Patent Law Weblog
recent posts
- Constellation Designs, LLC v. LG Electronics Inc. (Fed. Cir. 2026)
- Actelion Pharmaceuticals Ltd. v. Mylan Pharmaceuticals Inc. (Fed. Cir. 2026)
- PTAB Reverses § 101 Rejection Where Examiner Failed to Follow the Office’s Own Guidance
- Collision Communications v. Samsung: What Good Did the Government’s Statement of Interest Do?
- United States Files Statement of Interest in Patent Infringement Proceedings
about
-
By Kevin E. Noonan –
In what was an otherwise run-of-the-mill affirmance of a decision by the Patent Trial and Appeal Board (PTAB) (albeit somewhat noteworthy in affirming the Board's determination that the challenged claims were not invalid), the Federal Circuit heard but deigned not to consider the question of whether claims to methods for achieving a therapeutic outcome needed to be supported by a showing of actual reduction to practice (i.e., that the method had been shown to be operative for the claimed result) in Medtronic, Inc. v. Teleflex Life Sciences Ltd. (precedential).
The case arose in two inter partes review decisions over a reissue patent U.S. Patent No. RE46,116 directed to methods for "using a guide extension catheter with a guide catheter." A portion of claim 25 of the '116 patent was reproduced in the opinion as being representative:
25. A method, comprising:
advancing a distal end of a guide catheter having a lumen through a main blood vessel to an ostium of a coronary artery; . . .The original patent was filed in 2006 and the Board's assessment of the sufficiency of Medtronic's validity challenge was carried out under the provisions of the 1952 Patent Act. Accordingly, the Board considered evidence of conception in 2005 and diligence from its conception date until filing a patent application in May 2006 (constituting constructive reduction to practice). Medtronic asserted U.S. Patent No. 7,604,612 against claims 52 and 53 of the '116 patent for anticipation (the Board held for Medtronic and this decision was not appealed); asserted the combination of the '612 patent and U.S. Patent No. 7,736,355 for obviousness of claims 25-40, 42, 44-48, 52 and 53; and asserted the '612 and '355 patents in combination with U.S. Patent Application Publication No. 2005/0015073 for obviousness of claim 45 (wherein in each case in which it was asserted Medtronic argued the '355 patent was prior art under 35 U.S.C. § 102(e)).* The opinion notes that Medtronic did not challenge the sufficiency of Teleflex's evidence for conception prior to the critical date of the '355 patent (September 21, 2005). With regard to the '355 patent, the Board held that Teleflex had reduced to practice the invention claimed in the remaining challenged claims prior to the filing date of the ‘355 patent and had also satisfied the diligence requirements for its May 2006 filing date to qualify as a constructive reduction to practice. This decision, the opinion notes, was consistent with the Board's earlier decision on whether the '355 patent qualified as prior art for claims to Teleflex's corresponding apparatus claims.
The distinction ("unique to this case") that raised one of Medtronic's arguments in this appeal was the question of "whether or not in vivo testing was required for actual reduction to practice" because here the challenged claims recited the step of "advancing . . . a guide catheter . . . through a main blood vessel to an ostium of a coronary artery." The Board held such evidence was not necessary and that evidence from alternative physical models could suffice. This appeal followed.
The Federal Circuit affirmed, in an opinion by Judge Lourie, joined by Judges Prost and Chen. The sole issues raised in this opinion were "(1) whether or not in vivo testing was required for actual reduction to practice and (2) whether or not the patentee exercised reasonably continuous diligence until constructive reduction to practice," wherein the panel determined that Teleflex had established constructive reduction to practice and did not reach the issue of actual reduction to practice (noting that a showing of either species of reduction to practice was enough to affirm the Board's opinion). On this question, the Federal Circuit held that Medtronic had waived any challenge regarding the diligence issue by attempting to incorporate its arguments in that regard by reference to the Federal Circuit's judgment in a separate, related IPR. The panel rejected this attempt, first, because in that related case the Court had not vacated the Board's decision on sufficiency of asserted diligence, and second, that trying to "incorporate by reference twenty pages from another brief in another case, amounting to over 4,000 extra words" was "a clear violation of both the motions panel's order [denying Medtronic's motion for leave to expand its brief to 20,000 words] and our rules" (emphasis in opinion). The panel characterized this attempt as a strategic decision by Medtronic not to include arguments directed at the diligence issue into its principle brief and, its attempt having failed held that Medtronic's challenge of the Board's finding of diligence had been waived. Combined with Medtronic's decision not to contest conception of the claimed invention the panel affirmed the Board's decision that the '355 patent was not prior art and that Medtronic's obviousness challenges failed.
*Medtronic asserted unsuccessfully these references as well as U.S. Patent Application Publication No. 2007/0260219 in a second IPR against the '116 patent, but Federal Circuit decisions in three related cases rendered moot Medtronic's appeal of the Board's decisions in this IPR, which was not further discussed in this opinion.
Medtronic, Inc. v. Teleflex Life Sciences Ltd. (Fed. Cir. 2023)
Panel: Circuit Judges Lourie, Prost, and Chen
Opinion by Circuit Judge Lourie -
By Michael Borella —
We are at the beginning of what promises to be a wave (potentially a tsunami) of complaints filed against the companies behind generative AI models (e.g., OpenAI). Recent lawsuits from Paul Tremblay and Mona Awad (Tremblay and Awad v. OpenAI Inc. et al. — Northern District of California, No. 3:23-cv-03223), Sarah Silverman (Silverman v. OpenAI, Inc. — Northern District of California, No. 3:23-cv-03416-AMO), and the Authors Guild (Authors Guild et al v. OpenAI Inc. et al. — Southern District of New York, No. 1:23-cv-8292)[1] contend that OpenAI and others have hoovered up thousands of copyrighted publications, including those of the named plaintiffs, and used them to train large language models (LLMs) such as GPT-4. As these initial cases proceed, and possibly go up on appeal, they are likely to define the contours of how copyright law applies to the new world of generative AI and whether is it proper to train such models on copyrighted works without permission to do so.
The authors' theories of infringement vary as do their ancillary claims. While acknowledging the risk of over-simplify complex issues, we can boil the merits of these cases down to two main questions:
1) Is the ingestion of a copyrighted work into the training process of an LLM without the author's permission an infringement of the copyright?
2) What if an LLM trained in this fashion produces a new work that is substantially similar to the copyrighted work?

These questions can be thought in terms of pigs and sausage.[2] Pigs can be turned into sausage, but it is generally accepted to be impossible to turn sausage back into a pig. Mathematicians would consider the transformation from pig to sausage to be an irreversible one-way function.It is important to understand that all computer data are just organized collections of numbers. This includes digital copies of books, images, audio, video, web sites, etc. When a machine learning model such as an LLM is trained on a digital book, the arrangement of numbers representing the words, punctuation, front matter, and so on are transformed into a different arrangement of numbers — weights in a complex set of neural networks.
In most cases, there is no one-to-one mapping between the numbers used before and after transformation. One cannot point to a particular set of numbers in an LLM and identify a Game of Thrones novel. Indeed, the weights in an LLM are a complex amalgam of most or all data on which it was trained. Even the entities that design and build LLMs have yet to provide an understanding of what the weights actually represent.
So this leads to a likely answer to the first question. A similar set of facts were considered by the Second Circuit in Authors Guild, Inc. v. Google, Inc., in the context of using copyrighted books for search purposes. The Court ultimately ruled that the conversion of the copyrighted content into a form useful for searching was highly transformative, displaying small portions of the books was fair use, and such search and display did not provide a significant market substitute for the original works. Therefore, the mere use of a copyrighted work to train an LLM, even without permission, is unlikely to be a winning fact pattern.
But the emergent magic of LLMs is that they might know enough about an ingested Games of Thrones novel to be able to produce its plot summary, a list of main characters, and even quote a section or two.[3] These uses might also fall under the Second Circuit's definition of fair use.
But an LLM may be able to produce significant portions of the work or the work as a whole.[4] Or, the LLM may be able to generate alternative endings to the novel, new works in the style of the author, or new works involving the same characters and relying on the authors' world-building.
Thus, the answer to the second question is not clear, though it seems that the LLM would have to provide "more than just a little" of the copyrighted work. For example, copyright famously protects actual works and not styles. This issue may boil down to whether an LLM can reverse the transformation function and turn sausage back into a reasonable semblance of a pig, as well as whether an LLM operator can successfully prevent it from doing so.
As noted, the cases currently being litigated may provide some clarity — or, depending on how they proceed, maybe not. Also, Congress may step in and define new causes of action that specifically target LLMs and similar fact patterns.
Authors may ultimately have their strongest positions where they can argue that the operator of the LLM is unjustly enriching itself on the backs of the authors' labor or effectively competing in the same marketplace as the authors. At first blush it seems that imaging tools based on generative AI (e.g., DALL-E, Midjourney, and others), the use of which can eliminate the need for human illustrators, might be a better target for such claims.
[1] Here, the group of authors named in the complaint include John Grisham, George R. R. Martin, Jodi Picoult, and Scott Turow.
[2] Vegans should feel free to replace "pigs" with "plant-based protein."
[3] OpenAI appears to be aware of the issues that this capability might raise. If you ask ChatGPT 4 to "provide a Jon Snow quote from Game of Thrones," it falls back on a Bing search to do so.
[4] This is theoretically possible, though OpenAI and others have put guardrails in place in attempts to prevent their models from such blatant infringement.
-
By Kevin E. Noonan –
 It came as something of a shock in May, 2021 when the Biden administration voiced support for waiver of intellectual property protection mandated under the TRIPS agreement for COVID-19 vaccines (see "Biden Administration Supports Waiver of IP Protection for COVID-19 Vaccines"). The waiver had originally been proposed to the World Trade Organization (WTO) by India and South Africa; in that incarnation a much broader swath of intellectual property protections had been proposed (encompassing diagnostics and therapeutic drugs related to the pandemic). When eventually imposed under the WTO's June 17, 2022 Ministerial Decision, the waiver was limited to vaccines with further consideration of waivers having broader scope postponed (until later this year, in fact).
It came as something of a shock in May, 2021 when the Biden administration voiced support for waiver of intellectual property protection mandated under the TRIPS agreement for COVID-19 vaccines (see "Biden Administration Supports Waiver of IP Protection for COVID-19 Vaccines"). The waiver had originally been proposed to the World Trade Organization (WTO) by India and South Africa; in that incarnation a much broader swath of intellectual property protections had been proposed (encompassing diagnostics and therapeutic drugs related to the pandemic). When eventually imposed under the WTO's June 17, 2022 Ministerial Decision, the waiver was limited to vaccines with further consideration of waivers having broader scope postponed (until later this year, in fact).In anticipation of negotiations over expanding the IP waiver, the U.S. Trade Representative, Ambassador Katherine Tai sent a letter on December 16, 2022 to the International Trade Commission requesting the ITC to prepare a Report under section 332(g) of the Tariff Act of 1930 on the needs and justification for further expansion of the IP waiver. The Trade Representative asked that the ITC's Report be a product of market and other research within the Commission's expertise and also reflect the views of stakeholders and the public, to be garnered from public hearings and solicited comments from "foreign governments, non- governmental health advocates, organizations such as MPP and Foundation for Innovative New Diagnostics(FIND), and diagnostic and therapeutic manufacturers." The letter expressly disclaims any request for the Report to "draw any policy conclusions" and asked for the Report to be provided no later than October 17, 2023.
The ITC (Chairman David S. Johanson, Rhonda K. Schmidtlein, Jason E. Kearns, Randolph J. Stayin, and Amy A. Karpel) issued its almost 500-page Report, entitled "COVID-19 Diagnostics and Therapeutics: Supply, Demand, and TRIPS Agreement Flexibilities," on the deadline date (Ambassador's Tai's December 16, 2022 letter can be found as Appendix A to the Report). Containing as a prelude a list of abbreviations and glossary (necessary and advantageous for a report from any government agency or commission), which also identifies various companies, organizations, agencies in the U.S. and abroad, regulations, and proposals, the Report provides a succinct Executive Summary. Contained in this Summary are the sentiments that the COVID-19 pandemic had caused almost 7 million deaths worldwide and that there was a possibility (espoused by some) that IP could "act as a barrier to access, particularly in developing countries." Several highlights of this summary are:
• The TRIPS Agreement sets minimum standards for the protection and enforcement of IP rights. It also includes flexibilities (although there were differences of opinion regarding the scope of those flexibilities).
• Determining a definitive scope of what products are covered by the terms "diagnostics" and "therapeutics" as they pertain to COVID-19 and what constitutes relevant COVID-19 diagnostics and therapeutics covered by patents is complicated and subject to interpretation.
• The development and commercialization of COVID-19 diagnostics and therapeutics occurred on an extremely compressed timeline. Manufacturing of diagnostics and therapeutics involves multiple stages, each of which requires careful attention to detail and strict quality control measures. The two fields of diagnostics and therapeutics are composed of different producers, inputs, know-how, and so on (recognizing the unique characteristics of COVID-19 and thus suggesting caution in applying this recent history to future pandemics, but the commonalities such as diagnostics being marketed sooner than therapeutics). Data points: As of summer 2023, China (UMIC) reportedly had the highest number of manufacturers of COVID-19 diagnostics (247), and India (LMIC) had the largest number of reported therapeutics manufacturers (56).
• A variety of advantages and challenges are associated with the use of voluntary licenses to provide access to IP associated with COVID-19 therapeutics and compulsory licenses to access COVID-19 therapeutics and other patented pharmaceutical products (wherein neither licensing type was used during the pandemic).
• The availability of supplies to meet global demand for COVID-19 diagnostics and therapeutics has been a moving target throughout the pandemic (market demand or need? Varied over the time course of the pandemic).
• The disparity among countries of different income groups is wide in terms of access and availability to COVID-19 diagnostics and therapeutics (regarding government procurements: 80% HIC; 14% UMICs, 5% LMICs, none by LICs but multilateral organizations help in LICs. Recognition that this is multivariable, depending on "access to IP, prices and affordability, regulatory approvals, healthcare infrastructure, and the healthcare priorities of governments").
• Academic literature on the effects of patent protection, compulsory licenses, and the MPP is limited and would benefit from additional research ("From the available evidence, patent protection is generally found to be more beneficial to innovation in the health sector for developed countries and less so for developing countries.")
The Executive Summary also sets forth both the source of the information considered by the Commission (from academic and governmental literature, public and private sources, more than 120 interviews with stakeholders internationally, public hearings held March 29-20, 2023, and over 300 written submissions) and gaps in that information, which vary by country and region ("greatly"), in particular with regard to production costs. Also noted were gaps in information regarding IP-related agreements, including compulsory licenses and the existence of "very little price transparency" (which raises the question of the factual basis for the purported of the necessity for the waiver in the first place). Nor was there a great deal of information on consumption of COVID-19 related diagnostic or therapeutic moieties outside high income countries.
The Executive Summary also contains a brief review of IP and the TRIPS agreement, including an even briefer history of WTO grant of compulsory licenses in limited instances for pharmaceutical products. This portion of the Report identifies IP protections for COVID-19 related diagnostics and therapeutics (and expiry of the foundation IP on diagnostic methods) ("the best evidence that patents did not act as a primary barrier to the production of these tests is that many new producers and products, including in developing countries, came online relatively quickly"). The results in the Report include a "broad and varied" (and indeterminate) universe of the diagnostics that include "(1) tests used to diagnose an active infection of the SARS-CoV-2 virus, (2) tests that measure an adaptive immune response to the virus, and (3) tests used in the management of patients with COVID-19" and ancillary products (such as swabs, etc.). Therapeutic entities likewise include the drugs and delivery vehicles (syringes, etc.) for administering them. This portion of the Summary is devoid of any recognition or discussion of vaccines, in view of the mandate from the Trade Representative and the existing vaccine waiver. The Report provides schematic representations of how diagnostics and therapeutics were characterized:

With regard to supply chain issues (which in some ways were even more critical post-pandemic), the Report recognizes the multinational, interdependent nature of the pharmaceutical industry (see Cott et al., "How Pfizer Makes Its Covid-19 Vaccine," New York Times, April 28, 2021), and the complexities created by this situation. As of June/July 2023, the Report estimates there were 900 diagnostics manufacturers and 221 therapeutics manufacturers.This Summary of the Report also addresses the source of COVID-related IP, which include R&D agreements, manufacturing partnerships, and licensing agreements (only R&D agreements were informative for diagnostics technology). While small- and medium-sized entities (SME) were responsible for development of new COVID therapeutics, larger pharmaceutical companies and the Federal government supplied economic and development support (and, according to the Report, was a source of calls for the resulting products and IP to be broadly and more cheaply available and those contracts be more transparent). Voluntary licenses provide one source of IP, which would disclose not only the patented invention but also technical information, know-how, and trade secrets the disclosure of which would undoubtedly raise resistance to this disclosure. While geographical restrictions are discussed, the Report also somewhat unrealistically posits royalty-free licenses as a norm. One advantage of such licenses noted in the Report was somewhat more fulsome disclosure than was obtained from compulsory licenses. Also mentioned with approval was disclosure though the Medicines Patent Pool ("a United Nations-backed public health organization"). So-called "upper-middle-income countries," defined by the World Bank as those economies with incomes per capita between $4,256 and $13,205, were not included in voluntary licenses for four COVID-19 therapeutics.
Turning to compulsory licenses, the Report notes that while the subject and provisions for and under which compulsory licenses for COVID-related IP was the subject of the 2022 Ministerial decision, no compulsory licenses to patents related to COVID-19 vaccines have been granted (although such licenses to IP relating to COVID-19 therapeutics have been granted in Hungary, Israel, and Russia). The Report briefly sets forth purported advantages and real challenges that arise with using compulsory licenses, including for advantages reduced price, development of domestic manufacturing capacity, and "leverage" for negotiating with rightsholders. The Report recognizes the limitation (for now) that compulsory licenses involve solely patents and do not grant access to know-how and trade secrets (which the Report notes can be particularly important for biologic drugs). The Report acknowledges that compulsory licenses can be granted in the context of lack of availability of a qualified manufacturer and result in economic and political pressure and legal challenges under domestic law or TRIPS (although the latter may be something of a paper tiger in the current political climate).
The Report particularly acknowledges issues in "least developed countries" (LDC) as classified by the United Nations. Bangladesh, Laos, Myanmar, and Paraguay are provided as exemplars for taking advantage of the exceptions to TRIPS requirements related to IP, which in the COVID context has been used as a pretext for producing generic versions of nirmatrelvir (+ ritonavir), remdesivir, and baricitinib and exporting these drugs to countries without patent protection for them.
With regard to the availability and consumption of COVID-19 diagnostics and therapeutics, the Report assesses both parameters through the lens of price, actual purchase, donations, and consumption of diagnostics and therapeutics, as illustrated in this schematic for therapeutics:

For diagnostics, the data reviewed (albeit subject to inconsistencies in different countries, from different manufacturers, and different times) indicate government procurement and distribution of test kits (in the U.S. pledged to provide $1 billion in kits free of charge). Multilateral organizations made tests broadly available to middle- and low-income countries during the pandemic, which have reduced substantially by mid-2023, as the pandemic seems to have waned and WHO has ended the public health emergency.For therapeutics, the Report found very little price transparency but notes a tiered-pricing regime depending on income level, with low-income countries paying substantially less. Except for high income countries like the U.S. and Europe COVID therapeutics are obtained/procured through donations or purchase from governments (79% of 98 million treatment courses) and multinational organizations. Pfizer, Eli Lilly, and Merck have donated COVID therapeutics to low- and middle-income countries. Consistent with these data the Report states that "many consider the prices of COVID-19 therapeutics to be a significant barrier to access for many" countries (other than high income countries, of course), illustrating the challenge of therapeutic cost with Pfizer's nirmatrelvir (+ ritonavir) treatment, which at $250 per treatment course is about half of the average per capita healthcare expenditure across all upper-middle income countries. Coupled with this disincentive the waning of the pandemic has caused a deep reduction in demand for these drugs. Even when demand was higher regulatory approvals in each country caused delays, and many countries have pre-existing public health demands (HIV/AIDS, malaria, and tuberculosis are given as examples) that limited availability of COVID-19 drugs.
The Report next sets forth synopses of the views of "interested persons" on "(1) IP protection, R&D, and jobs; (2) the TRIPS Agreement and access to medicine; and (3) the TRIPS Agreement and COVID-19 diagnostics and therapeutics" obtained for eight topics in each of these categories. Not unexpectedly, the Report asserts that while some respondents attested to the beneficial effect of IP on development of technologies critical to addressing COVID-19 and future pandemics, others expressed the belief that extending the 2022 Ministerial Decision would not have a deleterious effect on the supply of diagnostics or therapeutics. Likewise respondents could be split into those who believe the IP protections in TRIPS facilitated access to medicines (including COVID-related diagnostics and therapeutics by incentivizing investment) while others thought the purported increased cost had the opposite effect. The pros and cons of compulsory licensing were also addressed and some respondents asserted that further clarification on the "flexibilities" in IP protection would be a useful pathway towards improving access to medicines worldwide, particularly in low- and middle-income countries. Respondents were in agreement, however, that "waiving or weakening IP rights on COVID-19 diagnostics and therapeutics would reduce incentives to invest in the R&D of tests and treatments for future pandemics." Nevertheless, respondents split reliably on lines wherein some advocated that IP protection was necessary to be prepare for future pandemic (providing incentives and financial rewards for innovation) while others stated that the 2022 Ministerial Decision should be extended to encompass future drug development. The former respondent group warned that "an extension of the 2022 Ministerial Decision to COVID-19 diagnostics and therapeutics would cover an overly broad set of diagnostics and therapeutics and reduce incentives for companies to look for COVID-19 applications for their products." Respondents also submitted statistics on the effect of the Decision on employment in 26 countries (including the U.S.) but these statistics included jobs unrelated to COVID-19 diagnostics and therapeutics and thus were of limited value.
At the request of the Trade Representative the Report contains a "literature review" on four topics: innovation in the health sector and access to medicines in relation to patent protection; the effects of compulsory licensing; and the effects of the Medicines Patent Pool (this topic having received limited academic study that focused in the main on HIV/AIDS drugs). These reviews found little consensus in the current academic literature on these topics, the Report characterizing them as "limited" and that would "benefit from additional research on these topics," due in part to "limited data availability" and "difficulty identifying the causal effect" of IP on these topics.
The remainder of the Report comprises the following sections.
Chapter 1: Introduction: explaining that the Report was in response to a letter dated December 16, 2022 from the U.S. Trade Representative asking the ITC to perform the study in response, in part, to the June 17, 2022 Ministerial Decision providing a waiver of certain TRIPS protections for intellectual property in response to a joint communique from India and South Africa. The scope and intention of the December 2022 letter indicated that the ITC's Report would be used by the U.S. before the WTO in its deliberations regarding whether to extend the IP waiver to include COVID-related diagnostics and therapeutics. In making these determinations, economies for WTO members were set forth according to this schema:

(where HIC = high-income countries; UMIC = upper-middle-income countries; LMIC = lower-middle-income countries; LIC = low-income countries). This section provides cumulative information that as of August 2023 the COVID pandemic had produced 769 million confirmed cases and almost 7 million deaths.Chapter 2: Background on IP and Regulations: providing background information on TRIPS and mandated IP protections required thereby. Notably, this section discloses that the "flexibilities" regarding IP protection of TRIPS Articles 31 and 31bis and the Doha Declaration (2001) have resulted in "172 instances (122 compulsory licenses; 46 extended transition implementation period until 2033 for LDCs; 3 exceptions to patent rights under TRIPS Article 30; and 1 instance of parallel importation) in which governments used or attempted to use TRIPS Agreement flexibilities for public health purposes as of May 2023. And modifications to the Doha declaration in 2003 permitted compulsory licenses to extend to production for export as well as domestic applications. This Chapter also sets forth the particulars of the 2022 Ministerial Declaration (in particular mentioning that the original proposal included waiver of "undisclosed information" meaning trade secrets), in apparent contradiction to the provisions of TRIPS Article 39.3. Finally in this regard, this Chapter sets forth information from both those in favor and against the 2022 Ministerial Declaration that IP rights did little or nothing to inhibit diagnostic testing for COVID-19 during the pandemic, due to expiration of most applicable IP.
The Report also discloses landscape searches performed by WIPO and the USPTO (824 COVID-19 diagnostic-specific published patent filings at the USPTO between December 2019 and the end of March 2023, in which 47% of relevant patent families were filed in China) directed to diagnostics. On the other hand, WIPO reported 4,787 applications filed between January 2020 and September 2022 directed to COVID-19 therapeutics.
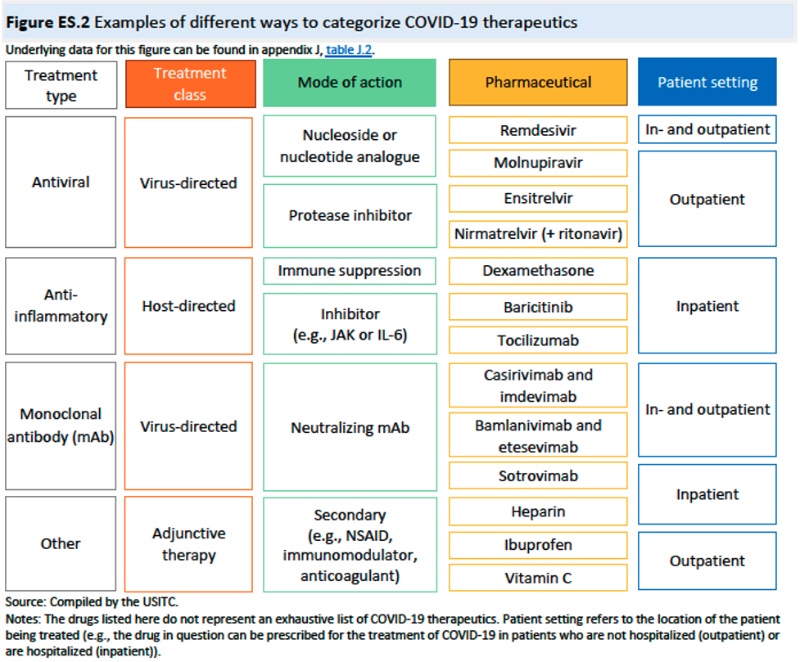
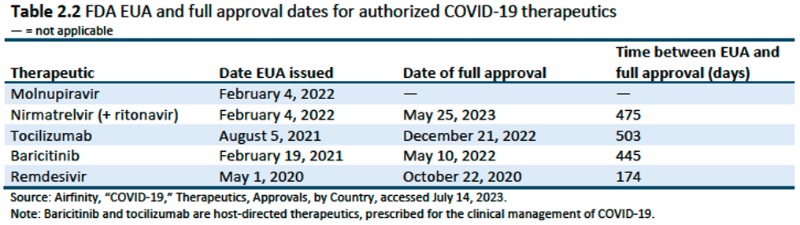
This Chapter concludes with an overview of the drug regulatory landscape worldwide (in each individual country and WHO) and the status of regulatory approval for COVID-19 related diagnostics and therapeutics. Five COVID therapeutics have been granted prequalification by WHO: tocilizumab, dexamethasone, molnupiravir, remdesivir, and nirmatrelvir (+ ritonavir), and FDA has authorized 278 molecular diagnostic tests and 64 antigen tests under EUAs as of July 2023.

Chapter 3: Definitions and the Universe or COVID-19 Diagnostics and Therapeutics (vaccines were not included in the Trade Representative's Request letter): specifically sets forth "the range of definitions for diagnostics and therapeutics in the medical field; (2) identifies and defines the universe of existing COVID-19 diagnostics and therapeutics covered by patents, as well as diagnostics and therapeutics in development; and (3) provides a broad overview of relevant COVID-19 diagnostics and therapeutics," acknowledging that these definitions are more complex than identifying vaccines, at least with regard to functional properties. These data are illustrated in several tables in the Chapter.

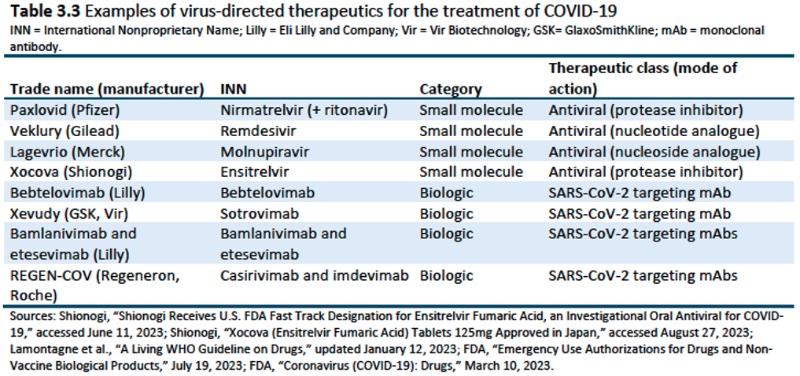

Chapter 4: COVID-19 Diagnostics and Therapeutics Manufacturing Supply Chain and Trade: provides information "describing the development, production, and trade of COVID-19 diagnostics and therapeutics; discussing the regulations needed to bring these goods to the global market; and providing an overview of the key producing countries and firms operating in the supply chain." Diagnostics were limited to PCR and rapid antigen tests, while therapeutics were limited to virus-directed drugs on the market during the pandemic (January 2020 to July 2023), including both small molecule and biologic drugs. The Report acknowledges the limitations in the data for a variety of reasons.Chapter 5: Approaches to Access the Intellectual Property Associated with COVID-19 Diagnostics and Therapeutics: This Chapter is focused more on therapeutics than diagnostics and addresses ways to accesses COVID-related IP, including "research and development (R&D) agreements, manufacturing partnerships, bilateral license agreements (BLAs) between firms, and license agreements involving the Medicines Patent Pool (MPP), a United Nations (UN)-backed public health organization" established in 2010 (these agreements comprising voluntary licensing). The consequences and benefits ("advantages and challenges") of compulsory licenses are also discussed. Statistics provided include that "87 percent of COVID-19 therapeutic R&D programs in the United States, and 75 percent of products in the global clinical development pipeline, originate from U.S.-based small- and medium-sized enterprises (SMEs)," according to the Biotechnology Innovation Organization (BIO). Also mentioned is the involvement of the Federal government in producing COVID-related therapeutics. Specific BLA agreements (to the extent they are public) discussed include Gilead/remdesivir, Merck/molnupiravir, and Eli Lilly/baricitinib, particularly as a means to avoid compulsory licenses and regulatory delays. Finally, the Chapter contains an extensive discussion of the advantages of using the MPP for voluntary licensing, stating:
Rather than negotiating individual license agreements directly with producers, licensors authorize the MPP to sublicense their technologies to third parties. In turn, the MPP solicits applications for sublicense agreements, reviews prospective producers, and handles all aspects of implementation of the sublicense agreements with selected companies.
(although conceding that academic research on the MPP is limited and what exists is focused on HIV/AIDS drugs). Merck is noted as the first company to enter into an MPP license in October, 2021 for molnupiravir (having the advantage that sublicenses come with a regulatory waiver), followed by Pfizer for nirmatrelvir (+ ritonavir) in November 2021, and Shionogi for ensitrelvir in October 2022. Examples of these MMP licenses are set forth in the following table:
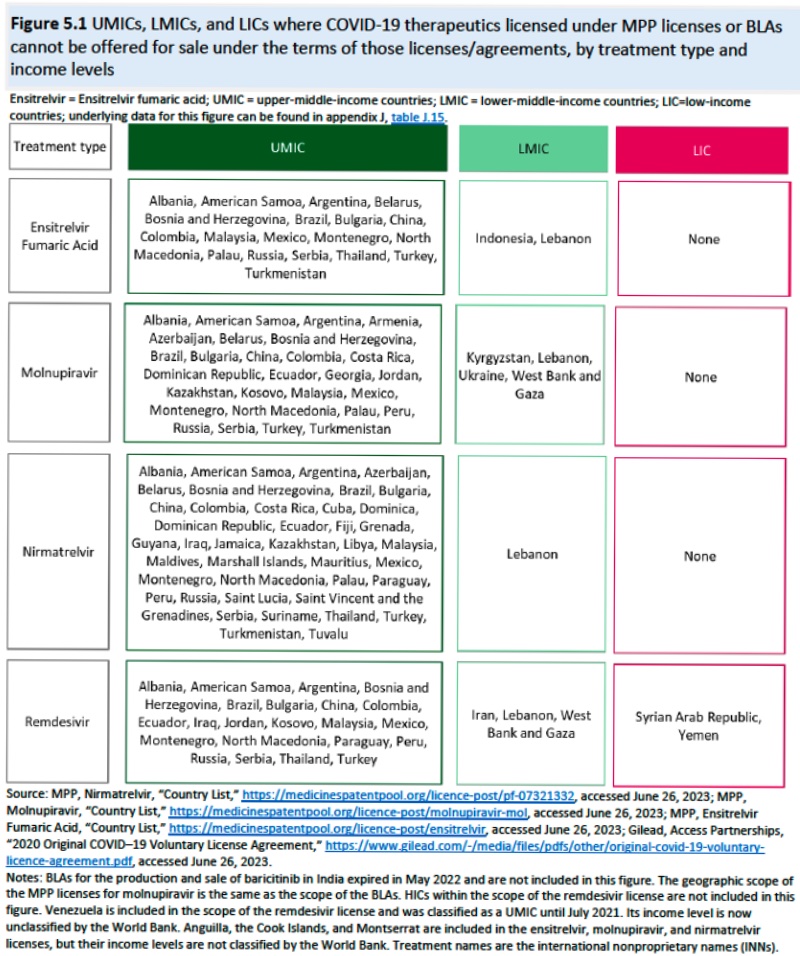
And the geographic distribution of MPP licenses shown in this diagram: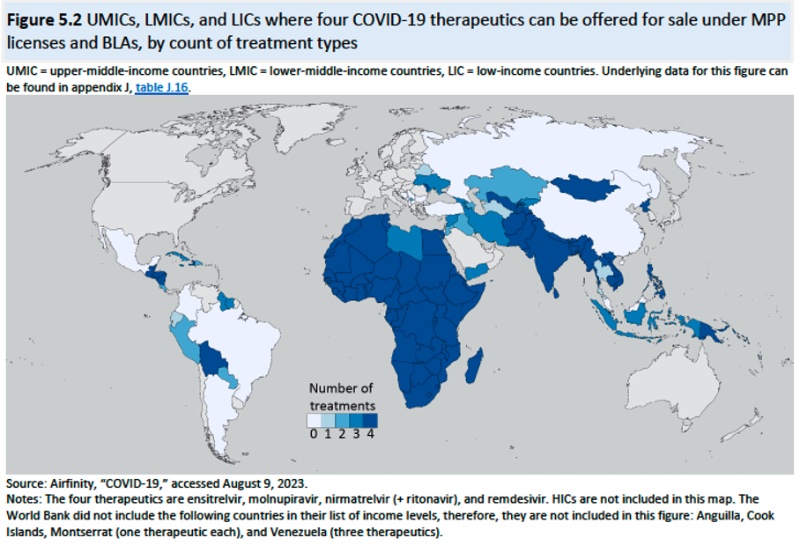
The Report also describes compulsory licenses which have not been used with COVID vaccines or diagnostics but have been imposed on COVID therapeutics. According to the Report, there have been approximately 121 attempts to issue a public health compulsory license (including 4 by the U.S.) since 2001, but only about 75% of threatened compulsory licenses were imposed, as illustrated by this diagram: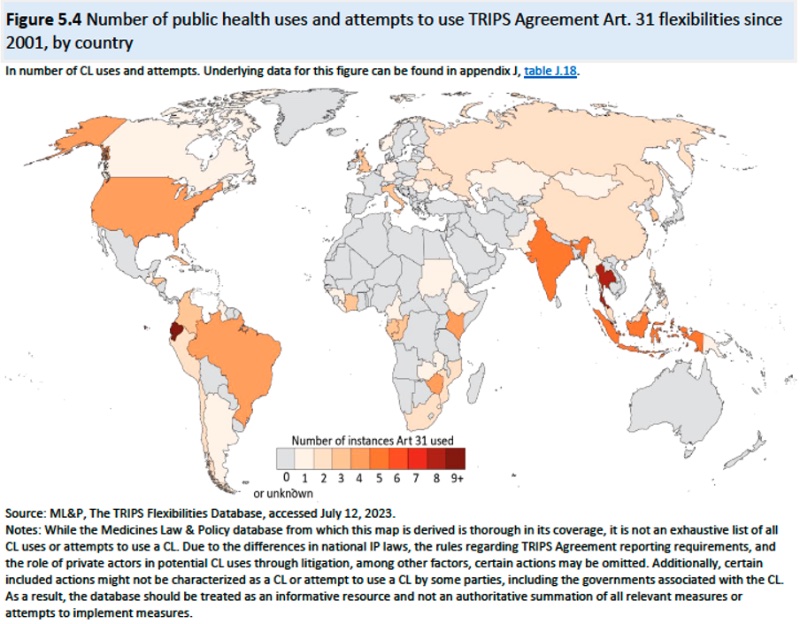
Advantages of such licenses include lowering drug prices, providing leverage in negotiations, and in some instances improving domestic manufacturing capacity. Of course, compulsory licensing has also resulted in retaliation against countries imposing them, including reducing investment or withholding drug products from the national market. Compulsory licenses are also imperfect in having the desired effect because they are limited to patents and cannot expropriate know-how necessary to produce a drug product. Specific instances of compulsory licensing discussed in the Report include Zambia, India, Brazil, Ecuador, Malaysia, South Africa, Thailand, and Canada (in this instance for importing HIV/AIDS drugs to Rwanda). And the effects of compulsory licenses on innovation are disputed by researchers according to the Report.Chapter 6: Availability and Consumption of COVID-19 Diagnostics and Therapeutics: This Chapter provides the Trade Representative with information regarding the necessity (or lack of it) of a COVID waiver extension based on global needs. This discussion is presented before the backdrop of fundamental disagreements between proponents and opponents of the waiver over the causes and even the existence of global deficiencies in providing COVID-19 diagnostics and therapeutics, arguing insufficient demand or insufficient (affordable) supply, with evidence on both sides of the debate. Needs assessments are limited almost entirely to therapeutics, there being no analogous studies or reports for diagnostics. The Report provides statistics relating to prices, purchases (including governmental purchases and multilateral programs and private sector purchases), donations, and consumption for diagnostics and therapeutics as well as factors affecting each of these statistics (such as "last mile delivery" effects). Regulatory authorizations (and needs therefor) as well as the effects of competing healthcare priorities in many countries is also discussed in the Report.
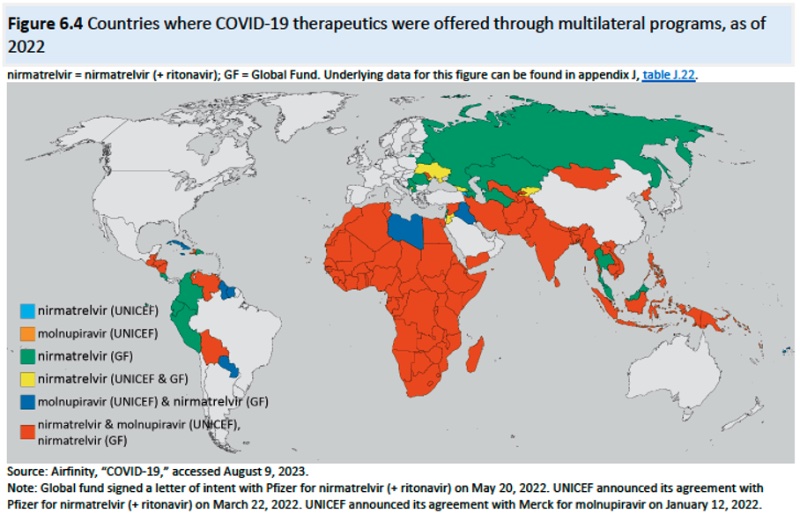
Chapter 7: Views of Interested Persons: This Chapter provides synopses and examples of public input requested by the Trade Representative. Providing a cautionary warning the Report states that "[t]he summaries of public views provided in this chapter do not attempt to assess, analyze, or draw conclusions about these views" and that "this chapter often uses language as provided by participants to avoid changing the meaning of their submitted views" and thus "some wording used in this chapter may not match wording used in other chapters of this report." The structure of the Report in this chapter is as follows:IP protection, R&D, and jobs
• The relationship between IP protection and corporate R&D expenditures, taking into account other expenditures, such as share buybacks, dividends, and marketing
• The location of jobs associated with the manufacturing of diagnostics and therapeutics, including in the United States
The TRIPS Agreement and access to medicine
• Whether and how existing TRIPS rules and flexibilities can be deployed to improve access to medicines
• Successes and challenges in using existing TRIPS flexibilities
• To what extent further clarifications of existing TRIPS flexibilities would be useful in improving access to medicines
The TRIPS Agreement and COVID-19 diagnostics and therapeutics
• How the TRIPS Agreement promotes innovation in and/or limits access to COVID-19 diagnostics and therapeutics
• The extent to which products not yet on the market, or new uses for existing products, could be affected by an extension of the Ministerial Decision to COVID-19 diagnostics and therapeutics
• The relevance, if any, of the fact that diagnostic and therapeutic products used with respect to COVID-19 may also have application to other diseases
Appendix D of the Report contains the positions and policy statements of witnesses at the public hearings
The Report is a wealth of information and data (albeit wherein some of the data is incomplete) and abides by the request by the Trade Representative not to espouse any particular course of action or outcome. Accordingly, the Report can be expected to be a useful tool during negotiations. It is the reality, however, that whatever course the Administration takes will, in the absence of fact- or data-driven outcomes predominantly be a political decision based more of the concessions that can be obtained than any demonstrated need for any extension of the IP waiver from these data. Should increasing the waiver come to pass the consequences are unlikely to be immediately felt, unless another virulent COVID-19 variant arises that cause higher morbidity and mortality than the current crop of variant COVID-19 species has developed. The greater risk is in the precedent that a widened waiver would set and whether such a waiver would in the next pandemic reduce the capability for the type of rapid development of vaccines, diagnostics, and therapeutics that have (at least for now) stemmed the tide of the COVID-19 pandemic to manageable levels. That is an outcome that will only become apparent through experience, and such an experience may be very costly indeed and completely unpredictably so. Prudence suggests it would be wise to court such an outcome carefully.
For additional information regarding this topic, please see:
• "Top Four Stories of 2022," January 18, 2023
• "WTO TRIPS Council Recommends That General Council Extend Waiver Deadline," December 18, 2022
• "Nine Countries Seek Extension of WTO Waiver to COVID-19 Therapeutics and Diagnostics," December 11, 2022
• "Status of Proposed Extension of TRIPS Waiver in WTO," December 8, 2022
• "C4IP Presents Webinar on COVID Waiver Extension," December 5, 2022
• "Senators Send Letter to Commerce Secretary Regarding WTO Waiver Compromise," March 28, 2022
• "The Proposed WTO IP Waiver: Just What Good Can It Do? — An Analysis," March 24, 2022
• "IP Associations "Concerned" by Reports of TRIPS Waiver Compromise," March 24, 2022
• "More on Leaked WTO COVID-19 Vaccine Patent Waiver Compromise," March 21, 2022
• "Compromise Reportedly Reached on COVID-19 Vaccine Patent Waiver," March 16, 2022
• "Sen. Tillis Writes to U.S. Trade Representative (Again) Regarding TRIPS Waiver," December 12, 2021
• "U.S. Trade Representative Responds to Letters from Senators Regarding TRIPS Waiver," November 14, 2021
• "U.S. Chamber of Commerce Urges Administration to 'Double Down' on Global Vaccine Distribution," November 3, 2021
• "Is This the WTO Waiver End Game?" July 25, 2021
• "BIO Declaration on Global Access to COVID-19 Vaccines and Treatments and Role of IP," June 24, 2021
• "GOP Legislators Write in Opposition to Proposed TRIPS Waiver," May 16, 2021
• "Population of Patents at Risk from Proposed WTO Patent Waiver," May 12, 2021
• "Sen. Daines Urges Biden Administration to Withdraw Support for COVID-19 IP Waiver," May 12, 2021
• "Pfizer CEO Pens Open Letter on COVID-19 Vaccine IP Waiver," May 10, 2021
• "If the Devil of the WTO IP Waiver Is in the Details, What Are the Details?" May 9, 2021
• "The Road to Hell Is Paved with What Everybody Knows," May 6, 2021
• "BIO & IPO Issue Statements on Biden Administration's Support for Proposed WTO Waiver," May 6, 2021
• "Biden Administration Supports Waiver of IP Protection for COVID-19 Vaccines," May 5, 2021
• "Suspending IP Protection: A Bad Idea (That Won't Achieve Its Desired Goals)," April 26, 2021
• "Sen. Tillis Asks Biden Administration to Oppose WTO Waiver Proposal," April 21, 2021
• "IP Organizations Support Continued Opposition to Waiver Proposal," April 5, 2021
• "Industry Coalition Supports Continued Efforts to Oppose Waiver Proposal," March 29, 2021
• "BIO and PhRMA Urge Biden Administration to Oppose Proposed WTO TRIPS Waiver," March 11, 2021
• "IPO Sends Letter on IP Law and Policy to President-Elect and Vice President-Elect," January 4, 2021 -
By Michael Borella —
 As a follow up to last week's article about the Biden executive order on artificial intelligence (AI), this is a brief overview of one of its provisions that has proven to be controversial — namely, the additional scrutiny that the government plans for AI models and computing clusters over a certain size. The order states that the Secretary of Commerce shall require that entities acquiring, developing, training, or using such models must report their activities to the government, including results of red-team testing intended to discover flaws and vulnerabilities of these systems.
As a follow up to last week's article about the Biden executive order on artificial intelligence (AI), this is a brief overview of one of its provisions that has proven to be controversial — namely, the additional scrutiny that the government plans for AI models and computing clusters over a certain size. The order states that the Secretary of Commerce shall require that entities acquiring, developing, training, or using such models must report their activities to the government, including results of red-team testing intended to discover flaws and vulnerabilities of these systems.The current size thresholds are:
(i) any model that was trained using a quantity of computing power greater than 1026 integer or floating-point operations, or using primarily biological sequence data and using a quantity of computing power greater than 1023 integer or floating-point operations; and
(ii) any computing cluster that has a set of machines physically co-located in a single datacenter, transitively connected by data center networking of over 100 Gbit/s, and having a theoretical maximum computing capacity of 1020 integer or floating-point operations per second for training AI.
For sake of comparison, today's high-end PCs typically can maintain a few trillion floating-point operations per second (teraflops), which is around 1012 floating-point operations. The NVIDIA DGX H100 server, which is specifically designed for supporting AI uses, is rated at 32 petaflops, which is around 1016 floating-point operations. Thus, to reach the 1020 floating-point operation threshold, one would need about 10000 DGX H100s. At a price of about $400,000 each, it would require exceptionally deep pockets to even consider building such a system, and this does not even take into account the cost of electrical power.[1]
According to some reports, OpenAI's GPT-3 was trained on a cluster that has about 800 petaflops of computing power (just under 1018 floating-point operations per second).[2] Hitting the regulatory threshold would require a system with about 100 times more capacity. OpenAI did not release the technical details of its training cluster for GPT-4 but did indicate that the training cost the company over $100 million.[3]
It is safe to say that only a handful of non-governmental entities currently have the ability to train models that would trigger the reporting required by the order. But, if anything, we have learned over the last several decades that the amount of computing power available per dollar increases by an order of magnitude every few years. Thus, the 1020 floating-point operation threshold may be within reach of many entities in the near future.
Despite the call for regulation coming from OpenAI and other leaders in generative AI, there has been some pushback. So far, this is largely originating with the open source community and venture capitalists.
Researcher Andrew Ng is concerned that regulations based on model size are an overreaction to the dangers of AI, would stifle innovation, and would advantage the incumbent players.[4] Researcher Yann LeCun, with a number of other tech executives and venture capitalists, sent a letter to President Biden arguing that the order would result in AI being dominated by a few well-heeled companies.[5] The letter also urges the president to consider the benefits that open source AI models can bring to the public by increasing marketplace competition while opening development and use of the technology to academia and individuals.
The argument for taking a lighter regulatory touch on open source AI models is that they foster collaboration, transparency, and security by democratizing the use of AI. An open source mantra is that "given enough eyeballs, all bugs are shallow." This can be interpreted to mean that the world's programming community being able to inspect these models would increase their reliability and usefulness while reducing harm. Yet, by their very nature, open source initiatives are unlikely to have the resources to engage with regulators. Furthermore, the concern over the most powerful AI in the world being black boxes understood and controlled by only a handful of people is very real.
While open source has proven to be an incredibly powerful tool for advancing technology and bringing it to the masses — note the rise of Linux over the last 30 years — a blind adherence to its principles ignores some obvious risks. Bad actors of all stripes could obtain and customize generative AI models at a low cost, potentially removing any guardrails and using them to produce an unlimited amount of misinformation, deepfakes, and cyberattacks, or even to design novel bioweapons. There is a big difference between open sourcing an operating system or web server versus open sourcing a large language model with even a fraction of GPT-4's capabilities.
At this juncture, we appear to be between the devil and the deep blue sea. The question comes down to who can be trusted more — a handful of wealthy tech firms, the governments of the world, or the public.
The order draws some lines in the sand, but is still a thoughtful response to an emerging technology that has the potential to be simultaneously useful and dangerous. And unlike many regulatory efforts, the Biden administration is not waiting for a disaster to happen before putting a framework into place. One thing that we do know about the generative AI space is that size matters — scaling up models can produce emergent capabilities. Given this, focusing initial scrutiny on models above a certain size is not a bad idea.
[1] This is just a back of the envelope calculation based on currently available information. The costs of the NVIDIA and other hardware is currently in flux due to market demand.
[2] https://www.linkedin.com/pulse/mind-boggling-processing-power-cost-behind-chat-gpt-what-thakur/
[3] https://www.wired.com/story/openai-ceo-sam-altman-the-age-of-giant-ai-models-is-already-over/
[4] https://www.cnbc.com/2023/11/02/biden-ai-executive-order-industry-civil-rights-labor-groups-react.html
-
By Kevin E. Noonan –
 Ever since Watson and Crick ended their seminal Nature paper in 1953 by saying that: "It has not escaped our notice that the specific pairing we have postulated immediately suggests a possible copying mechanism for the genetic material," James Watson & Francis Crick, "Molecular Structure of Nucleic Acids; A Structure for Deoxyribose Nucleic Acid," Nature 171: 737-38 (April 25, 1953), it has been envisioned that identification of genetic mutations causing disease would someday provide a way to repair them to achieve a cure. Those hopes have only increased over the years since then, particularly in light of the ever-increasing genetic information that arose from cloning specific human genes in the late 1970's and the 1980's and even more so with the crowning achievement of the Human Genome Project at the turn of the century. Unfortunately, these hopes have not been fulfilled, in large part due to failures in successful introduction of unmutated genes into appropriate cells in patients suffering from genetic diseases. The causes for these failures are many, ranging from delivery of intact genetic constructs into these cells to stable insertion of the genes into the genome at a chromosomal location capable of efficient transcription under circumstances that did not result in inappropriate or disease-causing) expression of endogenous genes.
Ever since Watson and Crick ended their seminal Nature paper in 1953 by saying that: "It has not escaped our notice that the specific pairing we have postulated immediately suggests a possible copying mechanism for the genetic material," James Watson & Francis Crick, "Molecular Structure of Nucleic Acids; A Structure for Deoxyribose Nucleic Acid," Nature 171: 737-38 (April 25, 1953), it has been envisioned that identification of genetic mutations causing disease would someday provide a way to repair them to achieve a cure. Those hopes have only increased over the years since then, particularly in light of the ever-increasing genetic information that arose from cloning specific human genes in the late 1970's and the 1980's and even more so with the crowning achievement of the Human Genome Project at the turn of the century. Unfortunately, these hopes have not been fulfilled, in large part due to failures in successful introduction of unmutated genes into appropriate cells in patients suffering from genetic diseases. The causes for these failures are many, ranging from delivery of intact genetic constructs into these cells to stable insertion of the genes into the genome at a chromosomal location capable of efficient transcription under circumstances that did not result in inappropriate or disease-causing) expression of endogenous genes.On October 31st, the U.S. Food and Drug Administration's Cellular, Tissue, and Gene Therapies Advisory Committee announced its recommendation that exagamglogene autotemcel (exa-cel) is safe for clinical use. This drug is the subject of biologics license application (BLA) 125787 from Vertex Pharmaceuticals, Inc. in collaboration with CRISPR Therapeutics (Zug, Switzerland) and provides for the first time gene therapy for sickle cell anemia. Sickle cell anemia was the first known human disease associated with a genetic polymorphism (an A→T single nucleotide polymorphism, or SNP), resulting in the substitution of a valine residue (encoded by a GTG codon) for a glutamic acid residue (encoded by a GAG codon) in the beta chain of human hemoglobin.* The Advisory Committee's announcement followed the FDA's earlier announcement on October 27th that exagamglogene autotemcel is effective, having 29 of 30 patients stay pain-free for 18 months.
The CRISPR-mediated effect on sickle cell disease from exa-cel is mediated by targeting the binding site of an inhibitor (BCL11a) of the gamma-globulin gene, which results in fetal globin expression that alleviates the symptoms of the disease. While there remains the possibility in some patients for "off-target" effects (some of which have been predicted by scanning human genomic DNA for such targets), the expected frequencies are low (1.6% according to David Altshuler MD, Vertex's Chief Science Officer). Scot Wolfe, PhD (UMass Chan Medical School), a member of the FDA advisory committee, voiced the sentiment that:
We don't want to let perfect be the enemy of the good. At some point we have to try things out in patients. There is a huge unmet need for sickle cell disease. It's important to advance therapies. I certainly think that this is one of them.
The FDA is scheduled to decide whether to approve the drug at a meeting scheduled for December 8th.
* The work, by Linus Pauling and colleagues at Cal Tech was published in the November 25, 1949 issue of the journal Science, about three and a half years before Watson and Crick deciphered the structure of DNA.
-
By Michael Borella —
 On October 30, President Biden signed a sprawling executive order governing the development, testing, and use of artificial intelligence (AI). Formally titled, "Executive Order on the Safe, Secure, and Trustworthy Development and Use of Artificial Intelligence," the order sets forth guiding principles and actions to be taken by federal agencies. These actions include research, coordination with industry, academia, and the international community, promulgation of regulations, publication of reference materials, and establishment of a White House Artificial Intelligence Council to assist the administration with all of the above.
On October 30, President Biden signed a sprawling executive order governing the development, testing, and use of artificial intelligence (AI). Formally titled, "Executive Order on the Safe, Secure, and Trustworthy Development and Use of Artificial Intelligence," the order sets forth guiding principles and actions to be taken by federal agencies. These actions include research, coordination with industry, academia, and the international community, promulgation of regulations, publication of reference materials, and establishment of a White House Artificial Intelligence Council to assist the administration with all of the above.The order does not have the staying power of legislation, as it may effectively expire or be revoked after Biden leaves office. Nonetheless, it touches on a significant number of areas relating to AI and may serve to kick-start a regulatory framework for these advancing technologies.
The motivation behind the order is undoubtedly the recent emergence of generative AI that has been rapidly adopted by businesses and the public. This includes well-known large language models (LLMs) and image generation models, as well as newer and still-evolving video generation and music generation models. Generative AI has proven to be revolutionary compared to traditional machine learning techniques, most of which are focused on accurate classification of information. It is also has been developing very quickly, as the LLMs and image generation models of 2023 are notably better than those of 2022.
This has led to a number of concerns regarding the training and use of generative AI, such as deepfakes, discrimination, job displacement, intellectual property infringement, privacy violations, misinformation, and weapons development, just to name a few. Popular media of the last several decades has mostly emphasized AI's potential for harm rather than its beneficial uses. Consequently, a natural response to these risks is to catastrophize. That said, anyone who is not feeling anxiety of the potential misuses of AI has probably not thought about them thoroughly. On the other hand, AI has great potential to cure diseases, simplify artistic processes, democratize education, offload boring and mundane manual tasks from humans, adapt content for disabled individuals, and advance science and technology across the board.
Thus, the order was probably inevitable and should be viewed as a positive first step. Of course, the devil is in the details and the order is lengthy. The purpose of this article is to introduce the content of the order by focusing on its eight partially-overlapping guiding principles and briefly touch on how they are intended to be applied.
Safety and security: The order states that "[m]eeting this goal requires robust, reliable, repeatable, and standardized evaluations of AI systems, as well as policies, institutions, and, as appropriate, other mechanisms to test, understand, and mitigate risks from these systems before they are put to use." These risks include those related to "biotechnology, cybersecurity, critical infrastructure, and other national security dangers." The order proposes to establish testing, evaluation, and performance monitoring standards and practices, as well as "effective labeling and content provenance mechanisms, so that Americans are able to determine when content is generated using AI and when it is not."
Promoting responsible innovation, competition, and collaboration: The goal of this principle is to facilitate the United States being a leader in all things AI. "This effort requires investments in AI-related education, training, development, research, and capacity, while simultaneously tackling novel [IP] questions and other problems to protect inventors and creators." The order also emphasizes a need for fairness and competition by "stopping unlawful collusion and addressing risks from dominant firms' use of key assets such as semiconductors, computing power, cloud storage, and data to disadvantage competitors, and . . . supporting a marketplace that harnesses the benefits of AI to provide new opportunities for small businesses, workers, and entrepreneurs."
Support of American workers: The order indicates that the administration "will seek to adapt job training and education to support a diverse workforce and help provide access to opportunities that AI creates." However, "AI should not be deployed in ways that undermine rights, worsen job quality, encourage undue worker surveillance, lessen market competition, introduce new health and safety risks, or cause harmful labor-force disruptions."
Equity and civil rights: The order recognizes that "[AI] systems deployed irresponsibly have reproduced and intensified existing inequities, caused new types of harmful discrimination, and exacerbated online and physical harms." Being able to trust that new AI systems are treating individuals fairly is critical to its growth and adoption. Thus, Biden seeks to "promote robust technical evaluations, careful oversight, engagement with affected communities, and rigorous regulation."
Consumer protection: Continuing on the goal of trust, the order states that the government will "enforce existing consumer protection laws and principles and enact appropriate safeguards against fraud, unintended bias, discrimination, infringements on privacy, and other harms from AI." It notes that these laws and safeguards are particularly relevant to "critical fields like healthcare, financial services, education, housing, law, and transportation" where unregulated use of AI could cause great harm to individuals and society.
Privacy and civil liberties: A further goal is to combat the risk that sensitive "personal data could be exploited and exposed." Here, the order instructs agencies to "use available policy and technical tools, including privacy-enhancing technologies (PETs) where appropriate, to protect privacy and to combat the broader legal and societal risks — including the chilling of First Amendment rights — that result from the improper collection and use of people's data."
The Federal Government's use of AI: The order seeks to "attract, retain, and develop public service-oriented AI professionals, including from underserved communities, across disciplines . . . and ease AI professionals' path into the Federal Government to help harness and govern AI." This would also involve training the federal workforce to "understand the benefits, risks, and limitations of AI for their job functions, and . . . ensure that safe and rights-respecting AI is adopted, deployed, and used."
International leadership: Finally, the order points to the importance of working with the international community on issues of AI development as well as safeguards. Thus, the administration intends to "engage with international allies and partners in developing a framework to manage AI's risks, unlock AI's potential for good, and promote common approaches to shared challenges." Particular emphasis is on promoting "responsible AI safety and security principles and actions with other nations."
The meat of the order focuses on initial steps to be taken in these directions, with many including particular requirements and timelines. For example, the order instructs the Secretary of Energy to develop a plan for "tools to evaluate AI capabilities to generate outputs that may represent nuclear, nonproliferation, biological, chemical, critical infrastructure, and energy-security threats or hazards" within 270 days.
A type of AI model that the order specifically addresses is a so-called "dual-use foundation model", defined as "an AI model that is trained on broad data; generally uses self-supervision; contains at least tens of billions of parameters; is applicable across a wide range of contexts; and that exhibits, or could be easily modified to exhibit, high levels of performance at tasks that pose a serious risk to security, national economic security, national public health or safety." Currently, generative AI models would likely fall into this category. The order requires that companies developing or planning on developing dual-use foundational models to periodically report to the government on:
• "any ongoing or planned activities related to training, developing, or producing dual-use foundation models, including the physical and cybersecurity protections taken to assure the integrity of that training process against sophisticated threats;"
• "the ownership and possession of the model weights of any dual-use foundation models, and the physical and cybersecurity measures taken to protect those model weights;" and
• "the results of any developed dual-use foundation model's performance in relevant AI red-team testing." To this end, the order also instructs the National Institute of Standards and Technology (NIST) to develop red-team testing standards that evaluate to what extent an AI model can be used for harmful purposes.
Additional reporting requirements include divulging the existence, locations, and ownership of large-scale computing clusters — arguably, those that can be used to train and execute dual-use foundation models.
Thus, the order requires that the government know what entities are doing, where they are doing it, and how they are doing it with regard to such dual use models. The Biden administration appears to be viewing generative AI like a dangerous munition that cannot be developed under the veil of secrecy by a non-governmental entity.
In the past, Silicon Valley's response to the government attempts to rein it in has been along the lines of "Don't regulate us, bro!" So far, reaction to the order has been largely positive. The major players may have realized that an absence of regulation in this area could ultimately slow AI adoption by businesses and the public due to a lack of trust. If the framework set forth in the order is intelligently implemented, these concerns may be largely assuaged, allowing AI to continue its remarkable growth but with many of its inherent risks understood, limited, or mitigated.
-
By Donald Zuhn —
Patent Center to Replace EFS-Web and Private PAIR in November
In a press release issued last month, the U.S. Patent and Trademark Office announced that the Patent Center system will fully replace the EFS-Web system and the Private Patent Application Information Retrieval (Private PAIR) tool for the electronic filing and management of patent applications beginning on November 8, 2023. The Office noted that since the launch of Patent Center six years ago, the system has undergone rigorous user testing and iterative improvements, based largely on public feedback, and that the Office would continue to use the feedback it receives from stakeholders to further refine Patent Center to meet stakeholder needs. The Office also noted that suggestions and ideas for improvements to the Patent Center system can be submitted by contacting emod@uspto.gov.
USPTO to Use Applicant-Suggested Figure in Patent Application Publications
In a notice published in the Official Gazette (1514 OG 107) last month, the U.S. Patent and Trademark Office announced that the Office will begin exclusively using the drawing figure suggested by an Applicant for the front page of a patent application publication when the suggestion is included on a compliant Application Data Sheet (ADS) that has been timely filed before the application publication process has begun. Prior to this change in procedure, which took effect earlier this month, the Office had not been bound by an Applicant's suggestion. If an Applicant does not include a figure suggestion in a compliant ADS filed before the publishing process begins, the Office will select a figure.
The Office noted that the publication process typically begins about fourteen weeks before the projected publication date listed on the filing receipt. The OG notice also indicates that although the Office will make every effort to use the figure selected by an Applicant, the Office "continues to not be bound by the applicant's figure suggestion," and the Office "will not treat its failure to use the figure suggested by the applicant for the first page of the pre-grant publication as a material mistake in the publication for purposes of a corrected patent application publication under 37 CFR 1.221(b)."
USPTO Eliminates e-Office Action Postcard
 In a notice published in the Federal Register (88 Fed. Reg. 64899) last month, the U.S. Patent and Trademark Office announced the elimination of the courtesy postal postcard that the Office had been sending to stakeholders participating in the Electronic Office (e-Office) Action program. The Office had been sending postal postcards to e-Office Action users as a reminder when communications that were available had not been viewed or downloaded within seven calendar days after the date of the notification and at least one of the listed Office communications required an Applicant's reply. The Office will still offer e-postcards (courtesy postcards sent by email). The Office noted that it had sought public comment on the elimination of the postal postcards and had received a single comment, which expressed views on Patent Center and not on the postal postcard.
In a notice published in the Federal Register (88 Fed. Reg. 64899) last month, the U.S. Patent and Trademark Office announced the elimination of the courtesy postal postcard that the Office had been sending to stakeholders participating in the Electronic Office (e-Office) Action program. The Office had been sending postal postcards to e-Office Action users as a reminder when communications that were available had not been viewed or downloaded within seven calendar days after the date of the notification and at least one of the listed Office communications required an Applicant's reply. The Office will still offer e-postcards (courtesy postcards sent by email). The Office noted that it had sought public comment on the elimination of the postal postcards and had received a single comment, which expressed views on Patent Center and not on the postal postcard.
USPTO Virtual Assistant Now Available to UsersIn a notice published on the Office's website earlier this month, the U.S. Patent and Trademark Office announced that it had launched the USPTO Virtual Assistant on selected Patents webpages. The Virtual Assistant will provide answers to common customer questions and make it easier to determine the status of patent applications. Users can simply type "What is the status of my application?" into the chat box and enter the application number when prompted. The Office noted that the Virtual Assistant is an example of its efforts to leverage machine learning to offer improved services, and that the Office expected to expand the Virtual Assistant's knowledge base and improve its ability to recognize and answer user questions as the tool is further developed.
-
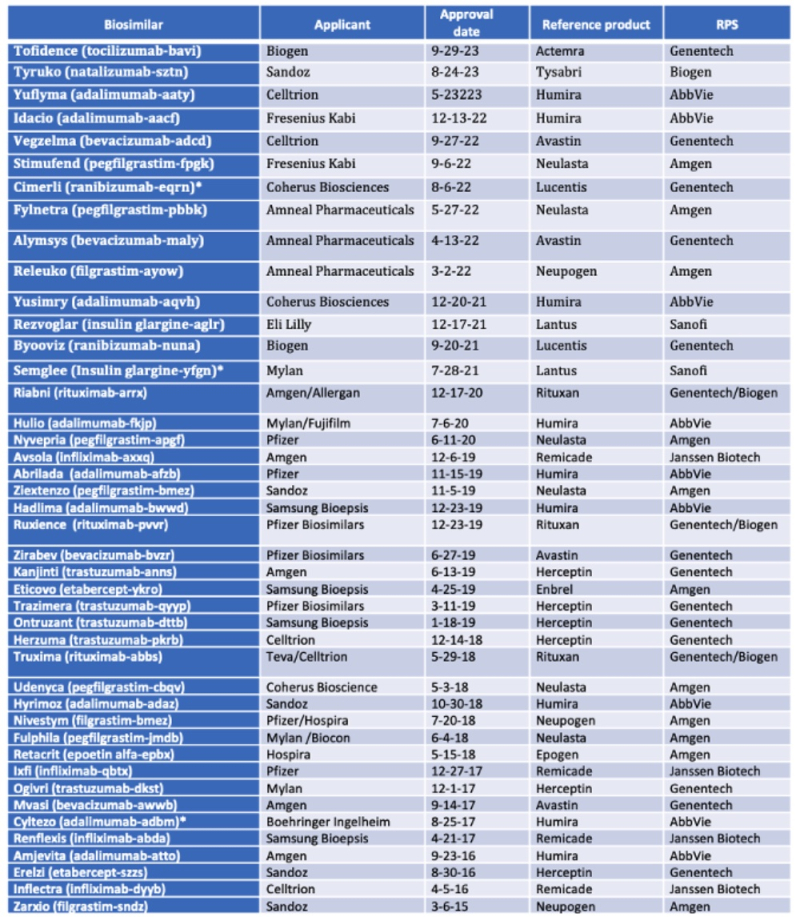
By Kevin E. Noonan –
 On September 29, the U.S. Food and Drug Administration announced approval of Biogen's Tofidence® (tocilizumab-bavi) as a biosimilar to Genentech's Actemra®, a drug approved for treating COVID-19, rheumatoid arthritis, giant cell arteritis, scleroderma, polyarticular or systemic juvenile idiopathic arthritis, and severe cytokine release syndrome. Tofidence® is the first Actemra biosimilar, and was approved as an intravenous formulation for treatment of moderately to severely active rheumatoid arthritis, polyarticular juvenile idiopathic arthritis and systemic juvenile idiopathic arthritis.
On September 29, the U.S. Food and Drug Administration announced approval of Biogen's Tofidence® (tocilizumab-bavi) as a biosimilar to Genentech's Actemra®, a drug approved for treating COVID-19, rheumatoid arthritis, giant cell arteritis, scleroderma, polyarticular or systemic juvenile idiopathic arthritis, and severe cytokine release syndrome. Tofidence® is the first Actemra biosimilar, and was approved as an intravenous formulation for treatment of moderately to severely active rheumatoid arthritis, polyarticular juvenile idiopathic arthritis and systemic juvenile idiopathic arthritis.This approval brings to 44 the number of approved biosimilar products.
-
By Kevin E. Noonan —
In view of the unprecedented uncertainty in patent law generated by counter-doctrinal Supreme Court decisions over the past decade or so and a cowed Federal Circuit relegated to complaining that their hands are tied on most matters (even when acknowledging that the decisions they are rendering do violence to the U.S. patent system; see now-Chief Judge Moore's dissent in Athena Diagnostics, Inc. v. Mayo Collaborative Services, LLC), an occasional clear decision that establishes a bright line in the law (once the Court's stock in trade) is refreshing even if the clarity comes with unwanted consequences. The Court's recent In re Cellect is one of those decisions, which held (as "once-and-for-all" as can be expected for the Court in its current incarnation) that the primary concern when assessing the interplay between obviousness-type double patenting (ODP) and patent term adjustment (PTA) is whether a patentee can obtain claim term that extends past 20 years from its earliest priority date for any reason (other than patent term extension under 35 U.S.C. § 156 for FDA delay in approving a patented drug, a small and unique subset of all granted patents) in favor of the public's right to having the patented invention freely available upon expiration of the earliest to expire patent claiming patentably indistinguishable embodiments of the invention.
The issue, of course, is ODP and the extent to which terminal disclaimers are required under circumstances where a patent is entitled by statute to patent term adjustment (PTA) under circumstances where the claims in such a patent is deemed to be an obvious variant of claims in a related, earlier-expiring patent. To recap, the issue arose in a series of ex parte reexaminations over five patents owned by Cellect, U.S. Patent Nos. 6,424,369; 6,452,626; 6,982,742; and 7,002,621, that involve "solid state image sensors which are configured to be of a minimum size and used within communication devices specifically including video telephones" according to the '621 patent (only four of these patents were invalidated, the fifth, U.S. Patent No. 6,862,036 not having any PTA that raised the issue). The chronological situation is set forth in an exhibit from Cellect's Federal Circuit appeal brief and reproduced in modified form in the opinion:

There was no dispute that the claims in these applications were patentably indistinct. The Board issued four Decisions on Appeal affirming the reexamination division's invalidation of the '369, '626, '621, and '742 patents, all on the grounds that the provisions of 35 U.S.C. § 154(b)(2)(B) mandated that a terminal disclaimer be filed under circumstances where obviousness-type double patenting arose due to extension of patent term as PTA, i.e., that ODP must be determined after application of PTA. (The Federal Circuit reached a different conclusion with regard to patent term extension (PTE) under 35 U.S.C. § 156 in Novartis AG v. Ezra Ventures LLC, the Court expressly refusing to permit "a judge-made doctrine to cut off a statutorily-authorized time extension.") In its consolidated decision, the Board emphasized the potential inequities to the public due to the possibility of harassment by different parties owning patents to obvious variants of one another (in the absence of a terminal disclaimer preventing this potentiality) as representing an unjust extension of patent term to the public's detriment; see In re Fallaux, 564 F.3d 1313 (Fed. Cir. 2009)). The Board further held that under In re Longi, the public was entitled to the assumption that it is free to practice what is claimed in the patent and obvious modifications and variants thereof once the patent has expired. 759 F.2d 887 (Fed. Cir. 1985).The Federal Circuit affirmed the Board's judgment in these re-examinations in an opinion by Judge Lourie joined by Judges Dyk and Reyna. The primary basis for the Court's decision was that it is inequitable to the public that a second, later-expiring patent should be obtained ("an unjustified timewise extension of patent term") on an obvious variant of a patented invention, based on AbbVie Inc. v. Mathilda & Terence Kennedy Inst. of Rheumatology Tr., 764 F.3d 1366, 1373 (Fed. Cir. 2014). The panel's opinion found support in the statute (as had the Board), wherein application of PTA was limited under circumstances where there was or should have been a terminal disclaimer filed, which stated that "no patent the term of which has been disclaimed beyond a specified date may be adjusted under this section beyond the expiration date specified in the disclaimer." 35 U.S.C. § 154(b)(2)(B)* This conclusion was based in part by the Court's precedent, particularly AbbVie, and by the panel's determination that the overriding policy consideration is the need to "ensure that the applicant does not receive an unjust timewise extension of patent term" (as it has for over a decade; see "In re Janssen Biotech, Inc.; G.D. Searle LLC v. Lupin Pharmaceuticals, Inc."; "AbbVie Inc."; "Gilead Sciences, Inc. v. Natco Pharma Ltd."; "Eli Lilly & Co. v. Teva Parenteral Medicines, Inc."; and "Sun Pharmaceutical Industries, Ltd. v. Eli Lilly & Co.").
Having spoken plainly (and seeing that the likelihood the Supreme Court will weigh in is substantially zero), the question is what prosecution stratagems can be devised to permit maximizing patent term by (at a minimum) having a patent receive its statutory PTA without running afoul of concerns over public expectations. Some have argued that filing "preemptive" terminal disclaimers would at least prevent patents from being invalidated on ODP grounds after the exclusivity of such patents has been relied upon to protect investment in drugs and other inventions requiring exclusivity terms that provide a sufficiently robust return on investment (see, for example, Mitra et al., 2023, In re Cellect: The Federal Circuit Alters Terminal Disclaimer Strategy; Huis et al., 2023, Patent Owners Must Consider New Terminal Disclaimer Strategies in View of the Federal Circuit's Decision in In re Cellect). The drawback of these strategies is that by definition they relinquish PTA even before entitlement to such PTA has been challenged and thus such terminally disclaimed patents may not provide the requisite exclusivity to support investment in the claimed technology.
There are, however, several alternative approaches. The most direct is to take advantage of the safe harbor provisions of 35 U.S.C. § 121 for claims subject to a restriction requirement. Particularly for inventions having claims encompassing several statutory categories under 35 U.S.C. § 101 (compositions, methods of making, methods of using, etc.) having a restriction requirement issued can provide a basis to avoid having to file a terminal disclaimer because the various claim groupings have been judged by the patent examiner to be patentably distinct. While not immune from later challenge, the presumption is that the Office's determination was correct and thus will be subject to the clear and convincing standard for invalidating patents for ODP. This tactic carries the responsibility to make certain that claims in the various groups are kept within their patentably distinct "silos" to maintain the safe harbor (see Boehringer Ingelheim Int'l. v. Barr Laboratories, Inc. (Fed. Cir. 2010); Obviousness-type Double Patenting after Amgen v. F. Hoffmann-LaRoche), a concept termed "consonance" in this regard. And it can also be prudent to avoid taking the opportunity to request rejoinder under M.P.E.P. § 821.04 of certain otherwise patentably distinct groups of claims (such as method claims being rejoined to allowed composition claims) which has the effect of having these patentably distinct claims issued in the same granted patent and thus having the same expiration date (which could in some instances be less than could be obtained under the PTA statute).
Substantively, assertion of ODP can be addressed as with any other obviousness rejection, by challenging motivation-to-combine disclosure of related applications with other prior art references, or whether species encompassed by ODP-rejected claims have features (like unexpected results) not shared with earlier claimed embodiments. Other objective indicia (like commercial success) may be available for species claims to the eventual commercial embodiment. Such strategies will require more careful consideration of what claims are pursued and in what order, which in turn will benefit from close coordination between business development actors and patent prosecutors that, while always recognized as being beneficial has not always been pursued with sufficient diligence. In this regard, it is important to remember that "the patent disclosure [of an earlier related patent asserted in an ODP rejection] is not 'prior art' and cannot be looked to for what it teaches," In re Baird, 348 F.2d 974, 979 (C.C.P.A. 1965), which renders such patents much more limited than prior art to another (which can be considered for obviousness purposes for all that it teaches and is not limited to whether the reference is enabling, see Amgen Inc. v. Hoechst Marion Roussel Inc., 314 F.3d 1313, 1357 (Fed. Cir. 2003) ("Under [§]103, however, a reference need not be enabled; it qualifies as a prior art, regardless, for whatever is disclosed therein) (emphasis added). Moreover, a species claim is not necessarily obvious over an earlier genus claim and can be found to be patentably distinct using the analytical rubrics contemplated herein. In re Sarett, 327 F.2d 1004 (C.C.P.A. 1964); an illustrative example is In re Vogel, where the Court of Customs and Patent Appeals held that claims to a method of preparing a beef product in a later application was patentably distinct from claims to a similar but not identical method for preparing a pork product but claims to a similar method for making a meat product were not patentably distinct. 422 F.2d 438 (C.C.P.A. 1970). This is also true for distinguishing claims to compositions and methods for using them. In re Boylan, 392 F.2d 1017, 1022-23 (C.C.P.A. 1968).
Implementing these various ways of avoiding ODP rejections and the need for PTA-destroying terminal disclaimers takes intentional planning when drafting claims and assessments that heretofore have not had compelling reasons to be performed for time-saving and drafting-efficiency reasons. In view of the Federal Circuit's decisions precluding any consideration of what is fair to the patentee (as opposed to the public), making the effort to avoid the need for filing a terminal disclaimer seems worth doing to the broadest extent possible.
* There is no such limitation in 35 U.S.C. § 156, even though both statutes recite that an extension of the term shall be granted.