By Kevin E. Noonan –
 In what everyone (including the Patent Trial and Appeal Board) considers an unprecedented administrative action, the PTAB late last year set out an order (Patent Trial and Appeal Board's Order, 2017 WL 5067421, P.T.A.B. Nov. 3, 2017) inviting amicus briefing on the question of whether the transfer of patent rights from Allergan to the St. Regis Mohawk Nation should lead the Board to dismiss several consolidated inter partes review actions (Nos. IPR2016-01127, IPR2016-01128, IPR2016-01129, IPR2016-01130, IPR2016-01131, and IPR2016-01132, instituted against U.S. Patent Nos. 8,685,930, 8,629,111, 8,642,556, 8,633,162, 8,648,048, and 9,248,191, respectively) on the grounds of sovereign immunity (see "Mohawk Nation Exercises Sovereign Immunity in Inter Partes Review"). The Board has ruled previously that state universities, as "arms of the state," could so assert the immunity and have such IPRs dismissed (see Covidien LP v. Univ. of Fla. Research Found., Inc., Case IPR2016-01274 (PTAB Jan. 25, 2017); NeoChord, Inc. v. Univ. of Md., Balt., Case IPR2016-00208 (PTAB May 23, 2017); and Reactive Surfaces Ltd., LLP v. Toyota Motor Corp., Case IPR2016-01914 (PTAB July 13, 2017)). The question is not as clear for the Native American tribes, because their sovereign immunity is more proscribed and subject to abrogation by Congress (albeit requiring express and unequivocal government action; see, Santa Clara Pueblo v. Martinez and Ransom v. St. Regis Mohawk Educ. and Cmty. Fund, Inc., 86 N.Y.2d 553, 560 (1995)). Even assertion of sovereign immunity by the several States is controversial (see "The Wall Street Journal's Problem with the U.S. Constitution"); indeed, the Board denied a motion to dismiss recently based on assertion of the patent in parallel district court litigation (see "PTAB Decides Patent Infringement Lawsuit Waives Eleventh Amendment Sovereign Immunity to Inter Partes Review").
In what everyone (including the Patent Trial and Appeal Board) considers an unprecedented administrative action, the PTAB late last year set out an order (Patent Trial and Appeal Board's Order, 2017 WL 5067421, P.T.A.B. Nov. 3, 2017) inviting amicus briefing on the question of whether the transfer of patent rights from Allergan to the St. Regis Mohawk Nation should lead the Board to dismiss several consolidated inter partes review actions (Nos. IPR2016-01127, IPR2016-01128, IPR2016-01129, IPR2016-01130, IPR2016-01131, and IPR2016-01132, instituted against U.S. Patent Nos. 8,685,930, 8,629,111, 8,642,556, 8,633,162, 8,648,048, and 9,248,191, respectively) on the grounds of sovereign immunity (see "Mohawk Nation Exercises Sovereign Immunity in Inter Partes Review"). The Board has ruled previously that state universities, as "arms of the state," could so assert the immunity and have such IPRs dismissed (see Covidien LP v. Univ. of Fla. Research Found., Inc., Case IPR2016-01274 (PTAB Jan. 25, 2017); NeoChord, Inc. v. Univ. of Md., Balt., Case IPR2016-00208 (PTAB May 23, 2017); and Reactive Surfaces Ltd., LLP v. Toyota Motor Corp., Case IPR2016-01914 (PTAB July 13, 2017)). The question is not as clear for the Native American tribes, because their sovereign immunity is more proscribed and subject to abrogation by Congress (albeit requiring express and unequivocal government action; see, Santa Clara Pueblo v. Martinez and Ransom v. St. Regis Mohawk Educ. and Cmty. Fund, Inc., 86 N.Y.2d 553, 560 (1995)). Even assertion of sovereign immunity by the several States is controversial (see "The Wall Street Journal's Problem with the U.S. Constitution"); indeed, the Board denied a motion to dismiss recently based on assertion of the patent in parallel district court litigation (see "PTAB Decides Patent Infringement Lawsuit Waives Eleventh Amendment Sovereign Immunity to Inter Partes Review").
Despite its unconventionality, fifteen amicus briefs were filed, almost evenly split between those believing that the Tribe could properly assert sovereign immunity and the Board should dismiss the IPRs and those distinguishing this case from the grants of dismissal to state universities on both legal and policy grounds. (As set forth below, one brief breaks the tie in favor of denying the motion, under a unique contract law theory.) The briefs were filed by the following parties:
In favor of granting the motion to dismiss
• Amicus Curiae Brief of the Oglala Sioux Tribe
• Amicus Curiae Brief of The National Congress of American Indians et al.
• Amicus Curiae Brief of Luis Ortiz and Kermit Lopez
• Amicus Curiae Brief of Scholars
• Amicus Curiae Brief of the Seneca Nation
• Amicus Curiae Brief of the NAIPEC
• Amicus Curiae Brief of U.S. Inventor, LLC
In favor of denying the motion
• Amicus Curiae Brief of the SIIA
• Amicus Curiae Brief of The Association for Accessible Medicines
• Amicus Curiae Brief of The BSA | The Software Alliance
• Amicus Curiae Brief of the Public Knowledge and the Electronic Frontier Foundation
• Amicus Curiae Brief of Askeladden LLC
• Amicus Curiae Brief of Deva Holding A.S.
• Amicus Curiae Brief of the High Tech Inventors Alliance
• Amicus Curiae Brief of James R. Major
One of these briefs, by practitioners Luis Ortiz and Kermit Lopez challenged the Board's authority to solicit amicus briefs on this question:
[T]he APA only allows an agency to engage in rulemaking through the notice and comment procedure set forth in 5 U.S.C. § 553, unless the agency is creating rules that "are required by statute to be made on the record after opportunity for an agency hearing."
And:
Allowing amici briefs in this IPR by parties aligned with the petitioners does nothing more than allow various commercial interests to complain in the wrong forum. Only Congress, whose elected members have the authority and duty to make the political decision whether to abrogate sovereign immunity, and who must face voters after such decisions, can choose to eliminate sovereign immunity for Indian tribes in IPRs [emphasis in brief].
A similar sentiment was echoed in the brief in favor of denying the Tribe's motion, filed by the Public Knowledge and Electronic Frontier Foundation (as amicus collegii), which argued that public notice and comment under the APA are the proper route to permit public comment — "[t]hat procedure would enable participation by members of the public with key knowledge or interests, would guarantee consistency across Board decisions, and would work to avoid conflicts with other policy decisions and positions of the Office." Certifying the question to the Director would have the advantages that 1) "a notice and comment proceeding would enable all interested parties to participate and provide useful information to the Office and the Board, and to do so without expensive and onerous requirements of retaining registered counsel"; 2) "certification to the Director will avoid inconsistent policy across panels of the Board"; and 3) the Director is best positioned to assess any consequences for the Office as a whole."
Otherwise, as might be expected, the briefs recite common themes. Briefs in favor of granting the motion and dismissing the IPRs focus on it being the exclusive provenance of Congress to make any determinations on the scope of sovereign immunity for the Tribes, and that even the Supreme Court has been circumspect in encroaching on Congressional purview in this regard. These briefs also advocate the policy goals of tribal self-sufficiency; for example, the Oglala Sioux tribe's brief with regard to public policy of encouraging tribes for commercial activity and self-sufficiency, cities White Mountain Apache Tribe v. Bracker to the effect that these efforts can't just be "casinos alone." Several of these briefs take solace in Justice Sotomayor's concurring opinion in Michigan v. Bay Mills Indian Community, 134 S.Ct. 2024(2014). In that case, the Justice stated that:
"Tribes face a number of barriers to raising revenue in traditional ways," and, thus, "[i]f Tribes are ever to become more self-sufficient, and fund a more substantial portion of their own governmental functions, commercial enterprises will likely be a central means of achieving that goal." 134 S.Ct. at 2041 (Sotomayor, J., concurring) (emphasis added).
The Ortiz and Lopez brief argues that "[t]he PTO has no expertise in determining the scope of sovereign immunity in any context" and addressed the policy arguments by saying "[t]he Board cannot interpret the statutes it administers to meet a policy goal contrary to clearly stated Congressional will, nor can the Board manipulate the statutes it interprets in a way not commensurate with Congressional intent," citing Utility Air Regulatory Grp. v. EPA, 134 S.Ct. 2427, 2445 (2014). And, using the types of assessments the Supreme Court has been known to employ, their brief argues that the Leahy-Smith America Invents Act was enacted in light of current precedent regarding tribal sovereign immunity, including Kiowa Tribe of Okla. v. Manuf'g Techs., Inc., 523 U.S. 751 (1998), Okla. Tax Comm'n v. Citizen Band Potawatomi Indian Tribe of Okla., 498 U.S. 505 (1991) ("Potawatomi I"), and Santa Clara Pueblo v. Martinez, 436 U.S. 49 (1978) — Potawanomi being particularly relevant because it involved sovereign immunity regarding a tribe's commercial activity — ("not a close call") under Michigan v. Bay Mills Indian Community, 134 S.Ct. 2024, 2037 (2014), that it is up to Congress to limit tribal sovereign immunity. Finally, this brief uniquely cites Executive Order 13175 in support of "tribal sovereignty and self-determination" and that "[a]gencies shall respect Indian tribal self-government and sovereignty."
The brief from the National Congress of American Indians raised the pending Supreme Court decision in Oil States and Congressional action (including the STRONGER Patents Act) as reasons for the Board to delay making a decision, counsel that may be less persuasive in view of the statutory 12-month time limit on rendering a decision (albeit tempered by the additional 6-month extension available under extraordinary circumstances, a standard the current situation should easily satisfy). This brief also finds support for sovereign immunity outside the Constitution (which could be restricted to the States), finding the basis for the immunity in English common law, ALEXANDER HAMILTON, THE FEDERALIST NO. 81, 548-49 (Jacob E. Cooke ed., 1961). Insofar as the immunity derives from common law it should be "applicable to all sovereigns: federal, state, tribal, and foreign."
The common themes that 1) abrogation of sovereign immunity is limited to Act of Congress, and 2) the PTAB would be outside of its Constitutional scope as an Article I court is found in briefs from the Seneca Nation and U.S. Invent, an inventor group, the latter brief arguing:
[I]t would be inconceivable that an Article I administrative agency has the authority to make binding decisions concerning the applicability of Native American tribal sovereign immunity when the U.S. Supreme Court — which has plenary judicial oversight of that same administrative agency — has declared that absent Congressional legislation, the Court does not have the power to qualify or limit the assertion of Native American sovereign immunity.
This point is also the focus of a brief from a group of Constitutional scholars, including Lawrence Tribe, William Eskridge Jr., Erwin Chemerinsky, Joseph Singer, David Orozco, and Jonathan Massey. This brief (as well as the Seneca Nation brief) cites Republic of Philippines v. Pimentel, 553 U.S. 851, 864 (2008), which states that "once a tribunal recognizes that an assertion of sovereign immunity is 'not frivolous,' it is 'error' for the tribunal to proceed further to address the merits." The brief also asserts that the question of tribal sovereign immunity is beyond the Board's "statutory purview and institutional expertise":
The Supreme Court has established that administrative agencies should not decide questions — especially complex and sensitive questions, such as those arising from Petitioners' arguments against tribal sovereign immunity — beyond their statutory purview and institutional competence. In such situations, agencies lack the expertise to resolve broader policy issues and risk interference with Congress's legislative prerogatives. Further, an agency acting beyond its purview lacks legitimacy and accountability. Controversial measures such as Petitioners' proposed restrictions on tribal sovereign immunity require a broader national democratic debate than an agency like the Board can provide [citing Hampton v. Mow Sun Wong, 426 U.S. 88, 116 (1976), Greene v. McElroy, 360 U.S. 474 (1959), and King v. Burwell, 135 S. Ct. 2480, 2489 (2015)].
This institutional incompetence is exacerbated by the allegations that:
[T]he Allergan/Mohawk contract is a 'sham agreement'[,] a 'sham assignment'[,] a 'scheme[] to buy tribal immunity for dubious activities'[,] and even an example of 'rent-a-tribe' schemes" [internal quotation marks and citation omitted] because "[n]ot only are these arguments highly disrespectful to the sovereign Tribe, but adjudicating them will embroil the Board in an intrusive and politically charged inquiry into tribal motivations and the policy wisdom of tribal economic freedom. These are issues for Congress, not the courts, and not an agency.
Allegations that the underlying agreement is a sham are also addressed in an amicus brief from the Native American Intellectual Property Enterprise Council.
In like manner, briefs from amici arguing that the PTAB should deny the Tribe's petition make many of the same arguments, the most prominent of which is that IPRs are proceedings in rem ("one against the world") and not in personem, and that sovereign immunity does not bar these proceedings, analogous to bankruptcies. The brief from the BSA/The Software Alliance argues that the IPR provisions of the AIA are "statutes of general applicability" (an argument also made in the brief from Askeladden LLC) subject to what they call the Tuscarora rule, from Fed. Power Comm'n v. Tuscarora Indian Nation, 362 U.S. 99, 116 (1960), that such statutes apply unless the application is related to or would abrogate some aspect of tribal autonomy or self-governance. This brief, as well as most of the other briefs on this side of the question, take the language of the Supreme Court's Cuozzo opinion (see "Cuozzo Speed Technologies LLC v. Lee (2016)") that IPRs are not "mini-trials" or in the form of a lawsuit but rather specialized agency proceedings (despite the rhetoric from supporters of these proceedings that they are indeed intended to be in the form of an adversarial proceeding as a substitute for litigation).
The policy argument in this brief, as well as others in favor of denial, is that "the IPR process serves to advance the important public policy—rooted in the Constitution's Patent and Copyright Clause—of ensuring that the American public and economy are not burdened by unwarranted monopolies." The brief also analogizes the IPR proceedings to other agency actions, including NLRA (NLRB v. Little River Band of Ottawa Indians Tribal Gov't, 788 F.3d 537, 555–56 (6th Cir. 2015)), OSHA (Menominee Tribal Enters. v. Solis, 601 F.3d 669, 674 (7th Cir. 2010); Reich v. Mashantucket Sand & Gravel, 95 F.3d 174, 177–82 (2d Cir. 1996)), ERISA (Smart v. State Farm Ins. Co., 868 F.2d 929, 932–36 (7th Cir. 1989)), and EEOC (EEOC v. Fond du Lac Heavy Equip. & Constr. Co., 986 F.2d 246, 249 (8th Cir. 1993).
The brief from the Association for Accessible Medicines makes similar arguments, characterizing their "significant interest in ensuring that brand-name drug manufacturers are not allowed to abuse the U.S. patent system by attempting to invoke rented Native American tribal immunity to frustrate the Board's authority to review erroneously issued patents." This brief emphasizes the purported "sham" nature of the assignments and echoing the in rem arguments made by other amici. This brief also analogizes to other cases involving private property and inapplicability of sovereign immunity, all under state law (Anderson & Middleton Lumber Co. v. Quinault Indian Nation, 929 P.2d 379, 381 (Wash. 1996); Smale v. Noretep, 208 P.3d 1180, 1180 (Wash. Ct. App. 2009); Cass Cnty. Joint Water Res. Dist. v. 1.43 Acres of Land, 643 N.W.2d 685, 688 (N.D. 2002)). Their contribution to the policy argument is to assert that:
[A]llowing a drug company patent owner to force an IPR to halt on the eve of an IPR hearing, by paying tens of millions of dollars to rent tribal immunity, would reward bad-faith behavior. It would also provide other holders of weak but extremely profitable patents with a roadmap for shielding their patents from review while wasting the valuable resources of their competitors and the Board.
And:
Prior Board decisions involving sovereign state entities are of limited relevance here because none of those cases involved last-minute bad-faith transfers specifically intended to divest the Board of its authority to take a second look at an issued patent—actions that cannot be squared with equity or good conscience.
The brief from the Software and Information Industry Association characterizes IPRs as a compromise intended to improve patent quality and "restore confidence in the presumption of validity afforded patents issued by the Patent and Trademark Office." The brief (as is also argued in the brief from Askeladden LLC) relies heavily on Judge Bryson's decision granting the Tribe's motion to join ANDA litigation involving some of these same patents:
The essence of the matter is this: Allergan purports to have sold the patents to the tribe, but in reality it has paid the Tribe to allow Allergan to purchase—or perhaps more precisely, to rent—the Tribe's sovereign immunity in order to defeat the pending IPR proceedings in the PTO . . . . If that ploy succeeds, any patentee facing IRO proceedings would presumably be able to employ the same artifice. In short, Allergan's tactic, if successful, could spell the end of the PTO's IPR program, which was a central component of the America Invents Act of 2011.
According to this brief, if the Board does not deny this motion, tribal immunity will be asserted in most if not all IPRs (a fear not without some justification; see "More Instances of Tribal Sovereign Immunity Shielding Patents from PTAB Invalidation").
A brief from the High Tech Inventors Alliance, Computer & Communications Industry Association, and Internet Association repeats the argument that patent laws are law of general applicability from which sovereign immunity provides no protection, basing this characterization in part because "[private petitioners] cannot compel the Patent Office to conduct a review and their ongoing participation is unnecessary to complete one" (another argument also made in the brief from Askeladden LLC). According to this brief, "[i]nter partes review is thus 'less like a judicial proceeding and more like a specialized agency proceeding' in which third parties that petition for review 'need not have a concrete stake in the outcome; indeed, they may lack constitutional standing,'" citing Cuozzo Speed Techs., LLC v. Lee, 136 S. Ct. 2131, 2143-44 (2016). According to this brief (in an argument made nowhere else), "tribes that own patents must, like all other patentees, pay the fees to maintain the patent, or else the patent will expire. See 35 U.S.C. § 41(b). The condition that the Patent Office may reexamine or review the patent is no different."
At least one of the amici arguing for denial of the Tribe's motion has a specific interest in the decision: Deva Holding A.S. is a Turkish company, in an "early stage" lawsuit against Allergan. This amicus argues that substitution is unnecessary (and thus tribal sovereign immunity does not prevent IPRs to proceed) because the Tribe and Allergan have identical interests based on litigation conduct, and so Allergan can represent the Tribe's interests — "actions speak louder than words." In this argument, the purported inconsistency of positions before the Board and the Court, citing a specific September 8, 2017 letter to the Court, mandate this conclusion and provide sufficient basis to deny the Tribe's motion.
In addition to its recommendation that the Board would be better to certify the question to the PTO Director, the amicus collegii brief from the Public Knowledge and Electronic Frontier Foundation cites Hazel-Atlas Glass Co. v. Hartford-Empire Co. and Precision Instrument Mfg. Co. v. Auto. Maint. Mach. Co. (inequitable conduct) for the "patents in the public interest" concept, saying that the PTAB is an "agency of public justice" and draws analogies to Supreme Court decisions precluding limitations on challenging validity, including Laboratories v. University of Illinois Foundation, Medimmune v. Genentech, Cardinal Chem. Co. v. Morton Int'l, Inc., and Lear v Atkins.
The brief from James R. Major, D. Phil., opposes the tribe's motion on contractual grounds, arguing that Tribe's patent rights outside the grantback to Allergan are illusory and thus the assignment nugatory, and that the Tribe has no independent interests to assert. These rights include "research, scholarly use, teaching, education, patient care incidental to the foregoing, sponsored research for itself and in collaborations with Non-Commercial Organizations . . . ." Dr. Major's point is that the Tribe already had these rights because they were immune from patent infringement liability due to the exercise of sovereign immunity:
In sum, any infringement suit that the Tribe has the first right to bring would fail. Therefore, the right of the Tribe to enforce the Licensed Patents in infringement suits unrelated to a Generic Equivalent is nugatory.
In Dr. Major's view, "the Agreement has stripped the Tribe of any meaningful rights in the Licensed Patents."
The time for filing amicus briefs in this matter has passed, and the Board is scheduled to render its decision by spring.
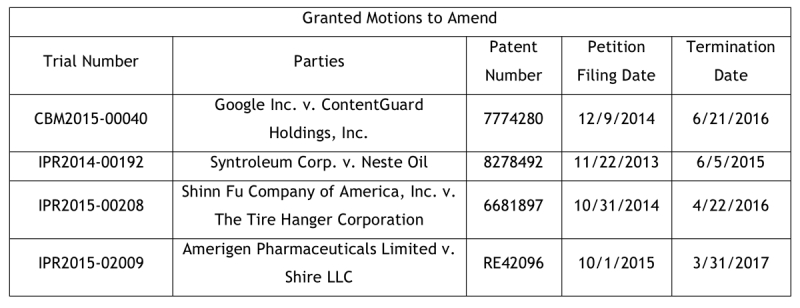
 On January 12, 2018, the U.S. Patent and Trademark Office's Patent Trial and Appeal Board announced that it had updated its Motion to Amend Study to include all AIA trials through the end of Fiscal Year 2017 (which ended on September 30, 2017). The original study included data through April 30, 2016. At that time, only two motions to amend had been granted outright, with another four granted-in-part. This study was updated last year with data through May 31, 2017. At that time, two additional motions to amend had been granted outright, with an additional four granted-in-part. The current study includes no new motions to amend granted outright, but there are two more that have been granted-in-part. In total, fourteen motions to amend had been granted at least in part as of September 30, 2017 (out of a total of 275 completed trials in which a motion to amend was filed). Of course, the cut off for this study misses any impact of the October 4, 2017 Aqua Products, Inc. v. Matal decision. We, however, believe that no additional motions to amend have been granted so far this fiscal year.
On January 12, 2018, the U.S. Patent and Trademark Office's Patent Trial and Appeal Board announced that it had updated its Motion to Amend Study to include all AIA trials through the end of Fiscal Year 2017 (which ended on September 30, 2017). The original study included data through April 30, 2016. At that time, only two motions to amend had been granted outright, with another four granted-in-part. This study was updated last year with data through May 31, 2017. At that time, two additional motions to amend had been granted outright, with an additional four granted-in-part. The current study includes no new motions to amend granted outright, but there are two more that have been granted-in-part. In total, fourteen motions to amend had been granted at least in part as of September 30, 2017 (out of a total of 275 completed trials in which a motion to amend was filed). Of course, the cut off for this study misses any impact of the October 4, 2017 Aqua Products, Inc. v. Matal decision. We, however, believe that no additional motions to amend have been granted so far this fiscal year.
 Tellingly, the third chart shows the outcome of the 170 decided motions to amend in which substitute claims were proposed. In 156 of these cases, or 92%, the Board denied the motion. The four motions to amend that were granted outright represent 2% of all motions to amend with substitute claims that were decided, and represent a much smaller percentage of all motions to amend. The ten motions to amend that were granted-in-part represented 6% of the total decided. The Study included a separate spreadsheet that contained the data from the 275 trials. This spreadsheet explains in which post-issuance proceedings that motions to amend were granted. The following tables were compiled from this data:
Tellingly, the third chart shows the outcome of the 170 decided motions to amend in which substitute claims were proposed. In 156 of these cases, or 92%, the Board denied the motion. The four motions to amend that were granted outright represent 2% of all motions to amend with substitute claims that were decided, and represent a much smaller percentage of all motions to amend. The ten motions to amend that were granted-in-part represented 6% of the total decided. The Study included a separate spreadsheet that contained the data from the 275 trials. This spreadsheet explains in which post-issuance proceedings that motions to amend were granted. The following tables were compiled from this data: