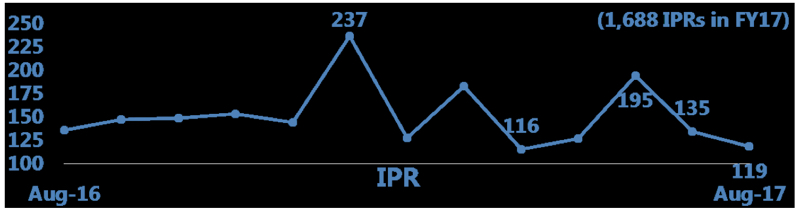
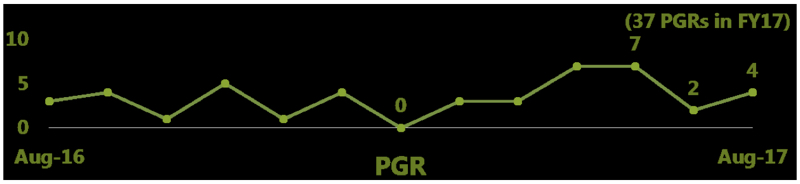
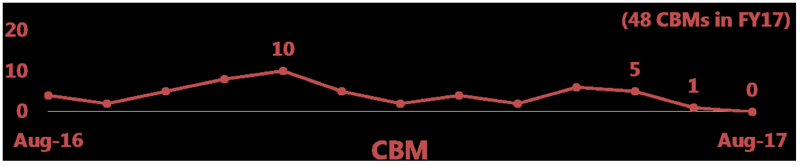
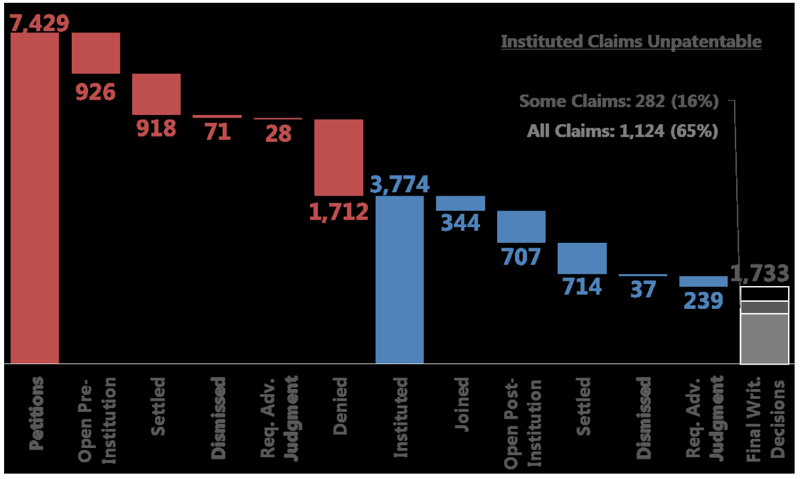
By Andrew Williams —
 Last Spring, the Supreme Court in TC Heartland LLC v. Kraft Foods Group Brands LLC held that the word "resides" in the patent venue statute, 28 U.S.C. § 1400(b), "refers only to the State of incorporation" of the alleged infringer. Correspondingly, the first prong of the statute is limited as to where a patent owner can bring an infringement suit. However, TC Heartland left open the question as to where an alleged infringer could be sued pursuant to the second prong of § 1400(b). The statute reads in its entirety:
Last Spring, the Supreme Court in TC Heartland LLC v. Kraft Foods Group Brands LLC held that the word "resides" in the patent venue statute, 28 U.S.C. § 1400(b), "refers only to the State of incorporation" of the alleged infringer. Correspondingly, the first prong of the statute is limited as to where a patent owner can bring an infringement suit. However, TC Heartland left open the question as to where an alleged infringer could be sued pursuant to the second prong of § 1400(b). The statute reads in its entirety:
Any civil action for patent infringement may be brought in the judicial district where the defendant resides, or where the defendant has committed acts of infringement and has a regular and established place of business.
However, in ANDA litigation, as well as lawsuits filed pursuant to the BPCIA, the act of infringement is artificial and the questions that a court must address are prospective in nature. Therefore, it is also an open question as to what it means for an FDA applicant to have committed an act of infringement as that term is used in 1400(b)? On September 11, 2017, in the Bristol-Myers Squibb Company v. Mylan case, Chief Judge Stark of the District of Delaware set out his framework for addressing venue challenges post-TC Heartland. Interestingly (but only coincidentally), it was ultimately Judge Stark that was reversed by the Supreme Court resulting from his adoption of the recommendation of Magistrate Judge Burke to deny TC Heartland's motion to dismiss on venue grounds.
This case arose when Mylan Pharmaceuticals Inc. ("Mylan") submitted an ANDA to market a generic version of BMS' Eliquis®, 2.5 mg and 5 mg strength apixaban tablets. BMS filed suit in the District of Delaware asserting U.S. Patent Nos. 6,967,208 and 9,326,945, generally directed to chemical compounds and formulations of apixaban. On July 25, 2017, after TC Heartland was decided, Mylan moved to dismiss the case for improper venue under Federal Rule of Civil Procedure 12(b)(3). The Court heard oral arguments in a hearing that concerned this case and other similar post-TC Heartland venue motions with both Chief Judge Stark and Magistrate Judge Burke presiding. In denying Mylan's motion without prejudice, Judge Stark analyzed what the second prong of § 1400(b) requires in the Hatch-Waxman context.
Who Bears the Burden of Proof?
The first issue addressed by the Court was who bears the burden in motions to dismiss for improper venue. The Court acknowledged that the majority of courts that have considered this issue have determined that it is the Plaintiff that must establish that the chosen district is a proper venue. However, the Third Circuit, in which Delaware is situated, has held that it is the defendant as the moving party that has the burden. The Federal Circuit, for its part, has not addressed this issue.
The Court first addressed whether Federal Circuit case law would control the case. To do this, the Court first determined whether this issue is one unique to patent law. According to Federal Circuit precedent, the procedural aspects of 12(b)(6) and judgments as a matter of law are controlled by the laws of the regional circuits. Because venue challenges also occur by operation of the Federal Rules of Civil Procedure, the Court concluded that improper venue motions are procedural, and controlled by Third Circuit law. Even so, the substantive questions relating to § 1400(b) are controlled by how the Federal Circuit would apply the second prong of this statute. Correspondingly, the Court determined that it was Mylan that had the burden of proof in establishing that venue was not proper.
Acts of Infringement in the ANDA Context
The next issue addressed by the Court was what would be considered an "act of infringement" such that venue would be proper. The Court first noted that the second prong of §1400(b) is written in the present perfect tense — "where the defendant has committed acts of infringement." The problem is that the temporal focus of a Hatch-Waxman infringement analysis is in the future, not the past or present. In other words, the question is what will the defendant manufacture and sell in the future, not what has been sold or is being sold. In footnote 9, the Court analogized the forward-looking nature of these cases to those of declaratory judgment actions. The prospective nature of the analysis is necessitated in part because there is no infringement liability for activities related to the submission of the application to the FDA because of the safe harbor provisions of 35 U.S.C. § 271(e)(1).
As a result, the "highly artificial act of infringement" in ANDA cases is the submission of application. Nevertheless, the resulting litigation is not about whether the documents submitted to the FDA were somehow unlawful, but rather whether a patent will be infringed by the manufacture, use or sale of the drug once the application is approved. Correspondingly, the Court concluded that in this context, "the 'acts of infringement' an ANDA filer 'has committed' includes all of the acts that would constitute ordinary patent infringement if, upon FDA approval, the generic drug product is launched into the market."
In reaching this decision, the Court relied heavily on the Federal Circuit's decision in Acorda Therapeutics Inc. v. Mylan Pharm. Inc., 817 F.3d 755 (Fed. Cir. 2016). The Acorda decision related to analyzing personal jurisdiction in the ANDA context, but the District Court reasoned that it was the best guidance as to how the Federal Circuit was likely to resolve these questions in the venue context. In that case, the Federal Circuit rejected the contention that the state must wait until conduct has occurred before exercising its judicial power: "[A]s long as the connection to the planned acts is close enough, the subject of such actions readily fits the terms of the minimum-contacts standard — conduct purposefully directed at the State that gives rise and is related to the suit." Similarly, the Court concluded that "the most reasonable conclusion after Acorda is that an ANDA filer's future, intended acts must be included as part of the 'acts of infringement' analysis for purposes of determining if venue is proper under the patent venue statute." The Court also noted that this reasoning stems from the fact that in the Hatch-Waxman context, the applicant has "reliably confirmed a plan to engage in real-world marketing" by the filing of the application. In fact, the Court pointed out that there would likely never be an act of actual infringement because either the patents will be found to be invalid or not infringed during the subsequent litigation, or the ANDA filer will be kept off the market during the life of the patents. And in the situation where the ANDA filer launches at risk, the pleadings will be amended to add claims of infringement under 35 U.S.C. § 271(a)-(c).
In reaching this conclusion, the Court specifically rejected the argument that the act of infringement either occurs at the location where the application submission is made (in this case, the home of the FDA, Bethesda, Maryland), where the submission is made from (either whether it is mailed or where it is electronically submitted), or where the center of gravity of the work associated with the preparation and submission occurred. The Court did acknowledge the potential problems with its interpretation. First, the verb tense of the statutory language is inconsistent with a prospective analysis. As a result, the term "act of infringement" would mean something different in Hatch-Waxman cases than in the non-Hatch-Waxman context. Also, the Supreme Court had previously warned against giving 1400(b) a liberal construction. Nevertheless, the Court reasoned that any other conclusion would be inconsistent with Acorda and the realities of Hatch-Waxman litigation.
The Court also provided a policy reason for its conclusion. Any contrary reading of § 1400 (b) would frustrate the goals of the expeditious resolution of the patent issues inherent in the Hatch-Waxman statute. If courts were required to address venue-related motions to dismiss in every case, it would be difficult for them to resolve the suits during statutory 30-month stay. Nevertheless, the Court recognized that such policy considerations were basically irrelevant to the question of statutory interpretation.
In applying this newly articulated standard, the Court found there was an act of infringement in this case. Specifically, Mylan did not deny that it planned to sell its product within Delaware. And the Court has already determined the fact that the ANDA was prepared in West Virginia and electronically submitted to the FDA located in Maryland was irrelevant to the present motion.
Regular and Established Place of Business
The Court next turned to what it means to have a regular and established place of business. First, the statute was parsed to reveal that what is required to meet this prong is that there be a (i) place of business that is (ii) regular and (iii) established. The Court focused on In re Cordis, 769 F.2d 733 (Fed. Cir. 1985), as the "most recent, precedential case applying" the second part of § 1400(b), even though it recognized that the Cordis decision was based on a mandamus petition, which utilizes a more deferential standard. The Federal Circuit in Cordis held that "in determining whether a corporate defendant has a regular and established place of business in a district, the appropriate inquiry is whether the corporate defendant does its business in that district through a permanent and continuous presence there and not . . . whether it has a fixed physical presence in the sense of a formal office or store." Nevertheless, the Delaware Court noted that there still needs to be a "place of business," which means the defendant needs to have a physical presence is district. As a result, there needs to be "some sort of meaningful physical manifestation in the district," an inquiry which is a factually driven.
The Court next cataloged the activities that have been determined, in and of themselves, to not be sufficient to amount to a regular and established place of business. These include: (1) simply doing business or being registered to do business in a district, (2) having sufficient "minimum contacts" with the district for the purposes of personal jurisdiction, (3) maintaining a website accessible within the district, and (4) shipping goods into a district, whether to an individual or for distribution by third parties. Accordingly, the Court concluded that it "must determine whether a defendant has a regular and established place of business by conducting a fact-intensive inquiry focused on whether the defendant does its business in this District through a permanent and continuous presence here."
Applying this test, the Court could not establish that Mylan has a regular and established place of business in Delaware. In the event that Mylan renewed its motion, the Court did provide BMS with an opportunity to take venue-related discovery. Along these lines, the Court did note the following facts: (1) Mylan has a nationwide and global footprint, (2) Mylan "'leverage[s] a broad network of local and global access channels that include physicians, institutions, governments, retailers and wholesalers," (3) the Mylan family includes at least 55 U.S. subsidiaries, more than 40 of which are incorporated in Delaware, and (4) Mylan has admitted that it does business in Delaware and that its products have been sold in the district. In addition, the Court noted that Mylan is a frequent litigant in Delaware federal court. For example, Mylan has been involved in more than 100 cases in the past ten years. In fact, Mylan recently got a Hatch-Waxman Act case transferred from the Northern District of West Virginia (its place of incorporation) to Delaware. Finally, there has been at least one Mylan case pending in Delaware in the past ten years. These facts are relevant because they demonstrate that Mylan's business model is predicated on participating in large amounts of litigation.
On the other hand, Mylan had alleged that it does not own or lease any manufacturing plants, corporate offices, facilities, or other real property in Delaware. Also, it does not have telephone listings or mailing addresses in Delaware. In addition, it does not have any employees working in Delaware. Nevertheless, the Court found that Mylan was unable to meet its burden, and so it could not say that Mylan "does not have a regular and established place of business in Delaware." Correspondingly, the motion to dismiss was denied without prejudice.
Do the Acts of Infringement and the Regular and Established Place of Business Need to be Related?
The Court next addressed the issue whether there must be a relationship between the accused acts of infringement and the regular and established place of business. The Court noted that other district has concluded that the two must be related. However, it came to the opposite conclusion. It relied on the statutory language to support this conclusion. For example, the statute does not require that the infringement "arise from" the regular and established place of business. The Court did recognize that there is language in Cordis that suggested a relationship must exist. Nevertheless, it concluded that the Federal Circuit was not actually confronted with this specific question, so that language amounted to more than dicta.
Venue-related discovery
Finally, because the Court dismissed the case without prejudice, it determined that granting BMS venue-related discovery was appropriate. It explained that "discovery will include understanding the relationship among the 40 Delaware Mylan entities" and the Mylan entities involved in the present suit. It also noted that discovery would be allowed to determine whether Mylan has sales representatives that come to Delaware to meet with doctors and hospitals. In addition, it suggested it would allow discovery to help understand how the industry operates, and the way sales are made and marketing is done. BMS may also allowed to explore Mylan's relationship with wholesalers. Finally, the Court noted that BMS may take discovery to determine Mylan's relationship with pharmacies and physician in Delaware. The Court concluded by allowing the case to proceed on the merits.
As a reminder, readers interested in this case and other related issues should consider attending American Conference Institute's 5th Annual Paragraph IV Disputes master symposium on October 2-3, 2017 in Chicago, IL. This author will also be in attendance, so if you are attending, please say hello. As a reminder, Patent Docs is a media partner for this event, and its readers are entitled to a 10% discount off of registration using discount code P10-999-PTD18.
 Strafford will be offering a webcast on "Negotiating IP Rights in Industry Sponsored Research Agreements — Structuring Ownership, Licensing, Assignment, Confidentiality, Publication and Use Provisions" on September 28, 2017 from 1:00 to 2:30 pm (EDT). Dr. Robert H. Underwood of McDermott Will & Emery and Matthew R. Wilmot of Stoel Rives will provide guidance to counsel on negotiating and structuring industry sponsored research agreements (SRAs) to allocate IP ownership, outline challenges of SRAs with nonprofit organizations, and discuss key provisions of the SRA to protect IP rights and avoid unintended consequences. The webinar will review the following topics:
Strafford will be offering a webcast on "Negotiating IP Rights in Industry Sponsored Research Agreements — Structuring Ownership, Licensing, Assignment, Confidentiality, Publication and Use Provisions" on September 28, 2017 from 1:00 to 2:30 pm (EDT). Dr. Robert H. Underwood of McDermott Will & Emery and Matthew R. Wilmot of Stoel Rives will provide guidance to counsel on negotiating and structuring industry sponsored research agreements (SRAs) to allocate IP ownership, outline challenges of SRAs with nonprofit organizations, and discuss key provisions of the SRA to protect IP rights and avoid unintended consequences. The webinar will review the following topics: