also in 2010 (which does not extend to granted patents); and
e) The reform to the IP Law ordering the patent office to publish all decisions which modify a granted patent or trademark.The study then explains that litigation has taken place in the form of patent infringement actions, challenges to applications or authorizations granted to generic drug manufacturers, and judicial petitions of recognition of Regulatory Data Exclusivity.
Two additional shots are fired at patent holders: on page 52, first paragraph, the study states:
"Upon making this study, we had knowledge that some holders of patents related to an active ingredient which are already in the public domain, have intended to cause confusion in authorities in charge of the purchase of drugs for the public sector and Courts, even trying to persuade their competitors by informally warning them of the initiation of and infringement proceed if (said competitor) does not refrain from manufacturing any product with the relevant active ingredient."
This statement refers to acts which could be subject to antitrust or even criminal investigations. Again, this becomes quite relevant when the source is not an independent writer, but a Government office.
The last paragraph of page 52 is also relevant:
"In summary, innovative laboratories can extend product market exclusivity through the abuse of judicial proceedings established in our laws and thus place obstacles to generic drug entry to the market. This calls for a review of the linkage system in order to have a transparent tool between marketing authorizations and industrial property rights and with this reduce possible spaces of litigation which seek to delay generic entry."
Whereas as mentioned in the first entry of this series, I agree with the need for transparency, the blanket reference to judicial proceedings as abusive does seem over the top. I would argue that those judicial proceedings are not necessarily abusive per se but are on many occasions the only resource available to prevent or correct the actual invasion of a granted right.
The section closes with a summary of litigation cases, by active ingredient, indicating the parties and patents involved, and those cases where regulatory data exclusivity has been requested by judicial means.
In part IV of the series on the COFECE study, we'll look at the first eight recommendations of policy changes.
* Mr. Serrano is Of Counsel with ROMO DE VIVAR V.IP SERVICES, S.C.
-
By Donald Zuhn —
 Last week, in Tinnus Enterprises, LLC v. Telebrands Corp., U.S. Magistrate Judge John D. Love of the U.S. District Court for the Eastern District of Texas issued a report and recommendation ("Recommendation") on a Motion For Summary Judgment of No Inequitable Conduct and No Unclean Hands filed by Plaintiffs Tinnus Enterprises, LLC and Zuru Ltd. ("Tinnus"). In particular, Magistrate Judge Love recommended that Tinnus' motion for summary judgment be granted.
Last week, in Tinnus Enterprises, LLC v. Telebrands Corp., U.S. Magistrate Judge John D. Love of the U.S. District Court for the Eastern District of Texas issued a report and recommendation ("Recommendation") on a Motion For Summary Judgment of No Inequitable Conduct and No Unclean Hands filed by Plaintiffs Tinnus Enterprises, LLC and Zuru Ltd. ("Tinnus"). In particular, Magistrate Judge Love recommended that Tinnus' motion for summary judgment be granted.Magistrate Judge Love's recommendation is related to a patent dispute initiated by Tinnus, which filed suit against Defendants Telebrands Corp. and Bulbhead.com, LLC ("Telebrands"), alleging that Telebrands infringed Tinnus' U.S. Patent Nos. 9,242,749. Tinnus subsequently amended their complaint to allege infringement of U.S. Patent No. 9,315,282. Following the issuance of an injunction against Telebrands, the District Court's claim construction opinion, and the denial by the Court of summary judgment as to indefiniteness, Tinnus moved for summary judgment on no inequitable conduct. In their motion, Tinnus asserted that Telebrands could not meet the clear and convincing evidence standard with respect to Telebrands' allegations that (1) Tinnus, the inventor, and patent counsel intentionally withheld the Patent Trial and Appeal Board's decision to institute post-grant review of U.S. Patent No. 9,051,066, from which both the '749 and '282 patents claim the benefit, and (2) the applications that issued as the '749 and '282 patents improperly claimed priority to the application that issued as the '066 patent.
With respect to the second allegation, the Recommendation notes that Telebrands did not address this issue in their opposition to Tinnus' motion, and therefore concludes that "[g]iven that there is no opposition to summary judgment on this argument, the Court finds that summary judgment is appropriate." The Court therefore recommended that Tinnus' motion be granted as to no inequitable conduct based upon improperly claimed priority.
As for the first allegation, Tinnus' argued that Telebrands failed to show that the decision to institute post-grant review of the '066 patent was withheld from the Examiner because the evidence showed that the Examiner was aware of the decision. Tinnus also argued that Telebrands failed to produce evidence that the decision was material or that Tinnus, the inventor, or patent counsel had any intent to deceive. Telebrands countered that the institution of post-grant review of the '749 and '282 patents on the same grounds as the decision instituting post-grant review of the '066 patent established "but for materiality," and that an intent to deceive the Patent Office for failing to provide the Examiner with a copy of the decision on the '066 patent was "the single most reasonable inference that can be drawn from the facts surrounding prosecution of the patents-in-suit."
Regarding Telebrands' argument of intent to deceive, the Recommendation notes that "Defendants do not dispute that the Examiner was aware of the '066 PGR Institution Decision by the PTAB," adding that "[i]ndeed, the evidence shows that the applicant's interview summary with the Examiner indicated that the Examiner was aware of the PTAB's PGR Institution Decision." Telebrands, however, had argued that Tinnus' interview summary was a self-serving document that may not accurately reflect the substance of the interview. The Court addressed this argument by pointing out that Telebrands had deposed the patent counsel who had submitted the interview summary and "presumably did not question him about the details of the interview or the veracity of his statements because Defendants do not cite any testimony in support of their contention." In addition, the Court noted that "knowing that the Examiner was indeed aware of the decision, Defendants' position assumes that Examiner is somehow incapable of finding the PTAB's PGR Institution Decision on his own," adding that "[i]t would be difficult to understand how the USPTO could not obtain the necessary publicly available documents from its own administrative agency when needed." Contrary to Telebrands' argument that an intent to deceive was the single most likely inference to be drawn from the evidence, the Court determined that "the more likely inference that can be drawn is that prosecution counsel believed he had satisfied his duty of candor when he informed the Examiner of the PGR Institution Decision during an interview with the Examiner."
Turning to Telebrands' materiality argument, the Recommendation determined that Telebrands' argument of but for materiality "holds little weight" since "an institution decision is simply a finding that it is 'more likely than not' that at least one challenged claim is unpatentable, or raises a novel or unsettled legal question." Significantly, the Court pointed out that in its final written decision regarding the '066 patent, "the PTAB did not invalidate the '066 Patent based on the prior art that Defendants contend establishes 'but for' materiality."
Finding that Telebrands could not show a "specific intent to deceive," the Court recommended that Tinnus' motion with respect to the first allegation be granted as well. The Court therefore recommended that Tinnus' Motion For Summary Judgment of No Inequitable Conduct and No Unclean Hands be granted.
Tinnus Enterprises, LLC v. Telebrands Corp. (E.D. Tex. 2017)
Report and Recommendation of United States Magistrate Judge by Magistrate Judge Love -
By Juan Luis Serrano* —
 In the first installment of this series, we outlined the general contents of the study published on August 9, 2017 by the Federal Commission for Economic Competition[1], highlighting that the Antitrust authority is calling for an overhaul of several aspects of the drug market in Mexico to promote competition. In this second installment, we delve into the patents and marketing authorizations sections, including the author's comments on the study's recommendations.
In the first installment of this series, we outlined the general contents of the study published on August 9, 2017 by the Federal Commission for Economic Competition[1], highlighting that the Antitrust authority is calling for an overhaul of several aspects of the drug market in Mexico to promote competition. In this second installment, we delve into the patents and marketing authorizations sections, including the author's comments on the study's recommendations.For the purposes of this entry, we'll refer to Mexican authorities by their acronyms; COFECE for the antitrust body, COFEPRIS for the regulatory authority, and IMPI for the patent office.
The patents section of the study (3.1), contains an explanation of the status in Mexico for patent granting, explaining that Mexico allows for several types of patents on pharmaceutical products, including those covering active ingredients and its variants, formulations, uses and processes. This section is mostly academic, and does not require much additional comment.
This is followed by a section on drug marketing authorizations (3.2), where the first shots are fired; COFECE criticizes some of the positions taken by COFEPRIS, such as forming "generic approval packages" (several approvals are issued in bulk, along with a press conference, instead of approving products on a first filed/first released basis), a lack of adherence to regulatory approval times, and a lack of transparency on available information on marketing authorizations at COFEPRIS' website.
Fixing these problems will fall mostly on COFEPRIS. Information available on authorization processes (both for innovators and generics) is indeed sorely lacking, and the rates of approval within the regulatory timeframes are low, for all industry participants.
The next section (3.3) delves into our linkage system, which was enacted in 2003, has been subject to partial Supreme Court review, and has substantial differences with the one established in the U.S. through the Drug Price Competition and Patent Term Restoration Act of 1984.
The study points two major problems with linkage; lack of transparency is the first, as inquiries on possible patent infringement are done in communications between COFEPRIS and IMPI without any immediate disclosure or right of hearing by the applicant or the patent holder. Transparency mechanisms do allow companies to obtain copies of these communications, but many times after decisions have already been taken.
The second problem is that patents listed in the linkage gazette are not necessarily related to an approved drug product. Any patent covering an active ingredient or formulation can be listed regardless of whether it covers a product which would be subject of generic competition or not.
This is one of the sections where I agree with COFECE; the system, in order to function properly for both innovators and generic manufacturers should allow interested parties to know precisely which patents are covering an approved product, and both generic drug applicants and patent holders should have an opportunity to be heard, either before administrative authorities or in an infringement proceeding, before an authorization is granted or denied.
The following section of the study will be quite interesting for both companies and policy makers; section 3.4 "Strategic behavior" refers to concepts that innovative companies will very likely object to, such as patent evergreening, patent clustering, and reference product switching based on new patents. There are also references to positions taken by other Latin American countries such as Brazil (which subjects some patents to prior approval by regulators) or Argentina (which forbids the grant of several types of pharmaceutical patents).
These sections are followed by an explanation on past litigation activity on these topics, mentioning that it has been used as a deterrent to generic competition, and several recommendations, both of which will be analyzed in a subsequent post.
* Mr. Serrano is Of Counsel with ROMO DE VIVAR V.IP SERVICES, S.C.
[1] https://www.cofece.mx/cofece/index.php/prensa/historico-de-noticias/cofece-el-vencimiento-de-patentes-no-se-ha-traducido-en-suficiente-presion-competitiva-en-el-mercado. The press statement also contains the link to download the full study.
-
By Kevin E. Noonan –
Human history is often something modern man only sees as through a glass, darkly. This is particularly the case when that history did not occur in the Mediterranean, the Nile Valley, India, or China, or when there is no written record on which scholars can rely. Exacerbating the disrupting effects of time on history can be when that history occurs in a region where extensive migration has disrupted whatever temporarily stable civilization happened to have taken root at that place at any particular time.
But humans leave traces of themselves in their history and a variety of such traces have been the source of reconstructions outside conventional sources. Luigi Cavalli-Sforza began the study of human population genetics as a way to understand this history in 1971 in The Genetics of Human Populations, and later extended these studies to include language and how it influences gene flow between human populations. More recent efforts to use genetics to reconstruct history include Deep Ancestry: The Landmark DNA Quest to Decipher Our Distant Past by Spencer Wells (National Geographic: 2006), and The Seven Daughters of Eve: The Science that Reveals our Genetic Ancestry by Brian Sykes (Carrol & Graf: 2002). And even more recently, genetic studies have illuminated the "fine structure" of human populations in England (see "Fine-structure Genetic Mapping of Human Population in Britain").
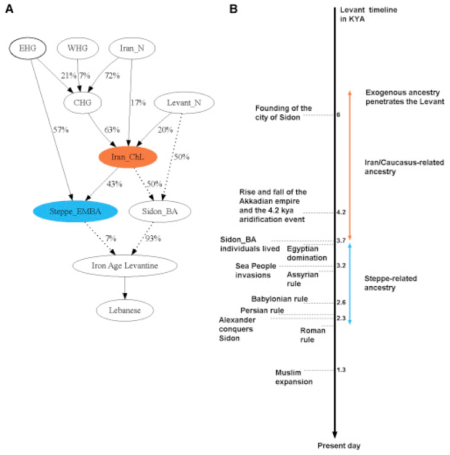
Two recent reports illustrate how genetics can inform history: the first, in the American Journal of Human Genetics entitled "Continuity and Admixture in the Last Five Millennia of Levantine History from Ancient Canaanite and Present-Day Lebanese Genome Sequences"; and a second in the Proceedings of the National Academy of Sciences USA, entitled "Genomic landscape of human diversity across Madagascar." In the first study, authors* from The Wellcome Trust Sanger Institute, University of Cambridge, University of Zurich, University of Otago, Bournemouth University, Lebanese American University, and Harvard University found evidence of genetic admixture over 5,000 years of a Canaanite population that has persisted in Lebanese populations into the modern era. This population is interesting for historians in view of the central location of the ancestral home of the Canaanites, the Levant, in the Fertile Crescent that ran from Egypt through Mesopotamia. The Canaanites also inhabited the Levant during the Bronze Age and provide a critical link between the Neolithic transition from hunter gatherer societies to agriculture. This group (known to the ancient Greeks as the Phoenicians) is also a link to the great early societies recognized through their historical writings and civilizations (including the Egyptians, Assyrians, Babylonians, Persians, Greeks, and Romans); if the Canaanites had any such texts or other writings they have not survived. In addition, the type of genetic analyses that have been done for European populations has not been done for descendants of inhabitants of the Levant from this historical period. This paper uses genetic comparisons between 99 modern day residents of Lebanon (specifically, from Sidon and the Lebanese interior) and ancient DNA (aDNA) from ~3,700 year old genomes from petrous bone of individuals interred in gravesites in Sidon. For aDNA, these analyses yielded 0.4-2.3-fold genomic DNA coverage and 53-264-fold mitochondrial DNA coverage, and also compared Y chromosome sequences with present-day Lebanese, two Canaanite males and samples from the 1000 Genomes Project. Over one million single nucleotide polymorphisms (SNPs) were used for comparison.
These results indicated that the Canaanite ancestry was an admixture of local Neolithic populations and migrants from Chalcolithic (Copper Age) Iran. The authors estimate from these linkage disequilibrium studies that this admixture occurred between 6,600 and 3,550 years ago, a date that is consistent with recorded mass migrations in the region during that time. Perhaps surprisingly, their results also show that the majority of the present-day Lebanese population has inherited most of their genomic DNA from these Canaanite ancestors. These researchers also found traces of Eurasian ancestry consistent with conquests by outside populations during the period from 3,750-2,170 years ago, as well as the expansion of Phoenician maritime trade network that extended during historical time to the Iberian Peninsula.
The second paper arose from genetic studies of an Asian/African admixture population on Mozambique. This group** from the University of Toulouse, INSERM, the University of Bordeaux, University of Indonesia, the Max Plank Institute for Evolutionary Anthropology, Institut genomique, Centre Nacional de Genotypage, University of Melbourne, and the Universite de la Rochelle, showed geographic stratification between ancestral African (mostly Bantu) and Asian (Austronesean) ancestors. Cultural, historical, linguistic, ethnographic, archeological, and genetic studies supports the conclusion that Madagascar residents have traits from both populations but the effects of settlement history are termed "contentious" by these authors. Various competing putative "founder" populations (including Arabic, Indian, Papuan, and/or Jewish populations as well as first settlers found only in legend, under names like "Vazimba," "Kimosy," and "Gola") have been posited as initial settlers. These researchers report an attempt to illuminate the ancestry of the Malagasy by a study of human genetics.These results showed common Bantu and Austronesian descent for the population with what the authors termed "limited" paternal contributions from Europe and Middle Eastern populations. The admixture of African and Austronesian populations occurred "recently" (i.e., over the past millennium) but was gender-biased and heterogeneous, which reflected for these researchers independent colonization by the two groups. The results also indicated that detectable genetic structure can be imposed on human populations over a relatively brief time (~ a few centuries).
Using a "grid-based approach" the researchers performed a high-resolution genetic diversity study that included maternal and paternal lineages as well as genome-wide data from 257 villages and over 2,700 Malagasy individuals. Maternal inheritance patterns were interrogated using mitochondrial DNA and patterns of paternity assayed using Y chromosomal sequences. Non-gender specific relationships were assessed through 2.5 million SNPs. Mitochondrial DNA analyses showed maternal inheritance from either African or East Asian origins (with one unique Madagascar variant termed M23) in roughly equal amounts, with no evidence of maternal gene flow from Europe or the Middle East. The M23 variant shows evidence of recent (within 900-1500 years) origin. Y chromosomal sequences, in contrast are much more prevalent from African origins (70.7% Africa:20.7% East Asia); the authors hypothesize that the remainder may reflect Muslim influences, with evidence of but little European ancestry.
Admixture assessments support Southeast Asian (Indonesian) and East African source populations for the Malagasy admixture. These results provide the frequency of the African component to be ~59%, the Asian component frequency to be ~37%, and the Western European component to have a frequency of about 4% (albeit with considerable variation, e.g., African ancestry can range from ~26% to almost 93%). Similar results were obtained when the frequency of chromosomal fragments shared with other populations were compared to the Malagasy population (finding the closest link to Asian populations from south Borneo, and excluding Indian, Somali, and Ethiopian populations, although the analysis was sensitive in one individual to detect French Basque ancestry). The split with ancestral Asian populations either occurred ~2,500 years ago or by slower divergence between ~2,000-3,000 years ago, while divergence with Bantu populations occurred more recently (~1,500 years ago).
There were also significant differences in geographic distribution between descendants of these ancestral populations. Maternal African lineages were found predominantly in north Madagascar, with material Asian lineages found in central and southern Madagascar (from mtDNA analyses). Paternal lineages were generally much lower overall for Asian descendants (~30% in central Madagascar) based on Y chromosome analyses. Genome-wide analyses showed "highlanders" had predominantly Asian ancestry (~65%) while coastal inhabitants had predominantly (~65%) African ancestry; these results depended greatly on the method of performing the analyses which affected the granularity of the geographic correlates. Finally, assessing admixture patterns indicated that the genetic results are consistent with single intermixing event (500-900 years ago) for all but one geographic area, which may have seen a first event 28 generations ago and a second one only 4 generations ago. These researchers also found evidence of at least one population bottleneck, where the number of individuals dropped to a few hundred people about 1,000-800 years ago.
These results are represented pictorially in the paper:

In view of the current political climate, the eloquent opening of the paper deserves attention:Ancient long-distance voyaging between continents stimulates the imagination, raises questions about the circumstances surrounding such voyages, and reminds us that globalization is not a recent phenomenon. Moreover, populations which thereby come into contact can exchange genes, goods, ideas and technologies.
* Marc Haber, Claude Doumet-Serhal, Christiana Scheib, Yali Xue, Petr Danecek, Massimo Mezzavilla, Sonia Youhanna, Rui Martiniano, Javier Prado-Martinez, Michał Szpak, Elizabeth Matisoo-Smith, Holger Schutkowski, Richard Mikulski, Pierre Zalloua, Toomas Kivisild, Chris Tyler-Smith
** Denis Pierrona, Margit Heiskea, Harilanto Razafindrazakaa, Ignace Rakotob, Nelly Rabetokotanyb, Bodo Ravololomangab, Lucien M.-A. Rakotozafyb, Mireille Mialy Rakotomalalab, Michel Razafiarivonyb, Bako Rasoarifetrab, Miakabola Andriamampianina Raharijesyb, Lolona Razafindralambob, Ramilisoninab, Fulgence Fanonyb, Sendra Lejamblec, Olivier Thomasc, Ahmed Mohamed Abdallahc, Christophe Rocherc,, Amal Arachichec, Laure Tonasoa, Veronica Pereda-lotha, Stéphanie Schiavinatoa, Nicolas Brucatoa, Francois-Xavier Ricauta, Pradiptajati Kusumaa,d,e, Herawati Sudoyod,e, Shengyu Nif, Anne Bolandg, Jean-Francois Deleuzeg, Philippe Beaujardh, Philippe Grangei, Sander Adelaarj, Mark Stonekingf, Jean-Aimé Rakotoarisoab, Chantal Radimilahy, and Thierry Letelliera
