By Kevin E. Noonan –
 In an extensive, 42-page per curiam opinion, the Patent Trial and Appeal Board (PTAB) surprised no one last Friday by denying the St. Regis Mohawk Tribe's motion to terminate several inter partes review proceedings based on the Tribe's assertion of tribal sovereign immunity. In an effort to avoid undue suspense, the Board provided a pithy summary of its decision at the beginning of the opinion:
In an extensive, 42-page per curiam opinion, the Patent Trial and Appeal Board (PTAB) surprised no one last Friday by denying the St. Regis Mohawk Tribe's motion to terminate several inter partes review proceedings based on the Tribe's assertion of tribal sovereign immunity. In an effort to avoid undue suspense, the Board provided a pithy summary of its decision at the beginning of the opinion:
Upon consideration of the record, and for the reasons discussed below, we determine the Tribe has not established that the doctrine of tribal sovereign immunity should be applied to these proceedings. Furthermore, we determine that these proceedings can continue even without the Tribe's participation in view of Allergan's retained ownership interests in the challenged patents. The Tribe's Motion is therefore denied.
The Board's decision is consistent with Board rule, 37 C.F.R. § 41.121(b), that the proponent of a motion bears the burden of establishing their right to the relief requested. It is also consistent with the Board's evident policy position from recent decisions (see Ericsson Inc. v. Regents of the University of Minnesota, Case IPR2017-01186 (PTAB Dec. 19, 2017) (Paper 14) ("Ericsson"), and LSI Corp. v. Regents of the University of Minnesota, Case IPR2017-01068 (PTAB Dec. 19, 2017) (Paper 19) ("LSI")) that, once instituted, it will not terminate post-grant review proceedings (absent settlement, and perhaps not even then) on sovereign immunity grounds, unless the Director, Congress, or the courts compel them to do so. And the opinion, in its recitation of the relevant facts, provided the panel with another opportunity to display its pique (see "The PTAB Strikes Back — Issues Order Prohibiting St. Regis Mohawk Tribe from Filing Any Additional Papers in IPR") at the Tribe's representatives, stating:
The Tribal Council Resolution states that the Tribe was approached by the law firm Shore Chan DePumpo LLP "to engage in new business activities related to existing and emerging technologies, which may include the purchase and enforcement of intellectual property rights, known as the 'Intellectual Property Project."
In setting forth the legal basis for its decision, the Board recognized that "[a]s a matter of federal law, an Indian tribe is subject to suit only where Congress has authorized the suit or the tribe has waived its immunity," citing Kiowa Tribe of Okla. v. Mfg. Techs., Inc., 523 U.S. 751, 754 (1998). Nevertheless, the Board applying at least Rule 121(b), refuges in the fact that there is no "controlling precedent or statutory basis" for applying sovereign immunity to IPR. Acknowledging that this is a "case of first impression" (making its mention of a lack controlling precedent at least inherently redundant), the opinion distinguishes Federal Maritime Commission v. South Carolina State Ports Authority, 535 U.S. 743 (2002) ("FMC"), on the grounds that FMC was concerned with State sovereign immunity which is distinct from tribal sovereign immunity by being protected by the Eleventh Amendment and not being subject to abrogation by Congress as tribal immunity is. The Board states that:
The Tribe and its supporting amici, however, have not pointed to any federal court or Board precedent suggesting that FMC's holding with respect to state sovereign immunity can or should be extended to an assertion of tribal immunity in similar federal administrative proceedings.
The opinion also distinguished State sovereign immunity decisions that recognized the immunity for state universities acting as an "arm of the state" (Covidien LP v. Univ. of Fla. Research Found. Inc., Case IPR2016-01274 (PTAB Jan. 25, 2017) (Paper 21); Neochord, Inc. v. Univ. of Md., Case IPR2016-00208 (PTAB May 23, 2017) (Paper 28); and Reactive Surfaces Ltd, LLP v. Toyota Motor Corp., Case IPR2016-01914, (PTAB July 13, 2017) (Paper 36)) on this basis.
The opinion also disregards analogous decisions of other agencies ("[w]e are not bound by those agency decisions") and also curiously distinguishes Bodi v. Shingle Springs Band of Miwok Indians, 832 F.3d 1011, 1021 (9th Cir. 2016), and Contour Spa at the Hard Rock, Inc. v. Seminole Tribe of Fla., 692 F.3d 1200, 1201 (11th Cir. 2012), both cases having declined to Lapides v. Bd. of Regents of the Univ. Sys. of Ga., 535 U.S. 613 (2002), concerning waiver of state's sovereign immunity based on litigation conduct, to tribal immunity). The opinion expressly relies on the Board's precedential decision in Athena Automation Ltd. v. Husky Injection Molding Sys. Ltd., Case IPR2013-00290, slip op. at 12–13 (PTAB Oct. 25, 2013), to assert that in contrast to 19 U.S.C. 1337(c) where "[a]ll legal and equitable defenses may be presented' in International Trade Commission (ITC) investigations" that is not the case for 35 U.S.C. § 311(a).
The Board went further than deigning not to grant the Tribe's motion; no doubt in part to stem the expected tide of similar assignment and licensing arrangements, the Board declared that tribal sovereign immunity does not apply to IPR proceedings. The basis for this decision represents somewhat of a turning of the tables: rather than addressing the Tribe's point that sovereign immunity should lie unless Congress has specifically abrogated it, the Board decision characterizes IPR proceedings as being the product of a "generally applicable statute" that does not implicate sovereign immunity concerns or protections. This determination is based on the applicability (or risk) of IPR proceedings against any patent "regardless of ownership," their being "subject to the conditions and requirements of [the Patent Act]." The opinion cites authority limiting application of tribal immunity for "laws of general applicability," including Donovan v. Coeur d'Alene Tribal Farm, 751 F.2d 1113, 1116 (9th Cir. 1985) (quoting U.S. v. Farris, 624 F.2d 890, 893–94 (9th Cir. 1980), to the effect that such laws must affect "exclusive rights of self-governance in purely intramural matters"; involve "abrogat[ing] rights guaranteed by Indian treaties"; or that there be legislative history that supports a conclusion that Congress did not intend the law to apply to an Indian tribe. Of course, in this instance none of these apply.
Portions of the opinion appear to imply that IPRs are an assertion of "superior sovereign powers" of the Federal government that tribal immunity cannot thwart, citing Quileute Indian Tribe v. Babbitt, 18 F.3d 1456, 1459 (9th Cir. 1994), and that tribal immunity has not prevented assertion by the Federal government of other administrative proceedings, citing Consumer Fin. Prot. Bureau v. Great Plains Lending, LLC, 846 F.3d 1049, 1058 (9th Cir. 2017), and Menominee Tribal Enters. v. Solis, 601 F.3d 669, 674 (7th Cir. 2010). In this the Board disregards at least the distinction that in those cases the action was by the government represented by a government lawyer. The Board also gives little credence to the argument that in an IPR a private third party remains involved, stating that "a private entity's continued involvement as a party in a federal administrative proceeding does not necessarily entitle a tribal entity to assert its immunity in the proceeding."
The opinion contains additional discussion of sovereign immunity as a "common law" concept granting immunity from suit, on the grounds that an IPR is not that sort of suit, and also notes the public interest inherent in IPRs, citing Cuozzo Speed Techs., LLC v. Lee, 136 S. Ct. 2131, 2144 (2016). While not basing its decision on the distinction made by several amici (see "Amicus Briefs Filed in Mohawk Tribe's Motion to Dismiss IPRs") between actions in personam and in rem (to the effect that IPRs as an in rem proceeding are not subject to an assertion of sovereign immunity), the Board does note that it has the authority to hold IPR proceedings in cases where the patent owner does not appear and the ability for the Board to continue an IPR even if the parties settle, stating "[t]he Board's authority to proceed without the parties' participation underscores its independent role in ensuring the correctness of granting patentable claims."
Finally, the Board says that the IPR can continue with Allergan alone (i.e., without the Tribe's participation), based on the rights in the patents granted by the license(s) and agreeing with Petitioners that Allergan remains the "patent owner" despite the assignment (while taking pains not to assert that the ownership transfer is a sham). The opinion cites the Federal Circuit for holding that the "party that has been granted all substantial rights under the patent is considered the owner regardless of how the parties characterize the transaction that conveyed those rights," citing Speedplay, Inc. v. Bebop, Inc., 211 F.3d 1245, 1250 (Fed. Cir. 2000), and also Alfred E. Mann Found. for Sci. Research v. Cochlear Corp., 604 F.3d 1354, 1358–59 (Fed. Cir. 2010), for the proposition that "[a] patent owner may transfer all substantial rights in the patents-in-suit, in which case the transfer is tantamount to an assignment of those patents to the exclusive licensee." The Board panel recognizes the following analysis of "the rights transferred and the rights retained under the license agreement, including:
(1) the nature and scope of the right to bring suit; (2) the exclusive right to make, use, and sell products or services under the patent; (3) the scope of the licensee's right to sublicense; (4) the reversionary rights to the licensor following termination or expiration of the license; (5) the right of the licensor to receive a portion of the proceeds from litigating or licensing the patent; (6) the duration of the license rights; (7) the ability of the licensor to supervise and control the licensee's activities; (8) the obligation of the licensor to continue paying maintenance fees; and (9) any limits on the licensee's right to assign its interests in the patent.
Azure Networks, LLC v. CSR PLC, 771 F.3d 1336, 1343 (Fed. Cir. 2014) (vacated on other grounds); see also Alfred E. Mann Found. for Sci. Research v. Cochlear Corp., 604 F.3d at 1360–61. The opinion compares the Allergan-Mohawk Tribe license using these factors; here is the scorecard:
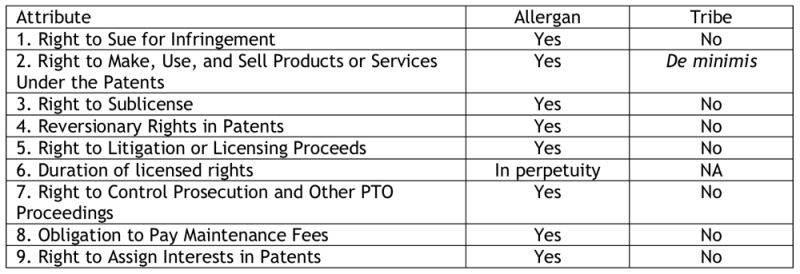
With regard to the first factor, the opinion notes that "[e]ach of the challenged patents is listed in the FDA's 'Orange Book.' . . . As such, we find that any viable infringement allegation for the challenged patents would have to necessarily be limited to drug products that require FDA approval . . ." and "[b]ased on the record before us, we find that the Tribe has not retained anything more than an illusory or superficial right to sue for infringement of the challenged patents." The Tribe's rights to make, use, and sell the patented invention was negligible in the Board's view, because "the claims of the challenged patents are directed to pharmaceutical compositions and methods used to treat human medical conditions, [and under these circumstances] we find Allergan's exclusive right to exploit the challenged patents 'or all FDA-approved uses in the United States' to be a substantial right." Allergan's rights under the license are "co-extensive in scope" with the claimed invention and the license thus does not limit Allergan's rights "in any meaningful sense." The opinion further notes that the Tribe's right to sublicense and assign its patents to a third party are restricted, and that "[w]ith respect to 'Contested PTO Proceedings' in particular, which include these inter partes review proceedings, the License provides that '[a]s between the Parties, Allergan shall have . . . the first right, but not the obligation, to defend and control the defense of the validity, enforceability and patentability of the Licensed Patents in such Contested PTO Proceeding.'"
In toto, the Board concluded from this analysis that:
We find these provisions to be significant restrictions on the Tribe's purported ownership rights. "The right to dispose of an asset is an important incident of ownership, and such a restriction on that right is a strong indicator" of whether a license agreement transferred all substantial rights under the patent.
and:
In sum, upon considering the relevant License terms, we find that Allergan obtained all substantial rights in the challenged patents. The Tribe points out that Allergan executed an assignment of the challenged patents to the Tribe, and this assignment was recorded at the PTO. . . . As recognized by the Tribe, however, a recordation of a patent assignment only creates a rebuttable presumption regarding ownership. See SiRF Tech., Inc. v. Int'l Trade Comm'n, 601 F.3d 1319, 1328 (Fed. Cir. 2010).
Importantly, the opinion also states that "[i]n reaching this conclusion, we do not comment on whether the License and the other agreements between the Tribe and Allergan constitute a 'sham' transaction, nor do we need to decide whether the agreements are otherwise improper under the law."
As a consequence, the Board further held that the Tribe is not an indispensible party under Federal Rule of Civil Procedure. This is important because Supreme Court precedent holds, in Republic of Philippines v. Pimentel, that:
"[a] case may not proceed when a required-entity sovereign is not amenable to suit . . . where sovereign immunity is asserted, and the claims of the sovereign are not frivolous, dismissal of the action must be ordered where there is a potential for injury to the interests of the absent sovereign. 553 U.S. 851, 867 (2008) and "once a tribunal recognizes that an assertion of sovereign immunity is 'not frivolous,' it is 'error' for the tribunal to proceed further to address the merits" (Pimentel, 553 U.S. at 864).
The Board distinguished Pimentel on its facts (involving a foreign sovereign), and on the need for courts to assess based on Rule 19(b) rather than the mere non frivolous assertion of sovereign immunity, citing Univ. of Utah v. Max-Planck-Gesellschaft Zur Forderung Der Wissenschaften E.V., 734 F.3d 1315, 1326 (Fed. Cir. 2013). The Board noted that its proceedings are not bound by the FRCP. Even if the Board applied Rule 19, the opinion states the Tribe is not an indispensible party:
Allergan has at least an identical interest to the Tribe—if not more of an interest as the effective patent owner for the reasons discussed above—in defending the challenged patents. Thus, we do not find that the Tribe will be significantly prejudiced in relation to the merits of the patentability challenges in these proceedings if it chooses not to participate based on its alleged tribal immunity because Allergan will be able to adequately represent any interests the Tribe may have in the challenged patents.
The original panel rendered this decision, putting at rest for the moment any inference of possible impropriety at least implied by the Tribe's motion for discovery on this issue (among others); see "Skeptical St. Regis Mohawk Tribe Requests Discovery Regarding Panel Selection Circumstances"). But this clearly is not the end of the matter; it remains to be seen whether the Board quickly follows with a Final Written Decision on the merits (due within the statutorily mandated twelve months on March 31st) or whether the Tribe files a Notice of Appeal on this issue (although the availability of such an appeal, likely to be considered interlocutory in nature, is uncertain; it is unlikely the PTAB will certify the question or otherwise facilitate an early appeal). And there remain efforts in Congress (inter alia, by Senator Claire McCaskill; see "A Bill to abrogate sovereign immunity of Indian tribes as a defense in inter partes review of patents") to abrogate by statute assertion of sovereign immunity by Indian tribes in patent cases. Stayed tuned.
 In a memorandum distributed last week to the U.S. Patent and Trademark Office patent examining corps, Robert Bahr, the Deputy Commissioner for Patent Examination Policy, sought to clarify the Office's guidance regarding the written description requirement of 35 U.S.C. § 112(a), and in particular, the examination of claims directed to antibodies for compliance with the written description requirement. The memorandum, entitled "Clarification of Written Description Guidance For Claims Drawn to Antibodies and Status of 2008 Training Materials," is divided into three sections, which address the Federal Circuit's recent decision in Amgen Inc. v. Sanofi, the Written Description Training Materials released by the Office in 2008, and the Office's current guidance on the written description requirement.
In a memorandum distributed last week to the U.S. Patent and Trademark Office patent examining corps, Robert Bahr, the Deputy Commissioner for Patent Examination Policy, sought to clarify the Office's guidance regarding the written description requirement of 35 U.S.C. § 112(a), and in particular, the examination of claims directed to antibodies for compliance with the written description requirement. The memorandum, entitled "Clarification of Written Description Guidance For Claims Drawn to Antibodies and Status of 2008 Training Materials," is divided into three sections, which address the Federal Circuit's recent decision in Amgen Inc. v. Sanofi, the Written Description Training Materials released by the Office in 2008, and the Office's current guidance on the written description requirement.










